Int J Stem Cells.
2019 Mar;12(1):84-94. 10.15283/ijsc18102.
Effect of Substrate Topography and Chemistry on Human Mesenchymal Stem Cell Markers: A Transcriptome Study
- Affiliations
-
- 1Institute of Biomedical Engineering, Department of Engineering Science, University of Oxford, Oxford, UK. hui.wang@eng.ox.ac.uk, hua.ye@eng.ox.ac.uk
- 2Department of Engineering Science, University of Oxford, Oxford, UK.
- 3BGI-Shenzhen, Shenzhen 518083, China.
- 4Oxford Suzhou Centre for Advanced Research, Suzhou Industrial Park, Jiangsu, China.
- KMID: 2447227
- DOI: http://doi.org/10.15283/ijsc18102
Abstract
- BACKGROUND AND OBJECTIVES
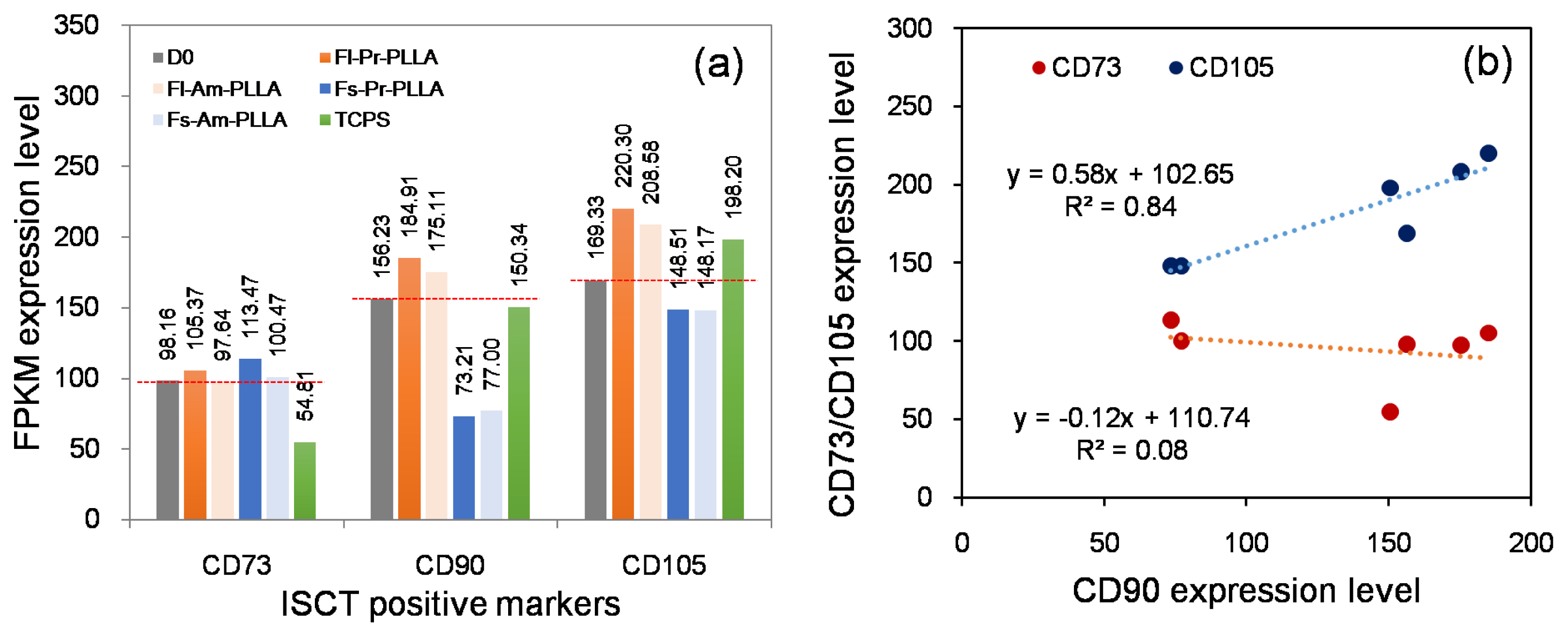
The International Society for Cellular Therapy (ISCT) proposed a set of minimal markers for identifying human mesenchymal stromal cells (hMSCs) in 2007. Since then, with the growing interest of better characterising hMSCs, various additional surface markers have been proposed. However, the impact of how culture conditions, in particular, the culture surface, vary the expression of hMSC markers was overlooked.
METHODS AND RESULTS
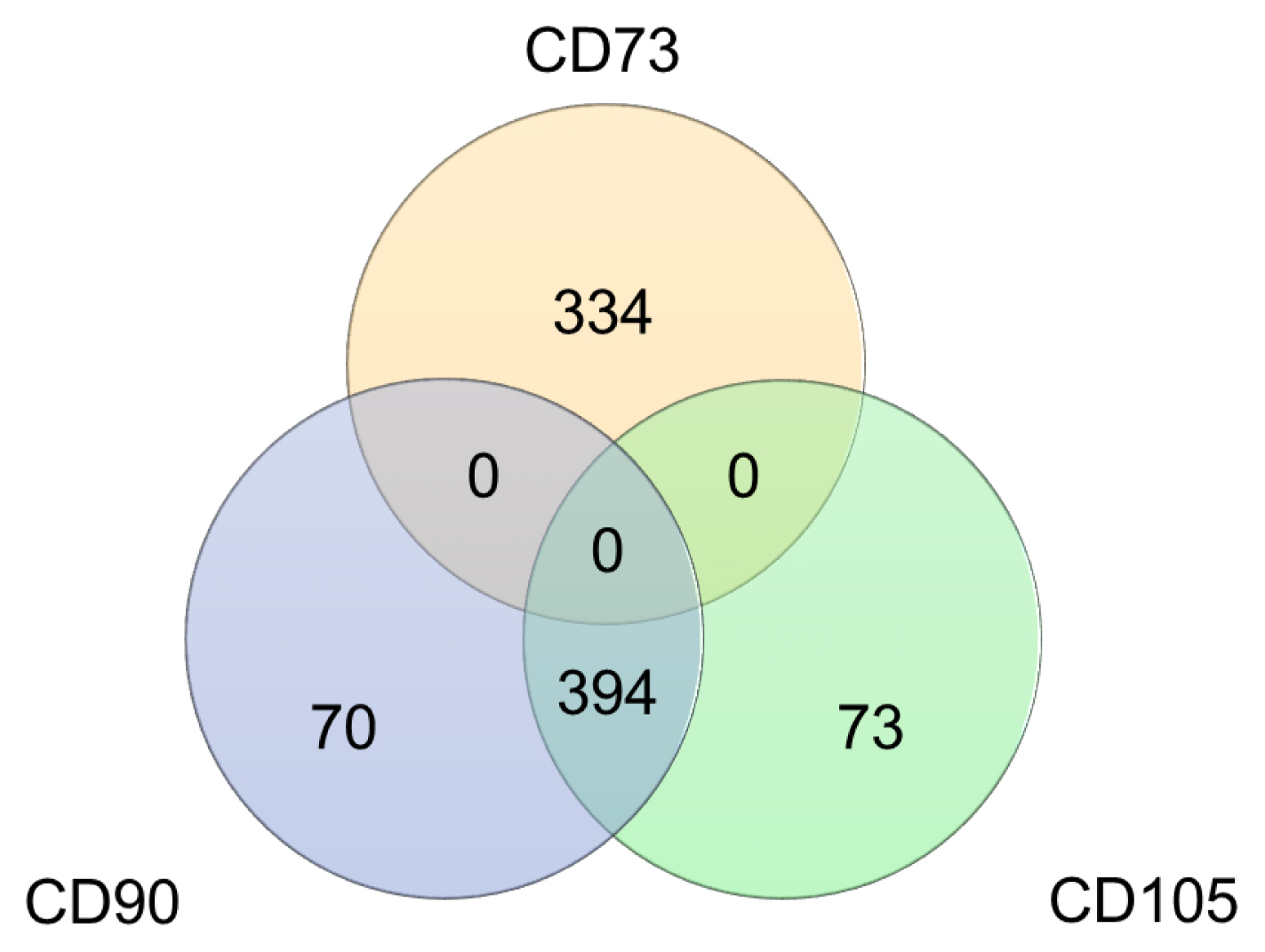
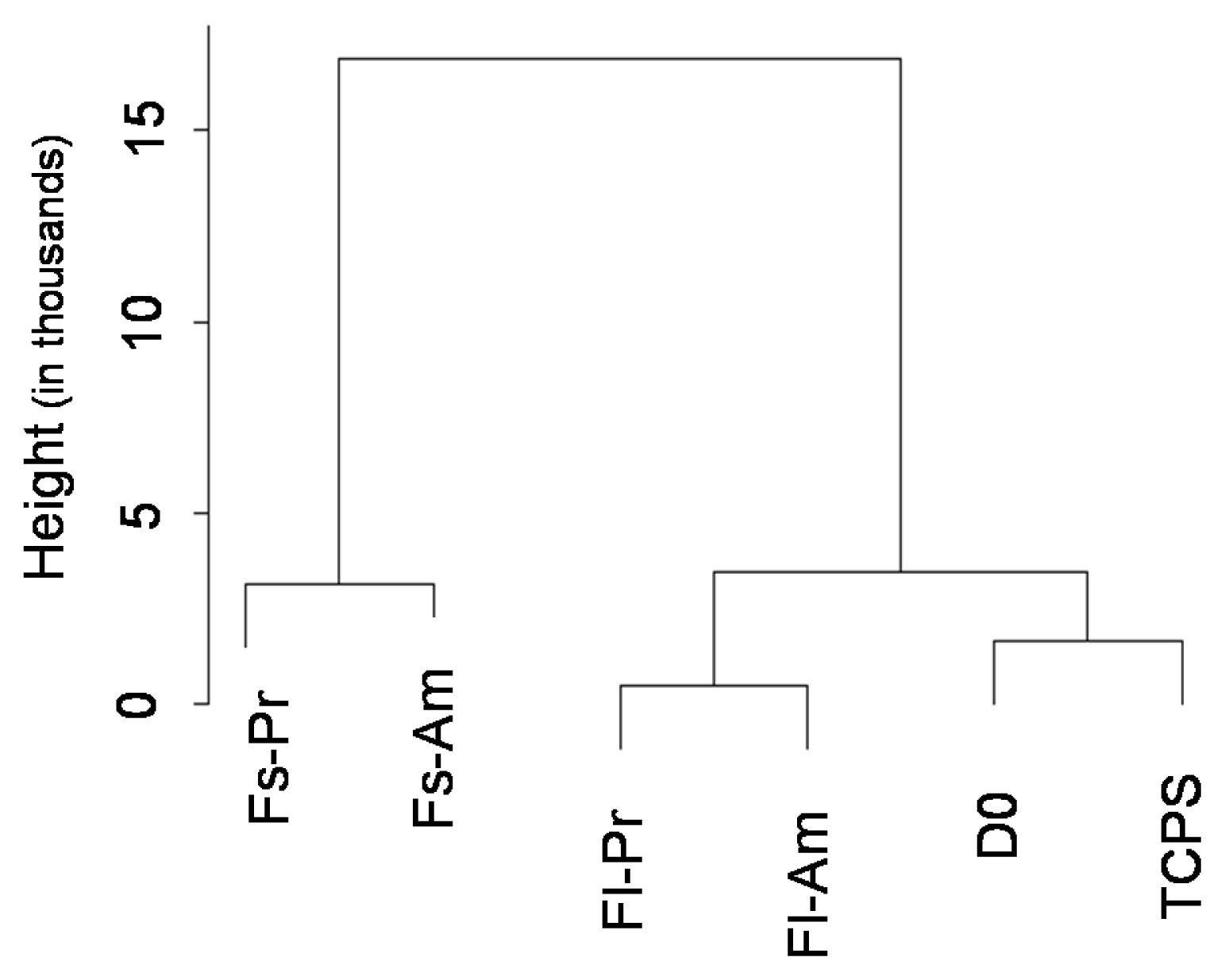
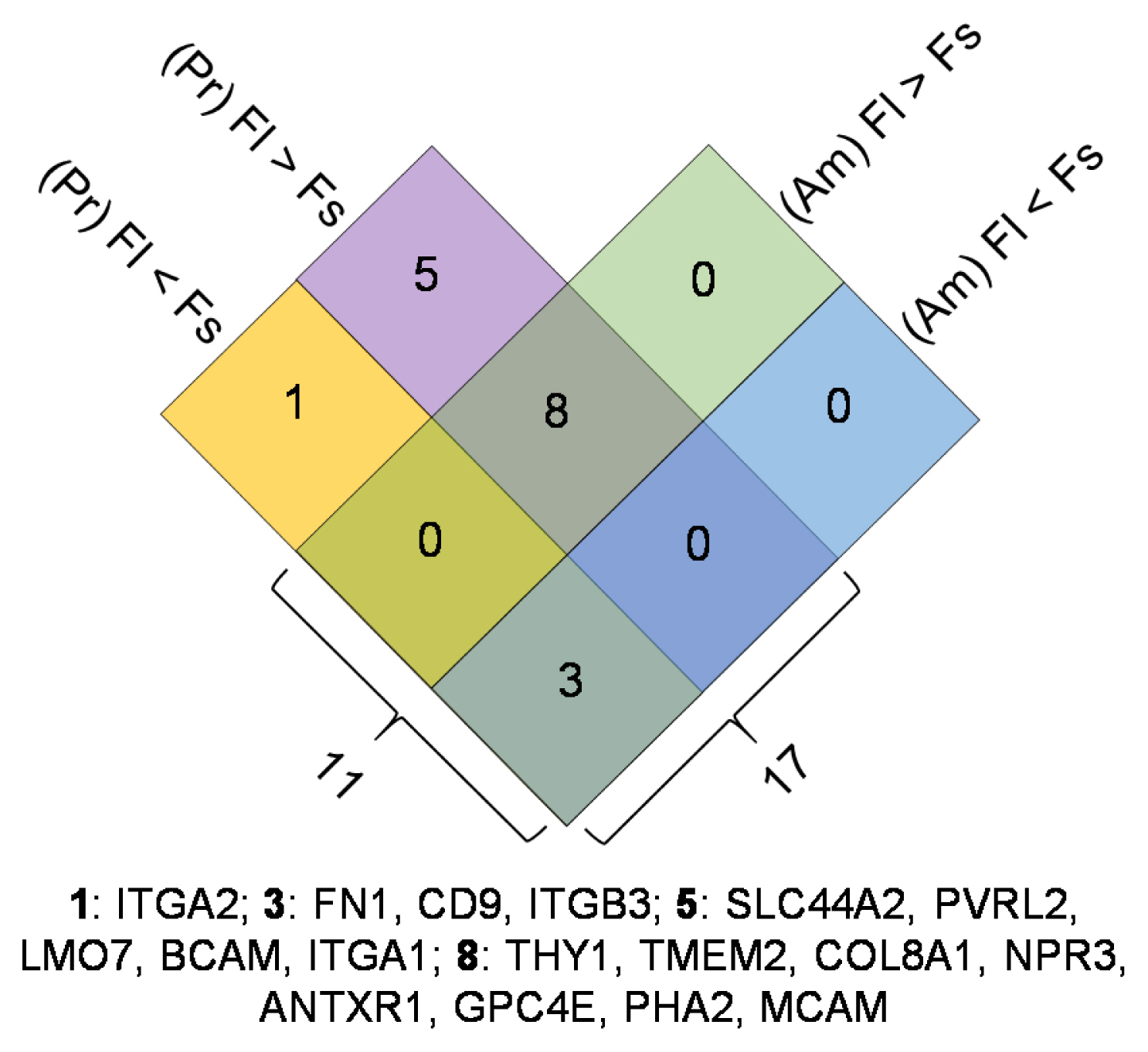
In this study, we utilized the RNA sequencing data on hMSCs cultured on different surfaces to investigate the variation of the proposed hMSC biomarkers. One of the three ISCT proposed positive biomarker, CD90 was found to be significantly down regulated on hMSCs culture on fibrous surfaces when compared to flat surfaces. The detected gene expression values for 177 hMSCs biomarkers compiled from the literature are reported here. Correlation and cluster analysis revealed the existence of different biomarker communities that displayed a similar expression profile. We found a list of hMSCs biomarkers which are the least sensitive to a change in surface properties and another list of biomarkers which are found to have high sensitivity to a change in surface properties.
CONCLUSIONS
This study demonstrated that substrate properties have paramount effect on altering the expressions of hMSCs biomarkers and the proposed list of substrate-stable and substrate-sensitive biomarkers would better assist in the population characterisation. However, proteomic level analysis would be essential to confirm the observations noted.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Ullah I, Subbarao RB, Rho GJ. Human mesenchymal stem cells - current trends and future prospective. Biosci Rep. 2015; 35. pii: e00191. DOI: 10.1042/BSR20150025. PMID: 25797907. PMCID: 4413017.
Article2. Song L, Tuan RS. Transdifferentiation potential of human mesenchymal stem cells derived from bone marrow. FASEB J. 2004; 18:980–982. DOI: 10.1096/fj.03-1100fje. PMID: 15084518.
Article3. Gao F, Chiu SM, Motan DA, Zhang Z, Chen L, Ji HL, Tse HF, Fu QL, Lian Q. Mesenchymal stem cells and immunomodulation: current status and future prospects. Cell Death Dis. 2016; 7:e2062. DOI: 10.1038/cddis.2015.327. PMID: 26794657. PMCID: 4816164.
Article4. Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop Dj, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006; 8:315–317. DOI: 10.1080/14653240600855905. PMID: 16923606.
Article5. Suila H, Pitkänen V, Hirvonen T, Heiskanen A, Anderson H, Laitinen A, Natunen S, Miller-Podraza H, Satomaa T, Natunen J, Laitinen S, Valmu L. Are globoseries glycosphingolipids SSEA-3 and -4 markers for stem cells derived from human umbilical cord blood? J Mol Cell Biol. 2011; 3:99–107. DOI: 10.1093/jmcb/mjq041. PMID: 21149348.
Article6. Lee RH, Seo MJ, Pulin AA, Gregory CA, Ylostalo J, Prockop DJ. The CD34-like protein PODXL and alpha6-integrin (CD49f) identify early progenitor MSCs with increased clonogenicity and migration to infarcted heart in mice. Blood. 2009; 113:816–826. DOI: 10.1182/blood-2007-12-128702. PMID: 18818395. PMCID: 2630267.
Article7. Pham H, Tonai R, Wu M, Birtolo C, Chen M. CD73, CD90, CD105 and Cadherin-11 RT-PCR screening for mesenchymal stem cells from cryopreserved human cord tissue. Int J Stem Cells. 2018; 11:26–38. DOI: 10.15283/ijsc17015. PMID: 29843192. PMCID: 5984056.
Article8. Lv FJ, Tuan RS, Cheung KM, Leung VY. Concise review: the surface markers and identity of human mesenchymal stem cells. Stem Cells. 2014; 32:1408–1419. DOI: 10.1002/stem.1681. PMID: 24578244.
Article9. Zhang Z, Gupte MJ, Ma PX. Biomaterials and stem cells for tissue engineering. Expert Opin Biol Ther. 2013; 13:527–540. DOI: 10.1517/14712598.2013.756468. PMID: 23327471. PMCID: 3596493.
Article10. Mahara A, Yamaoka T. Continuous separation of cells of high osteoblastic differentiation potential from mesenchymal stem cells on an antibody-immobilized column. Biomaterials. 2010; 31:4231–4237. DOI: 10.1016/j.biomaterials.2010.01.126. PMID: 20185169.
Article11. Chen AK, Reuveny S, Oh SK. Application of human mesenchymal and pluripotent stem cell microcarrier cultures in cellular therapy: achievements and future direction. Biotechnol Adv. 2013; 31:1032–1046. DOI: 10.1016/j.biotechadv.2013.03.006. PMID: 23531528.
Article12. Sambu S, Xu X, Schiffer HA, Cui ZF, Ye H. RGDS-fuctionalized alginates improve the survival rate of encapsulated embryonic stem cells during cryopreservation. Cryo Letters. 2011; 32:389–401. PMID: 22020461.13. Lee J, Abdeen AA, Kilian KA. Rewiring mesenchymal stem cell lineage specification by switching the biophysical microenvironment. Sci Rep. 2014; 4:5188. DOI: 10.1038/srep05188. PMID: 24898422. PMCID: 4046125.
Article14. Choi YS, Park SN, Suh H. Adipose tissue engineering using mesenchymal stem cells attached to injectable PLGA spheres. Biomaterials. 2005; 26:5855–5863. DOI: 10.1016/j.biomaterials.2005.02.022. PMID: 15949551.
Article15. Dalby MJ, Gadegaard N, Oreffo RO. Harnessing nano-topography and integrin-matrix interactions to influence stem cell fate. Nat Mater. 2014; 13:558–569. DOI: 10.1038/nmat3980. PMID: 24845995.
Article16. Walters NJ, Gentleman E. Evolving insights in cell-matrix interactions: elucidating how non-soluble properties of the extracellular niche direct stem cell fate. Acta Biomater. 2015; 11:3–16. DOI: 10.1016/j.actbio.2014.09.038. PMID: 25266503. PMCID: 5833939.
Article17. Jang HK, Kim BS. Modulation of stem cell differentiation with biomaterials. Int J Stem Cells. 2010; 3:80–84. DOI: 10.15283/ijsc.2010.3.2.80. PMID: 24855545. PMCID: 4021801.
Article18. McMurray RJ, Gadegaard N, Tsimbouri PM, Burgess KV, McNamara LE, Tare R, Murawski K, Kingham E, Oreffo RO, Dalby MJ. Nanoscale surfaces for the long-term maintenance of mesenchymal stem cell phenotype and multipotency. Nat Mater. 2011; 10:637–644. DOI: 10.1038/nmat3058. PMID: 21765399.
Article19. Dalby MJ, Gadegaard N, Tare R, Andar A, Riehle MO, Herzyk P, Wilkinson CD, Oreffo RO. The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder. Nat Mater. 2007; 6:997–1003. DOI: 10.1038/nmat2013. PMID: 17891143.
Article20. Benoit DS, Schwartz MP, Durney AR, Anseth KS. Small functional groups for controlled differentiation of hydrogel-encapsulated human mesenchymal stem cells. Nat Mater. 2008; 7:816–823. DOI: 10.1038/nmat2269. PMID: 18724374. PMCID: 2929915.
Article21. Phillips JE, Petrie TA, Creighton FP, García AJ. Human mesenchymal stem cell differentiation on self-assembled monolayers presenting different surface chemistries. Acta Biomater. 2010; 6:12–20. DOI: 10.1016/j.actbio.2009.07.023. PMID: 19632360. PMCID: 2787851.
Article22. Zamparelli A, Zini N, Cattini L, Spaletta G, Dallatana D, Bassi E, Barbaro F, Iafisco M, Mosca S, Parrilli A, Fini M, Giardino R, Sandri M, Sprio S, Tampieri A, Maraldi NM, Toni R. Growth on poly(L-lactic acid) porous scaffold preserves CD73 and CD90 immunophenotype markers of rat bone marrow mesenchymal stromal cells. J Mater Sci Mater Med. 2014; 25:2421–2436. DOI: 10.1007/s10856-014-5259-4. PMID: 24997163.
Article23. Duffy CR, Zhang R, How SE, Lilienkampf A, De Sousa PA, Bradley M. Long term mesenchymal stem cell culture on a defined synthetic substrate with enzyme free passaging. Biomaterials. 2014; 35:5998–6005. DOI: 10.1016/j.biomaterials.2014.04.013. PMID: 24780167.
Article24. Kasoju N, Wang H, Zhang B, George J, Gao S, Triffitt JT, Cui Z, Ye H. Transcriptomics of human multipotent mesenchymal stromal cells: retrospective analysis and future prospects. Biotechnol Adv. 2017; 35:407–418. DOI: 10.1016/j.biotechadv.2017.04.005. PMID: 28450077.
Article25. Sargent A, Shano G, Karl M, Garrison E, Miller C, Miller RH. Transcriptional profiling of mesenchymal stem cells identifies distinct neuroimmune pathways altered by CNS disease. Int J Stem Cells. 2018; 11:48–60. DOI: 10.15283/ijsc17062. PMID: 29699382. PMCID: 5984058.
Article26. Li Q, Zhang B, Kasoju N, Ma J, Yang A, Cui Z, Wang H, Ye H. Differential and interactive effects of substrate topography and chemistry on human mesenchymal stem cell gene expression. Int J Mol Sci. 2018; 19. pii: E2344. DOI: 10.3390/ijms19082344. PMID: 30096912. PMCID: 6121573.
Article27. Niehage C, Steenblock C, Pursche T, Bornhäuser M, Corbeil D, Hoflack B. The cell surface proteome of human mesenchymal stromal cells. PLoS One. 2011; 6:e20399. DOI: 10.1371/journal.pone.0020399. PMID: 21637820. PMCID: 3102717.
Article28. Cho KA, Park M, Kim YH, Woo SY, Ryu KH. RNA sequencing reveals a transcriptomic portrait of human mesenchymal stem cells from bone marrow, adipose tissue, and palatine tonsils. Sci Rep. 2017; 7:17114. DOI: 10.1038/s41598-017-16788-2. PMID: 29214990. PMCID: 5719355.
Article29. Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T. Cytoscape: a software environment for integrated models of bio-molecular interaction networks. Genome Res. 2003; 13:2498–2504. DOI: 10.1101/gr.1239303. PMID: 14597658. PMCID: 403769.
Article30. Girvan M, Newman ME. Community structure in social and biological networks. Proc Natl Acad Sci U S A. 2002; 99:7821–7826. DOI: 10.1073/pnas.122653799. PMID: 12060727. PMCID: 122977.
Article31. Bounova G. Octave networks toolbox. Zendo. 2015; DOI: 10.5281/zendo.22398.32. Mendicino M, Bailey AM, Wonnacott K, Puri RK, Bauer SR. MSC-based product characterization for clinical trials: an FDA perspective. Cell Stem Cell. 2014; 14:141–145. DOI: 10.1016/j.stem.2014.01.013. PMID: 24506881.
Article33. Skardal A, Mack D, Atala A, Soker S. Substrate elasticity controls cell proliferation, surface marker expression and motile phenotype in amniotic fluid-derived stem cells. J Mech Behav Biomed Mater. 2013; 17:307–316. DOI: 10.1016/j.jmbbm.2012.10.001. PMID: 23122714. PMCID: 3665276.
Article34. Colgan SP, Eltzschig HK, Eckle T, Thompson LF. Physiological roles for ecto-5′-nucleotidase (CD73). Purinergic Signal. 2006; 2:351–360. DOI: 10.1007/s11302-005-5302-5. PMID: 18404475. PMCID: 2254482.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Recent Trends and Strategies in Stem Cell Therapy for Alzheimer's Disease
- Stem cell properties of cells derived from canine periodontal ligament
- Characterization of Human Dental Pulp Cells from Supernumerary Teeth by Using Flow Cytometry Analysis
- Clinical Safety and Efficacy of Autologous Bone Marrow-Derived Mesenchymal Stem Cell Transplantation in Sensorineural Hearing Loss Patients
- Differentiation of human male germ cells from Wharton's jelly-derived mesenchymal stem cells