J Clin Neurol.
2019 Jul;15(3):292-300. 10.3988/jcn.2019.15.3.292.
Association of Nutritional Status with Cognitive Stage in the Elderly Korean Population: The Korean Brain Aging Study for the Early Diagnosis and Prediction of Alzheimer's Disease
- Affiliations
-
- 1Department of Neurology, Kangwon National University Hospital, Kangwon National University College of Medicine, Chuncheon, Korea.
- 2Department of Biomedical Engineering, Hanyang University, Seoul, Korea.
- 3Department of Neurology, Konyang University College of Medicine, Daejeon, Korea.
- 4Department of Neurology, Dong-A Medical Center, Dong-A University College of Medicine, Busan, Korea.
- 5Department of Neurology, Inje University College of Medicine, Haeundae Paik Hospital, Busan, Korea.
- 6Department of Neurology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 7Department of Neurology, Eulji University College of Medicine, Daejeon, Korea.
- 8Department of Neurology, Ewha Womans University School of Medicine, Seoul, Korea.
- 9Department of Neurology, Pusan National University School of Medicine, Busan, Korea.
- 10Department of Neurology, Keimyung University Dongsan Medical Center, Daegu, Korea.
- 11Department of Pharmacology and Medicinal Toxicology Research Center, Inha University School of Medicine, Incheon, Korea.
- 12Department of Neurology, Yonsei University Wonju College of Medicine, Wonju, Korea. mlunoilu@hanmail.net
- 13Department of Neurology, Inha University School of Medicine, Incheon, Korea. seonghye@inha.ac.kr
- KMID: 2451111
- DOI: http://doi.org/10.3988/jcn.2019.15.3.292
Abstract
- BACKGROUND AND PURPOSE
Epidemiological studies have suggested the presence of strong correlations among diet, lifestyle, and dementia onset. However, these studies have unfortunately had major limitations due to their inability to fully control the various potential confounders affecting the nutritional status. The purpose of the current study was to determine the nutritional status of participants in the Korean Brain Aging Study for the Early Diagnosis and Prediction of Alzheimer's Disease (KBASE) and to identify clinical risk factors for being at risk of malnutrition or being malnourished.
METHODS
Baseline data from 212 participants [119 cognitively unimpaired (CU), 56 with mild cognitive impairment (MCI), and 37 with dementia] included in the KBASE database were analyzed. All participants underwent a comprehensive cognitive test and MRI at baseline. The presence of malnutrition at baseline was measured by the Mini Nutritional Assessment score. We examined the cross-sectional relationships of clinical findings with nutritional status using multiple logistic regression applied to variables for which p<0.2 in the univariate analysis. Differences in cortical thickness according to the nutritional status were also investigated.
RESULTS
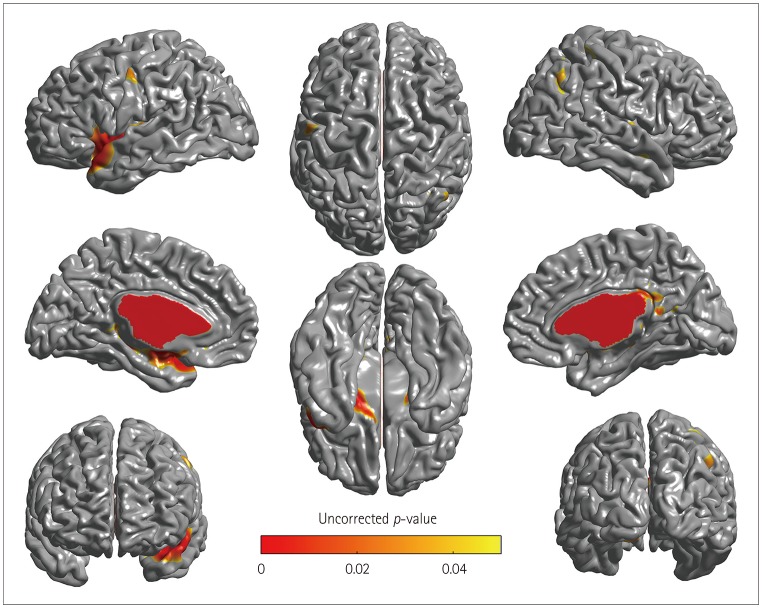
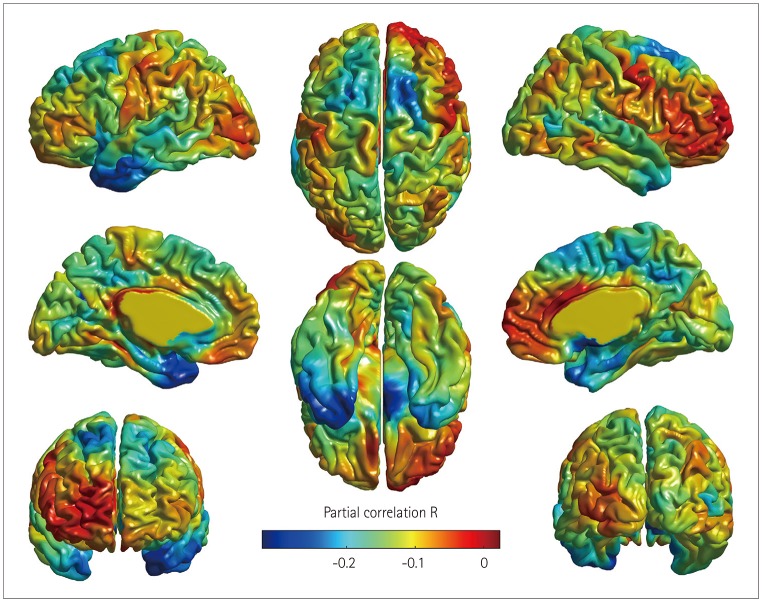
After adjustment for demographic, nutritional, and neuropsychological factors, participants with dementia had a significantly higher odds ratio (OR) for being at risk of malnutrition or being malnourished than CU participants [OR=5.98, 95% CI=1.20-32.97] whereas participants with MCI did not (OR=0.62, 95% CI=0.20-1.83). Cortical thinning in the at-risk/malnutrition group was observed in the left temporal area.
CONCLUSIONS
Dementia was found to be an independent predictor for the risk of malnutrition compared with CU participants. Our findings further suggest that cortical thinning in left temporal regions is related to the nutritional status.
Keyword
MeSH Terms
Figure
Reference
-
1. Ferri CP, Prince M, Brayne C, Brodaty H, Fratiglioni L, Ganguli M, et al. Global prevalence of dementia: a Delphi consensus study. Lancet. 2005; 366:2112–2117. PMID: 16360788.
Article2. Alzheimer's Disease International. Dementia statistics [Internet]. London: Alzheimer's Disease International;2015. cited 2018 Aug 1. Available from: https://www.alz.co.uk/research/statistics.3. Wimo A, Guerchet M, Ali GC, Wu YT, Prina AM, Winblad B, et al. The worldwide costs of dementia 2015 and comparisons with 2010. Alzheimers Dement. 2017; 13:1–7. PMID: 27583652.
Article4. Brookmeyer R, Evans DA, Hebert L, Langa KM, Heeringa SG, Plassman BL, et al. National estimates of the prevalence of Alzheimer's disease in the United States. Alzheimers Dement. 2011; 7:61–73. PMID: 21255744.
Article5. Otaegui-Arrazola A, Amiano P, Elbusto A, Urdaneta E, Martínez-Lage P. Diet, cognition, and Alzheimer's disease: food for thought. Eur J Nutr. 2014; 53:1–23.
Article6. Nicolia V, Lucarelli M, Fuso A. Environment, epigenetics and neurodegeneration: focus on nutrition in Alzheimer's disease. Exp Gerontol. 2015; 68:8–12. PMID: 25456841.
Article7. Greenwood CE, Parrott MD. Nutrition as a component of dementia risk reduction strategies. Healthc Manage Forum. 2017; 30:40–45. PMID: 28929899.
Article8. Berti V, Murray J, Davies M, Spector N, Tsui WH, Li Y, et al. Nutrient patterns and brain biomarkers of Alzheimer's disease in cognitively normal individuals. J Nutr Health Aging. 2015; 19:413–423. PMID: 25809805.
Article9. Mosconi L, Murray J, Davies M, Williams S, Pirraglia E, Spector N, et al. Nutrient intake and brain biomarkers of Alzheimer's disease in at-risk cognitively normal individuals: a cross-sectional neuroimaging pilot study. BMJ Open. 2014; 4:e004850.
Article10. Ahmed T, Haboubi N. Assessment and management of nutrition in older people and its importance to health. Clin Interv Aging. 2010; 5:207–216. PMID: 20711440.11. Muurinen S, Savikko N, Soini H, Suominen M, Pitkälä K. Nutrition and psychological well-being among long-term care residents with dementia. J Nutr Health Aging. 2015; 19:178–182. PMID: 25651443.
Article12. Park M, Song JA, Lee M, Jeong H, Lim S, Lee H, et al. National study of the nutritional status of Korean older adults with dementia who are living in long-term care settings. Jpn J Nurs Sci. 2018; 15:318–329. PMID: 29345106.
Article13. Lee DY, Lee KU, Lee JH, Kim KW, Jhoo JH, Kim SY, et al. A normative study of the CERAD neuropsychological assessment battery in the Korean elderly. J Int Neuropsychol Soc. 2004; 10:72–81. PMID: 14751009.
Article14. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. DSM-IV-TR [Internet]. Washington, D.C.: American Psychiatric Association;2000. cited 2018 May 7. Available from: https://dsm.psychiatryonline.org/doi/abs/10.1176/appi.books.9780890420249.dsm-iv-tr.15. McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH, et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011; 7:263–269. PMID: 21514250.
Article16. Lee JH, Lee KU, Lee DY, Kim KW, Jhoo JH, Kim JH, et al. Development of the Korean version of the Consortium to Establish a Registry for Alzheimer's Disease Assessment Packet (CERAD-K): clinical and neuropsychological assessment batteries. J Gerontol B Psychol Sci Soc Sci. 2002; 57:P47–P53. PMID: 11773223.
Article17. Zijdenbos AP, Forghani R, Evans AC. Automatic “pipeline” analysis of 3-D MRI data for clinical trials: application to multiple sclerosis. IEEE Trans Med Imaging. 2002; 21:1280–1291. PMID: 12585710.
Article18. Collins DL, Neelin P, Peters TM, Evans AC. Automatic 3D intersubject registration of MR volumetric data in standardized Talairach space. J Comput Assist Tomogr. 1994; 18:192–205. PMID: 8126267.
Article19. Sled JG, Zijdenbos AP, Evans AC. A nonparametric method for automatic correction of intensity nonuniformity in MRI data. IEEE Trans Med Imaging. 1998; 17:87–97. PMID: 9617910.
Article20. Smith SM. Fast robust automated brain extraction. Hum Brain Mapp. 2002; 17:143–155. PMID: 12391568.
Article21. Zijdenbos A, Evans AC, Riahi F, Sled JG, Chui J, Kollokian V. Automatic quantification of multiple sclerosis lesion volume using stereotaxic space. In : Höhne KH, Kikinis R, editors. Visualization in Biomedical Computing. Berlin: Springer;1996. p. 439–448.22. Kim JS, Singh V, Lee JK, Lerch J, Ad-Dab'bagh Y, MacDonald D, et al. Automated 3-D extraction and evaluation of the inner and outer cortical surfaces using a Laplacian map and partial volume effect classification. Neuroimage. 2005; 27:210–221. PMID: 15896981.
Article23. Lyttelton O, Boucher M, Robbins S, Evans A. An unbiased iterative group registration template for cortical surface analysis. Neuroimage. 2007; 34:1535–1544. PMID: 17188895.
Article24. Chung MK, Worsley KJ, Robbins S, Paus T, Taylor J, Giedd JN, et al. Deformation-based surface morphometry applied to gray matter deformation. Neuroimage. 2003; 18:198–213. PMID: 12595176.
Article25. Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage. 2002; 15:273–289. PMID: 11771995.
Article26. Wenham PR, Price WH, Blandell G. Apolipoprotein E genotyping by one-stage PCR. Lancet. 1991; 337:1158–1159.
Article27. Guigoz Y. The Mini Nutritional Assessment (MNA) review of the literature--What does it tell us? J Nutr Health Aging. 2006; 10:466–485. PMID: 17183419.28. Vellas B, Villars H, Abellan G, Soto ME, Rolland Y, Guigoz Y, et al. Overview of the MNA--Its history and challenges. J Nutr Health Aging. 2006; 10:456–463. PMID: 17183418.29. Neelemaat F, Kruizenga HM, De Vet HC, Seidell JC, Butterman M, Van Bokhorst-de van der Schueren MA. Screening malnutrition in hospital outpatients. Can the SNAQ malnutrition screening tool also be applied to this population? Clin Nutr. 2008; 27:439–446. PMID: 18395946.
Article30. Fox J, Monette G. Generalized collinearity diagnostics. J Am Stat Assoc. 1992; 87:178–183.
Article31. Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Series B Stat Methodol. 1995; 57:289–300.
Article32. Droogsma E, Van Asselt DZ, Schölzel-Dorenbos CJ, Van Steijn JH, Van Walderveen PE, Van der Hooft CS. Nutritional status of community-dwelling elderly with newly diagnosed Alzheimer's disease: prevalence of malnutrition and the relation of various factors to nutritional status. J Nutr Health Aging. 2013; 17:606–610. PMID: 23933871.
Article33. Guerin O, Soto ME, Brocker P, Robert PH, Benoit M, Vellas B. REAL.FR Group. Nutritional status assessment during Alzheimer's disease: results after one year (the REAL French Study Group). J Nutr Health Aging. 2005; 9:81–84. PMID: 15791350.34. Guérin O, Andrieu S, Schneider SM, Milano M, Boulahssass R, Brocker P, et al. Different modes of weight loss in Alzheimer disease: a prospective study of 395 patients. Am J Clin Nutr. 2005; 82:435–441. PMID: 16087990.35. Vellas B, Lauque S, Gillette-Guyonnet S, Andrieu S, Cortes F, Nourhashémi F, et al. Impact of nutritional status on the evolution of Alzheimer's disease and on response to acetylcholinesterase inhibitor treatment. J Nutr Health Aging. 2005; 9:75–80. PMID: 15791349.36. Spaccavento S, Del Prete M, Craca A, Fiore P. Influence of nutritional status on cognitive, functional and neuropsychiatric deficits in Alzheimer's disease. Arch Gerontol Geriatr. 2009; 48:356–360. PMID: 18448178.
Article37. Nourhashemi F, Amouyal-Barkate K, Gillette-Guyonnet S, Cantet C, Vellas B. REAL.FR Group. Living alone with Alzheimer's disease: cross-sectional and longitudinal analysis in the REAL.FR Study. J Nutr Health Aging. 2005; 9:117–120. PMID: 15791356.38. Saragat B, Buffa R, Mereu E, Succa V, Cabras S, Mereu RM, et al. Nutritional and psycho-functional status in elderly patients with Alzheimer's disease. J Nutr Health Aging. 2012; 16:231–236. PMID: 22456778.
Article39. Gillioz AS, Villars H, Voisin T, Cortes F, Gillette-Guyonnet S, Andrieu S, et al. Spared and impaired abilities in community-dwelling patients entering the severe stage of Alzheimer's disease. Dement Geriatr Cogn Disord. 2009; 28:427–432. PMID: 19907179.
Article40. Porter Starr KN, McDonald SR, Bales CW. Nutritional vulnerability in older adults: a continuum of concerns. Curr Nutr Rep. 2015; 4:176–184. PMID: 26042189.
Article41. Volkert D, Chourdakis M, Faxen-Irving G, Frühwald T, Landi F, Suominen MH, et al. ESPEN guidelines on nutrition in dementia. Clin Nutr. 2015; 34:1052–1073. PMID: 26522922.
Article42. Agarwal E, Miller M, Yaxley A, Isenring E. Malnutrition in the elderly: a narrative review. Maturitas. 2013; 76:296–302. PMID: 23958435.
Article43. Morley JE, Silver AJ. Nutritional issues in nursing home care. Ann Intern Med. 1995; 123:850–859. PMID: 7486469.
Article44. Fitten LJ, Morley JE, Gross PL, Petry SD, Cole KD. Depression. J Am Geriatr Soc. 1989; 37:459–472. PMID: 2564855.
Article45. Phillips RM. Nutrition and depression in the community-based oldest-old. Home Healthc Nurse. 2012; 30:462–471. PMID: 22936044.
Article46. Steenland K, Karnes C, Seals R, Carnevale C, Hermida A, Levey A. Late-life depression as a risk factor for mild cognitive impairment or Alzheimer's disease in 30 US Alzheimer's disease centers. J Alzheimers Dis. 2012; 31:265–275. PMID: 22543846.
Article47. Byers AL, Yaffe K. Depression and risk of developing dementia. Nat Rev Neurol. 2011; 7:323–331. PMID: 21537355.
Article48. Hardman RJ, Kennedy G, Macpherson H, Scholey AB, Pipingas A. Adherence to a mediterranean-style diet and effects on cognition in adults: a qualitative evaluation and systematic review of longitudinal and prospective trials. Front Nutr. 2016; 3:22. PMID: 27500135.
Article49. Staubo SC, Aakre JA, Vemuri P, Syrjanen JA, Mielke MM, Geda YE, et al. Mediterranean diet, micronutrients and macronutrients, and MRI measures of cortical thickness. Alzheimers Dement. 2017; 13:168–177. PMID: 27461490.
Article50. Mosconi L, Murray J, Tsui WH, Li Y, Davies M, Williams S, et al. Mediterranean diet and magnetic resonance imaging-assessed brain atrophy in cognitively normal individuals at risk for Alzheimer's disease. J Prev Alzheimers Dis. 2014; 1:23–32. PMID: 25237654.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Neuroimaging Markers for Alzheimer's Disease and Mild Cognitive Impairment in Alzheimer's Disease Neuroimaging Initiative (ADNI)
- New Diagnostic Criteria for Alzheimer's Disease : NIA-AA Diagnostic Guidelines
- Neuropsychological Assessment of Dementia and Cognitive Disorders
- EEG Coherence in Controls, Mild Cognitive Impairment and Different Stage of Alzheimer's Disease
- Memory Functions in Questionable Dementia of the Alzheimer Type