Immunological Characteristics in Refractory Chronic Rhinosinusitis with Nasal Polyps Undergoing Revision Surgeries
- Affiliations
-
- 1Department of Otorhinolaryngology-Head and Neck Surgery, Soonchunhyang University College of Medicine, Cheonan, Korea.
- 2Department of Otorhinolaryngology-Head and Neck Surgery and Institute of New Frontier Research, Chuncheon Sacred Heart Hospital, Hallym University College of Medicine, Chuncheon, Korea.
- 3Department of Otorhinolaryngology-Head and Neck Surgery, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. kkam97@gmail.com
- 4Department of Otorhinolaryngology-Head and Neck Surgery, Boramae Medical Center, Seoul National University College of Medicine, Seoul, Korea. kicubi@daum.net
- 5Department of Otorhinolaryngology-Head and Neck Surgery, Pyongchon Sacred Heart Hospital, Hallym University College of Medicine, Anyang, Korea.
- 6Department of Otorhinolaryngology-Head and Neck Surgery, Seoul National University College of Medicine, Seoul National University Hospital, Seoul, Korea.
- 7Division of Allergy-Immunology, Department of Internal Medicine, University of South Florida Morsani College of Medicine, Tampa, FL, USA.
- KMID: 2452757
- DOI: http://doi.org/10.4168/aair.2019.11.5.664
Abstract
- PURPOSE
Despite medical and surgical treatments, some cases of nasal polyps (NP) exhibit recidivism. However, the endotype of refractory chronic rhinosinusitis with NP (CRSwNP) remains unclear. Therefore, the objective of this study was to characterize the immunological profile of refractory CRSwNP.
METHODS
The control (n =23), primary NP group (pNP, n =70) and refractory NP group (rNP, n =86) were enrolled in this study. Patients who underwent revision surgeries due to failed maximal medical treatment after primary surgery were defined as the rNP group. A total of 18 inflammatory markers were investigated in nasal tissues using multiplex cytokine assay or enzyme-linked immunosorbent assay.
RESULTS
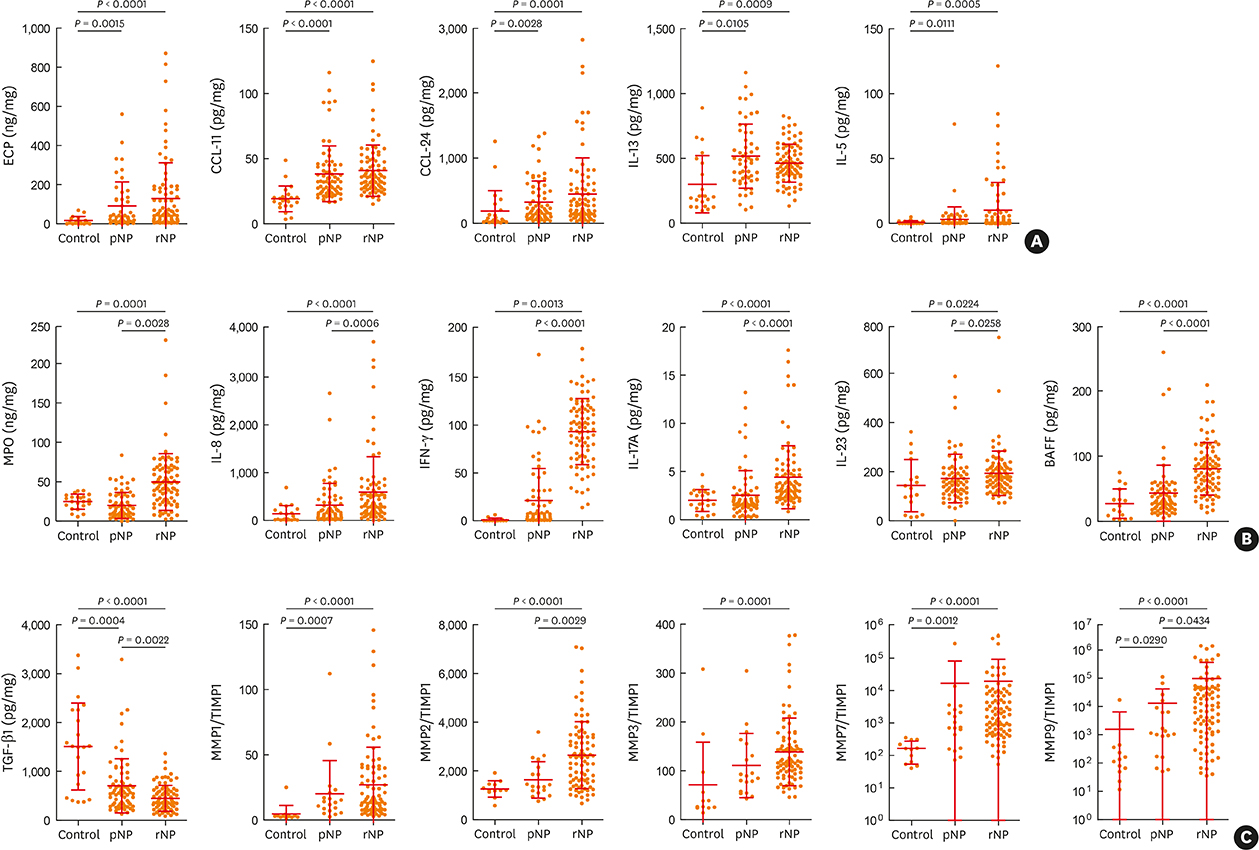
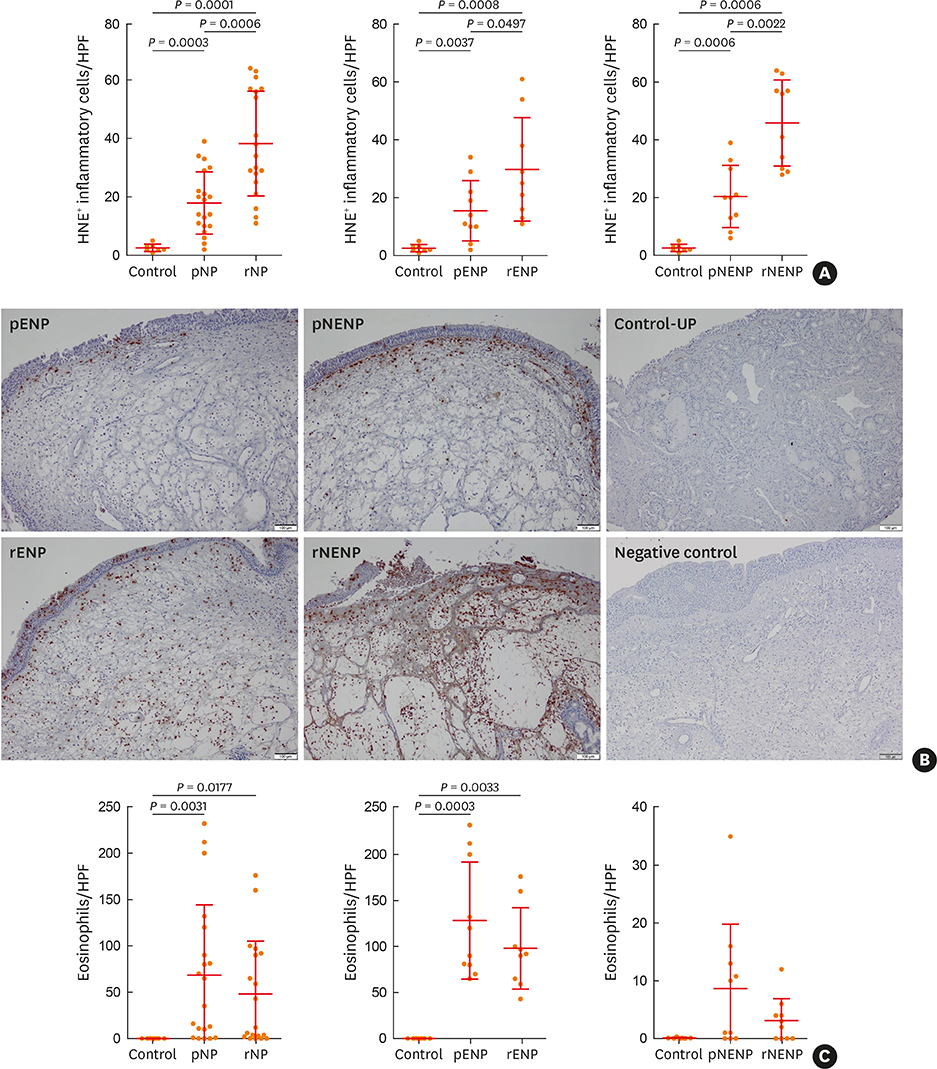
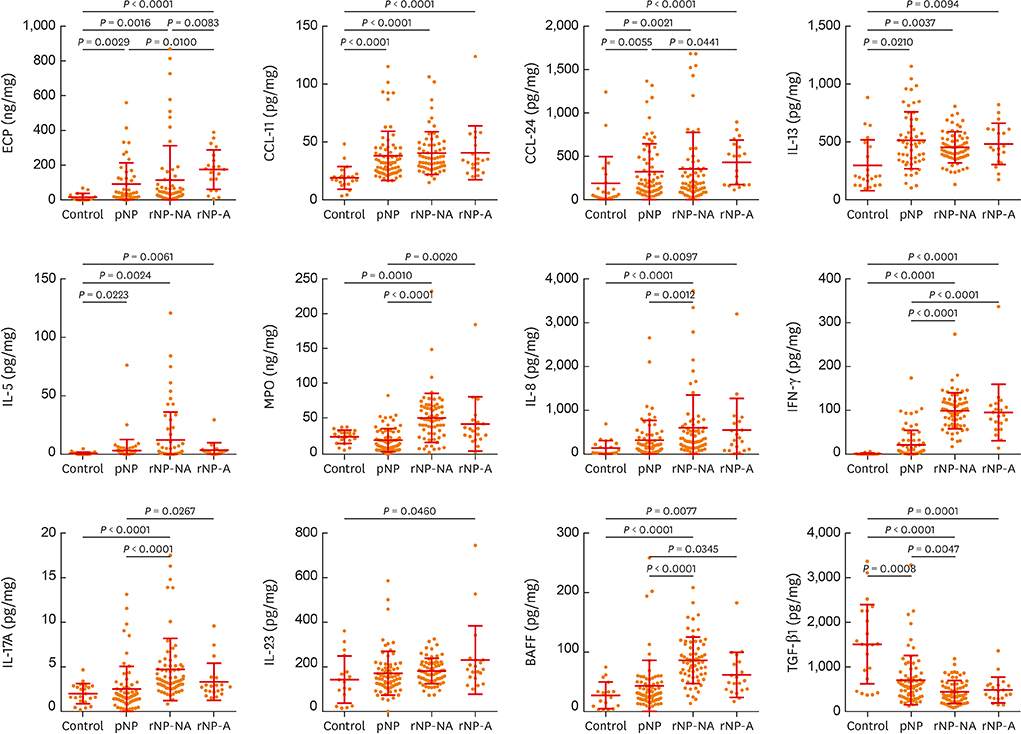
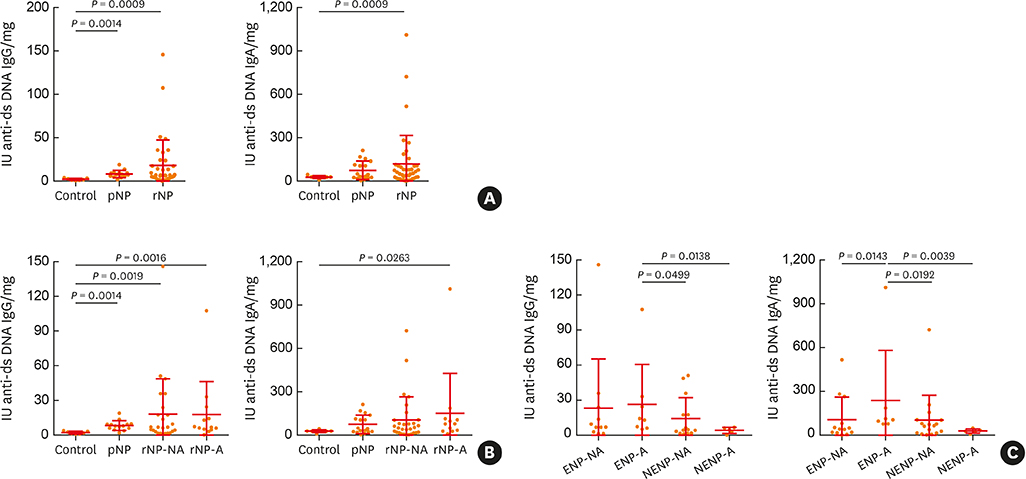
The clinical characteristics of rNP included more extensive disease and worse clinical course after surgery. Additionally, rNP subjects showed higher infection rate (mucopurulence and culture-positive rate), more frequent use of antibiotics and suffered from symptomatic bacterial infection, increased asthma morbidity compared to pNP. Cytokine profile analysis showed that levels of Th17-associated mediators (myeloperoxidase, interleukin (IL)-8, IL-17A and IL-23), B-cell activating factor (BAFF) and Th1 cytokine (interferon-γ) were up-regulated in rNP compared to controls and pNP. Human neutrophil elastase-positive cells were also enhanced in rNP compared with pNP. Upregulation of Th17/Th1mediators and BAFF were observed in rNP, regardless of tissue eosinophilia or asthmatic comorbidity. Interestingly, eosinophilic markers, such as eosinophil cationic protein and C-C motif chemokine ligand 24, were up-regulated in asthmatic rNP compared to pNP and controls. Levels of anti-dsDNA immunoglobulin (Ig) G and IgA were up-regulated in rNP and highest in asthmatic eosinophilic rNP among subtypes of rNP.
CONCLUSIONS
Our results suggest that Th17/Th1-associated mediators and BAFF may play a role and be a potential therapeutic target in refractory CRSwNP. Additionally, eosinophilic markers and autoantibodies may contribute to refractoriness in asthmatic rNP.
MeSH Terms
-
Anti-Bacterial Agents
Asthma
Autoantibodies
B-Cell Activating Factor
Bacterial Infections
Comorbidity
Enzyme-Linked Immunosorbent Assay
Eosinophil Cationic Protein
Eosinophilia
Eosinophils
Humans
Immunoglobulin A
Immunoglobulins
Interleukin-17
Interleukins
Nasal Polyps*
Neutrophils
Sinusitis
Th17 Cells
Up-Regulation
Anti-Bacterial Agents
Autoantibodies
B-Cell Activating Factor
Eosinophil Cationic Protein
Immunoglobulin A
Immunoglobulins
Interleukin-17
Interleukins
Figure
Cited by 3 articles
-
Chinese Society of Allergy and Chinese Society of Otorhinolaryngology-Head and Neck Surgery Guideline for Chronic Rhinosinusitis
Zheng Liu, Jianjun Chen, Lei Cheng, Huabin Li, Shixi Liu, Hongfei Lou, Jianbo Shi, Ying Sun, Dehui Wang, Chengshuo Wang, Xiangdong Wang, Yongxiang Wei, Weiping Wen, Pingchang Yang, Qintai Yang, Gehua Zhang, Yuan Zhang, Changqing Zhao, Dongdong Zhu, Li Zhu, Fenghong Chen, Yi Dong, Qingling Fu, Jingyun Li, Yanqing Li, Chengyao Liu, Feng Liu, Meiping Lu, Yifan Meng, Jichao Sha, Wenyu She, Lili Shi, Kuiji Wang, Jinmei Xue, Luoying Yang, Min Yin, Lichuan Zhang, Ming Zheng, Bing Zhou, Luo Zhang
Allergy Asthma Immunol Res. 2020;12(2):176-237. doi: 10.4168/aair.2020.12.2.176.Elastase-Positive Neutrophils Are Associated With Refractoriness of Chronic Rhinosinusitis With Nasal Polyps in an Asian Population
Dong-Kyu Kim, Jin Youp Kim, Young Eun Han, Joon Kon Kim, Hee-Suk Lim, Kyoung Mi Eun, Seung Koo Yang, Dae Woo Kim
Allergy Asthma Immunol Res. 2020;12(1):42-55. doi: 10.4168/aair.2020.12.1.42.Clinical Characteristics of Chronic Rhinosinusitis With Nasal Polyp According to Histopathological Endotypes and Staining Method for Neutrophilic Polyp Classification and Its Clinical Implication
Hyoyeon Kim, Shin Hyuk Yoo, Kwang Hyun Byun, Ji-Hun Mo
Korean J Otorhinolaryngol-Head Neck Surg. 2024;67(2):79-86. doi: 10.3342/kjorl-hns.2023.00332.
Reference
-
1. Fokkens WJ, Lund VJ, Mullol J, Bachert C, Alobid I, Baroody F, et al. EPOS 2012: European position paper on rhinosinusitis and nasal polyps 2012. A summary for otorhinolaryngologists. Rhinology. 2012; 50:1–12.
Article2. Baguley C, Brownlow A, Yeung K, Pratt E, Sacks R, Harvey R. The fate of chronic rhinosinusitis sufferers after maximal medical therapy. Int Forum Allergy Rhinol. 2014; 4:525–532.
Article3. Wynn R, Har-El G. Recurrence rates after endoscopic sinus surgery for massive sinus polyposis. Laryngoscope. 2004; 114:811–813.
Article4. Bassiouni A, Naidoo Y, Wormald PJ. When FESS fails: the inflammatory load hypothesis in refractory chronic rhinosinusitis. Laryngoscope. 2012; 122:460–466.
Article5. Stein NR, Jafari A, DeConde AS. Revision rates and time to revision following endoscopic sinus surgery: a large database analysis. Laryngoscope. 2018; 128:31–36.
Article6. DeConde AS, Mace JC, Levy JM, Rudmik L, Alt JA, Smith TL. Prevalence of polyp recurrence after endoscopic sinus surgery for chronic rhinosinusitis with nasal polyposis. Laryngoscope. 2017; 127:550–555.
Article7. Hull BP, Chandra RK. Refractory chronic rhinosinusitis with nasal polyposis. Otolaryngol Clin North Am. 2017; 50:61–81.
Article8. Van Zele T, Holtappels G, Gevaert P, Bachert C. Differences in initial immunoprofiles between recurrent and nonrecurrent chronic rhinosinusitis with nasal polyps. Am J Rhinol Allergy. 2014; 28:192–198.
Article9. Tokunaga T, Sakashita M, Haruna T, Asaka D, Takeno S, Ikeda H, et al. Novel scoring system and algorithm for classifying chronic rhinosinusitis: the JESREC study. Allergy. 2015; 70:995–1003.10. Mortuaire G, Leroy X, Gengler I, Chevalier D, Prin L, Picry A. Histopathological classification of refractory chronic rhinosinusitis with nasal polyps. Histol Histopathol. 2015; 30:1447–1454.11. Desrosiers M. Refractory chronic rhinosinusitis: pathophysiology and management of chronic rhinosinusitis persisting after endoscopic sinus surgery. Curr Allergy Asthma Rep. 2004; 4:200–207.
Article12. Wu AW, Ting JY, Platt MP, Tierney HT, Metson R. Factors affecting time to revision sinus surgery for nasal polyps: a 25-year experience. Laryngoscope. 2014; 124:29–33.
Article13. Bassiouni A, Ou J, Rajiv S, Cantero D, Vreugde S, Wormald PJ. Subepithelial inflammatory load and basement membrane thickening in refractory chronic rhinosinusitis with nasal polyposis: a histopathological study. Int Forum Allergy Rhinol. 2016; 6:248–255.
Article14. Lund VJ, Kennedy DW. Staging for rhinosinusitis. Otolaryngol Head Neck Surg. 1997; 117:S35–40.
Article15. Lund VJ, Mackay IS. Staging in rhinosinusitus. Rhinology. 1993; 31:183–184.16. Kim DK, Jin HR, Eun KM, Mo JH, Cho SH, Oh S, et al. The role of interleukin-33 in chronic rhinosinusitis. Thorax. 2017; 72:635–645.
Article17. Shin HW, Kim DK, Park MH, Eun KM, Lee M, So D, et al. IL-25 as a novel therapeutic target in nasal polyps of patients with chronic rhinosinusitis. J Allergy Clin Immunol. 2015; 135:1476–1485.e7.
Article18. Kim DK, Eun KM, Kim MK, Cho D, Han SA, Han SY, et al. Comparison between signature cytokines of nasal tissues in subtypes of chronic rhinosinusitis. Allergy Asthma Immunol Res. 2019; 11:201–211.
Article19. Kim DW, Eun KM, Roh EY, Shin S, Kim DK. Chronic rhinosinusitis without nasal polyps in Asian patients shows mixed inflammatory patterns and neutrophil-related disease severity. Mediators Inflamm. 2019; 2019:7138643.
Article20. Peters AT, Kato A, Zhang N, Conley DB, Suh L, Tancowny B, et al. Evidence for altered activity of the IL-6 pathway in chronic rhinosinusitis with nasal polyps. J Allergy Clin Immunol. 2010; 125:397–403.e10.
Article21. Tan BK, Li QZ, Suh L, Kato A, Conley DB, Chandra RK, et al. Evidence for intranasal antinuclear autoantibodies in patients with chronic rhinosinusitis with nasal polyps. J Allergy Clin Immunol. 2011; 128:1198–1206.e1.
Article22. Deng F, Chen J, Zheng J, Chen Y, Huang R, Yin J, et al. Association of BAFF and IL-17A with subphenotypes of primary Sjögren's syndrome. Int J Rheum Dis. 2016; 19:715–720.
Article23. Kalled SL. The role of BAFF in immune function and implications for autoimmunity. Immunol Rev. 2005; 204:43–54.
Article24. Wei F, Chang Y, Wei W. The role of BAFF in the progression of rheumatoid arthritis. Cytokine. 2015; 76:537–544.
Article25. Zheng Y, Gallucci S, Gaughan JP, Gross JA, Monestier M. A role for B cell-activating factor of the TNF family in chemically induced autoimmunity. J Immunol. 2005; 175:6163–6168.
Article26. Ricciardolo FL, Sorbello V, Folino A, Gallo F, Massaglia GM, Favatà G, et al. Identification of IL-17F/frequent exacerbator endotype in asthma. J Allergy Clin Immunol. 2017; 140:395–406.
Article27. Vazquez-Tello A, Halwani R, Hamid Q, Al-Muhsen S. Glucocorticoid receptor-beta up-regulation and steroid resistance induction by IL-17 and IL-23 cytokine stimulation in peripheral mononuclear cells. J Clin Immunol. 2013; 33:466–478.
Article28. Irvin C, Zafar I, Good J, Rollins D, Christianson C, Gorska MM, et al. Increased frequency of dual-positive TH2/TH17 cells in bronchoalveolar lavage fluid characterizes a population of patients with severe asthma. J Allergy Clin Immunol. 2014; 134:1175–1186.e7.29. Chen HH, Liu X, Ni C, Lu YP, Xiong GY, Lu YY, et al. Bacterial biofilms in chronic rhinosinusitis and their relationship with inflammation severity. Auris Nasus Larynx. 2012; 39:169–174.
Article30. Cleland EJ, Bassiouni A, Wormald PJ. The bacteriology of chronic rhinosinusitis and the pre-eminence of Staphylococcus aureus in revision patients. Int Forum Allergy Rhinol. 2013; 3:642–646.31. Zhang Z, Linkin DR, Finkelman BS, O'Malley BW Jr, Thaler ER, Doghramji L, et al. Asthma and biofilm-forming bacteria are independently associated with revision sinus surgeries for chronic rhinosinusitis. J Allergy Clin Immunol. 2011; 128:221–223.e1.
Article32. Munari F, Fassan M, Capitani N, Codolo G, Vila-Caballer M, Pizzi M, et al. Cytokine BAFF released by Helicobacter pylori-infected macrophages triggers the Th17 response in human chronic gastritis. J Immunol. 2014; 193:5584–5594.33. Calzas C, Goyette-Desjardins G, Lemire P, Gagnon F, Lachance C, Van Calsteren MR, et al. Group B Streptococcus and Streptococcus suis capsular polysaccharides induce chemokine production by dendritic cells via Toll-like receptor 2- and MyD88-dependent and -independent pathways. Infect Immun. 2013; 81:3106–3118.34. Boita M, Heffler E, Pizzimenti S, Raie A, Saraci E, Omedè P, et al. Regulation of B-cell-activating factor expression on the basophil membrane of allergic patients. Int Arch Allergy Immunol. 2015; 166:208–212.
Article35. Lim SG, Kim JK, Suk K, Lee WH. Crosstalk between signals initiated from TLR4 and cell surface BAFF results in synergistic induction of proinflammatory mediators in THP-1 cells. Sci Rep. 2017; 7:45826.
Article36. Neill DR, Saint GL, Bricio-Moreno L, Fothergill JL, Southern KW, Winstanley C, et al. The B lymphocyte differentiation factor (BAFF) is expressed in the airways of children with CF and in lungs of mice infected with Pseudomonas aeruginosa. PLoS One. 2014; 9:e95892.37. Liu K, Zhang Y, Hu S, Yu Y, Yang Q, Jin D, et al. Increased levels of BAFF and APRIL related to human active pulmonary tuberculosis. PLoS One. 2012; 7:e38429.
Article38. McNamara PS, Fonceca AM, Howarth D, Correia JB, Slupsky JR, Trinick RE, et al. Respiratory syncytial virus infection of airway epithelial cells, in vivo and in vitro, supports pulmonary antibody responses by inducing expression of the B cell differentiation factor BAFF. Thorax. 2013; 68:76–81.39. Kato A, Peters A, Suh L, Carter R, Harris KE, Chandra R, et al. Evidence of a role for B cell-activating factor of the TNF family in the pathogenesis of chronic rhinosinusitis with nasal polyps. J Allergy Clin Immunol. 2008; 121:1385–1392. 1392.e1–1392.e2.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Medical treatment according to phenotypes of chronic rhinosinusitis
- Tissue Remodeling in Rhinosinusitis
- Practical Review of Biologics in Chronic Rhinosinusitis With Nasal Polyps
- The Role of Superantigen in Nasal Polypogenesis
- Role of Fungal and Bacterial Superantigen in the Pathogenesis of Chronic Rhinosinusitis with Polyps