Cancer Res Treat.
2016 Apr;48(2):546-552. 10.4143/crt.2015.015.
Efficacy of Imatinib in Patients with Platelet-Derived Growth Factor Receptor Alpha-Mutated Gastrointestinal Stromal Tumors
- Affiliations
-
- 1Department of Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. ykkang@amc.seoul.kr
- KMID: 2454331
- DOI: http://doi.org/10.4143/crt.2015.015
Abstract
- PURPOSE
The incidence of gastrointestinal stromal tumors (GISTs) harboring platelet-derived growth factor receptor alpha (PDGFRA) mutations is low, therefore further investigation of the efficacy of imatinib in this subgroup was needed.
MATERIALS AND METHODS
Patients with PDGFRA-mutant GISTs who received imatinib as primary therapy for advanced disease between January 2000 and June 2012 were identified from the GIST registry of Asan Medical Center, Seoul, Korea.
RESULTS
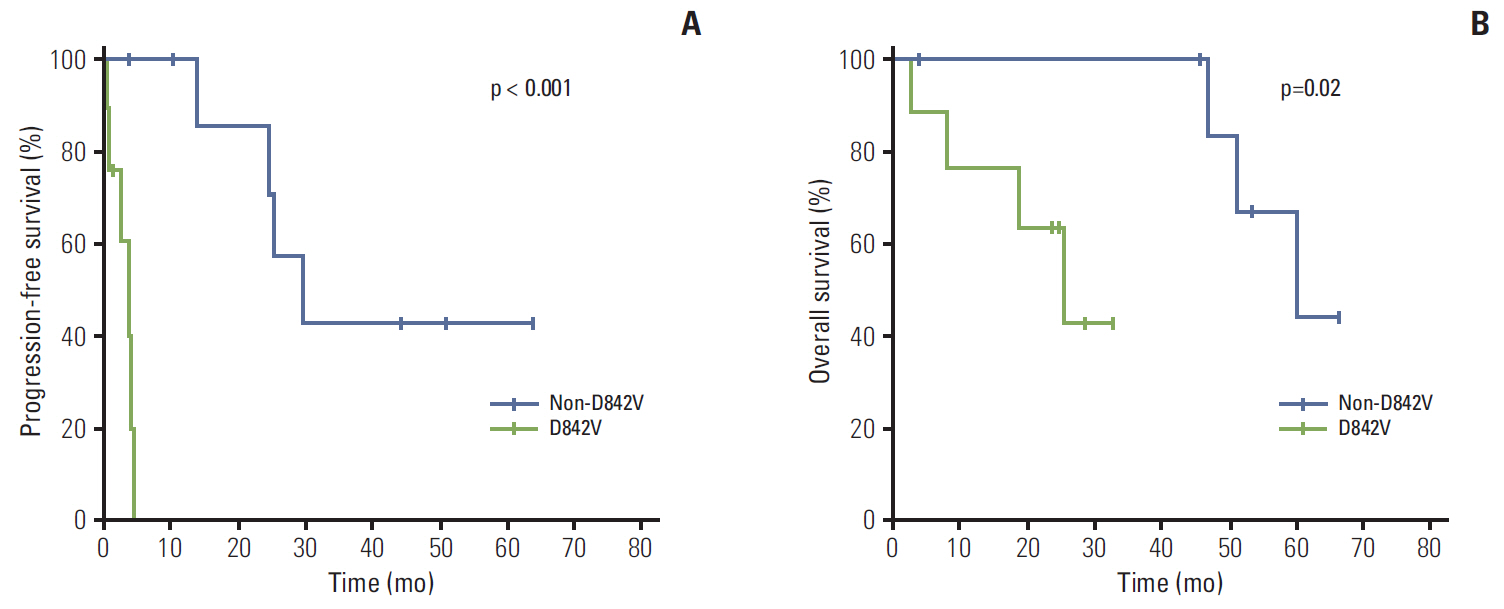
KIT and PDGFRA genotyping in 823 patients identified 18 patients (2%) with PDGFRA mutations who were treated with first-line imatinib. Exon 18 D842V substitution, non-D842V exon 18 mutations, and exon 12 mutations were detected in nine (50%), four (22%), and five (28%) patients, respectively. Objective response rate differed significantly between patients with the D842V mutation and those with non-D842V mutations (0% [0/5] vs. 71% [5/7], p=0.03). In all patients, median progression-free survival (PFS) and overall survival (OS) was 24.8 months (95% confidence interval [CI], 0.0 to 57.2) and 51.2 months (95% CI, 37.1 to 65.3), respectively. Significantly, poorer PFS was observed for patients with D842V-mutant GISTs than those with non-D842V PDGFRA-mutant GISTs: median 3.8 months (95% CI, 1.4 to 6.3) versus 29.5 months (95% CI, 18.3 to 40.7) (p < 0.001). Patients with the D842V mutation had poorer OS than those with non-D842V PDGFRA mutations: median 25.2 months (95% CI, 12.7 to 37.8) versus 59.8 months (95% CI, 43.0 to 76.5) (p=0.02).
CONCLUSION
Imatinib is active against non-D842V PDGFRA-mutant GISTs, whereas GISTs harboring the D842V mutation are primarily resistant to imatinib.
MeSH Terms
Figure
Reference
-
References
1. Lasota J, Miettinen M. Clinical significance of oncogenic KIT and PDGFRA mutations in gastrointestinal stromal tumours. Histopathology. 2008; 53:245–66.2. Wozniak A, Rutkowski P, Piskorz A, Ciwoniuk M, Osuch C, Bylina E, et al. Prognostic value of KIT/PDGFRA mutations in gastrointestinal stromal tumours (GIST): Polish Clinical GIST Registry experience. Ann Oncol. 2012; 23:353–60.
Article3. Emile JF, Brahimi S, Coindre JM, Bringuier PP, Monges G, Samb P, et al. Frequencies of KIT and PDGFRA mutations in the MolecGIST prospective population-based study differ from those of advanced GISTs. Med Oncol. 2012; 29:1765–72.
Article4. Cassier PA, Ducimetiere F, Lurkin A, Ranchere-Vince D, Scoazec JY, Bringuier PP, et al. A prospective epidemiological study of new incident GISTs during two consecutive years in Rhone Alpes region: incidence and molecular distribution of GIST in a European region. Br J Cancer. 2010; 103:165–70.5. Demetri GD, von Mehren M, Blanke CD, Van den Abbeele AD, Eisenberg B, Roberts PJ, et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med. 2002; 347:472–80.
Article6. Verweij J, Casali PG, Zalcberg J, LeCesne A, Reichardt P, Blay JY, et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: randomised trial. Lancet. 2004; 364:1127–34.
Article7. Blanke CD, Rankin C, Demetri GD, Ryan CW, von Mehren M, Benjamin RS, et al. Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the KIT receptor tyrosine kinase: S0033. J Clin Oncol. 2008; 26:626–32.
Article8. Ryu MH, Kang WK, Bang YJ, Lee KH, Shin DB, Ryoo BY, et al. A prospective, multicenter, phase 2 study of imatinib mesylate in korean patients with metastatic or unresectable gastrointestinal stromal tumor. Oncology. 2009; 76:326–32.
Article9. Heinrich MC, Owzar K, Corless CL, Hollis D, Borden EC, Fletcher CD, et al. Correlation of kinase genotype and clinical outcome in the North American Intergroup Phase III Trial of imatinib mesylate for treatment of advanced gastrointestinal stromal tumor: CALGB 150105 Study by Cancer and Leukemia Group B and Southwest Oncology Group. J Clin Oncol. 2008; 26:5360–7.
Article10. Debiec-Rychter M, Sciot R, Le Cesne A, Schlemmer M, Hohenberger P, van Oosterom AT, et al. KIT mutations and dose selection for imatinib in patients with advanced gastrointestinal stromal tumours. Eur J Cancer. 2006; 42:1093–103.
Article11. Gastrointestinal Stromal Tumor Meta-Analysis Group (MetaGIST). Comparison of two doses of imatinib for the treatment of unresectable or metastatic gastrointestinal stromal tumors: a meta-analysis of 1,640 patients. J Clin Oncol. 2010; 28:1247–53.12. Corless CL, Schroeder A, Griffith D, Town A, McGreevey L, Harrell P, et al. PDGFRA mutations in gastrointestinal stromal tumors: frequency, spectrum and in vitro sensitivity to imatinib. J Clin Oncol. 2005; 23:5357–64.
Article13. Cassier PA, Fumagalli E, Rutkowski P, Schoffski P, Van Glabbeke M, Debiec-Rychter M, et al. Outcome of patients with platelet-derived growth factor receptor alpha-mutated gastrointestinal stromal tumors in the tyrosine kinase inhibitor era. Clin Cancer Res. 2012; 18:4458–64.
Article14. Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000; 92:205–16.15. Ryu MH, Lee JL, Chang HM, Kim TW, Kang HJ, Sohn HJ, et al. Patterns of progression in gastrointestinal stromal tumor treated with imatinib mesylate. Jpn J Clin Oncol. 2006; 36:17–24.
Article16. Kang HJ, Ryu MH, Kim KM, Park YS, Choi J, Ryoo BY, et al. Imatinib efficacy by tumor genotype in Korean patients with advanced gastrointestinal stromal tumors (GIST): The Korean GIST Study Group (KGSG) study. Acta Oncol. 2012; 51:528–36.
Article17. Subramanian S, West RB, Corless CL, Ou W, Rubin BP, Chu KM, et al. Gastrointestinal stromal tumors (GISTs) with KIT and PDGFRA mutations have distinct gene expression profiles. Oncogene. 2004; 23:7780–90.
Article18. Miettinen M, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol. 2005; 29:52–68.19. Gramza AW, Corless CL, Heinrich MC. Resistance to tyrosine kinase inhibitors in gastrointestinal stromal tumors. Clin Cancer Res. 2009; 15:7510–8.
Article20. Kang YK, Kang HJ, Kim KM, Sohn T, Choi D, Ryu MH, et al. Clinical practice guideline for accurate diagnosis and effective treatment of gastrointestinal stromal tumor in Korea. Cancer Res Treat. 2012; 44:85–96.
Article21. Heinrich MC, Griffith D, McKinley A, Patterson J, Presnell A, Ramachandran A, et al. Crenolanib inhibits the drug-resistant PDGFRA D842V mutation associated with imatinib-resistant gastrointestinal stromal tumors. Clin Cancer Res. 2012; 18:4375–84.
Article22. Heinrich MC, Maki RG, Corless CL, Antonescu CR, Harlow A, Griffith D, et al. Primary and secondary kinase genotypes correlate with the biological and clinical activity of sunitinib in imatinib-resistant gastrointestinal stromal tumor. J Clin Oncol. 2008; 26:5352–9.
Article23. Dewaele B, Wasag B, Cools J, Sciot R, Prenen H, Vandenberghe P, et al. Activity of dasatinib, a dual SRC/ABL kinase inhibitor, and IPI-504, a heat shock protein 90 inhibitor, against gastrointestinal stromal tumor-associated PDGFRAD842V mutation. Clin Cancer Res. 2008; 14:5749–58.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Bowel perforation associated sunitinib therapy for recurred gastric gastrointestinal stromal tumor
- Mutational Analysis of the Epidermal Growth Factor Receptor Gene in Gastrointestinal Stromal Tumors
- A Case of Drug Eruption with Periorbital Edema and Exfoliative Dermatitis Induced by Imatinib Mesylate (STI571, Gleevec(TM))
- The KIT Exon 11 Stop Codon Mutation in Gastrointestinal Stromal Tumors: What Is the Clinical Meaning?
- Imatinib Mesylate (Gleevec(TM))-induced Lichenoid Drug Eruption Improved by Tentative Dose-reduction and Topical Steroid