Allergy Asthma Immunol Res.
2019 May;11(3):330-342. 10.4168/aair.2019.11.3.330.
Altered Sphingolipid Metabolism Is Associated With Asthma Phenotype in House Dust Mite-Allergic Patients
- Affiliations
-
- 1Department of Experimental Allergology and Immunology, Medical University of Bialystok, Bialystok, Poland. kowalkmd@umb.edu.pl
- 2Department of Allergology and Internal Medicine, Medical University of Bialystok, Bialystok, Poland.
- 3Department of Physiology, Medical University of Bialystok, Bialystok, Poland.
- KMID: 2442045
- DOI: http://doi.org/10.4168/aair.2019.11.3.330
Abstract
- PURPOSE
Sphingolipids play an important role in cell growth, survival, inflammation and tissue remodeling. House dust mite (HDM) allergy is a major risk factor for asthma. The aim of the study was to evaluate if allergic asthma phenotype is associated with altered sphingolipid metabolism.
METHODS
Twenty-two HDM-allergic asthmatic patients and 11 HDM-allergic rhinitis patients were challenged intrabronchially with biologically standardized Dermatophagoides pteronyssinus extract. Whole blood and platelet-poor plasma samples were collected before, during early asthmatic response (EAR), late asthmatic response (LAR) and 24 hours after the challenge. Concentrations of sphinganine (SFA), sphinganine-1-phosphate (SFA1P), ceramide, sphingosine (SFO) and sphingosine-1-phosphate (S1P) were measured using high performance liquid chromatography.
RESULTS
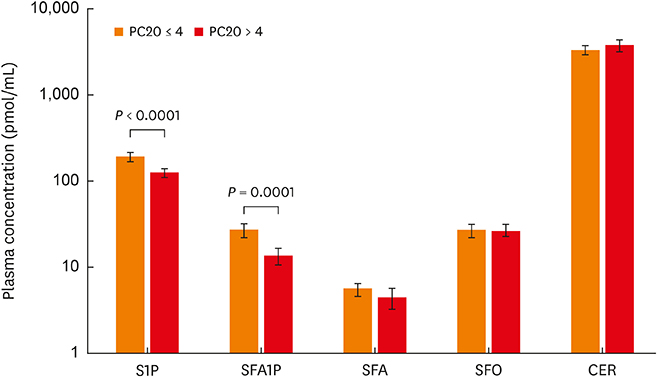
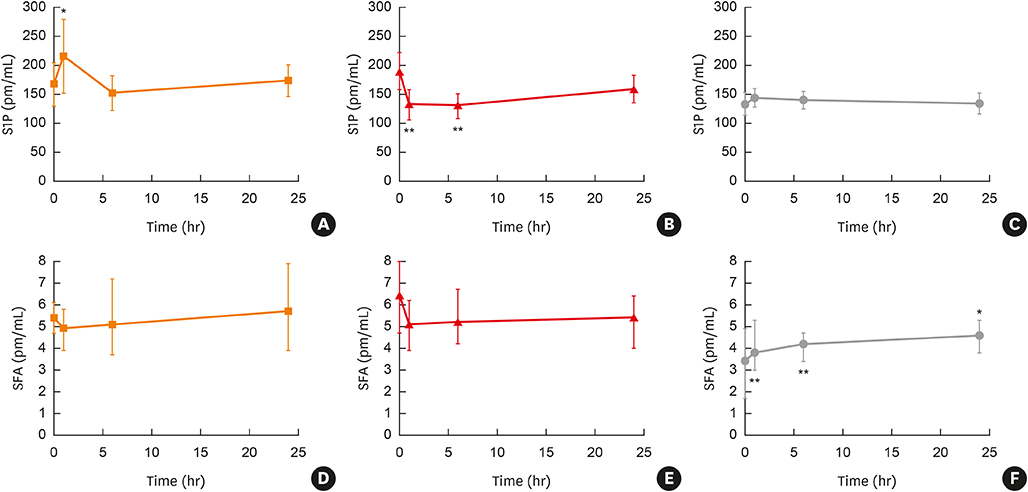
In all house dust mite-allergic patients (HDM-APs), baseline lung function and severity of airway hyperreactivity (AHR) correlated significantly with plasma S1P and SFA1P concentrations. Exhaled nitric oxide concentration, however, correlated with SFA and ceramide, but not with S1P or SFA1P concentration. Allergen challenge increased plasma S1P concentration during EAR, but only in patients who developed both EAR and LAR. The magnitude of the increase determined during EAR correlated with the severity of subsequently developed LAR. Platelet and eosinophil counts were independent predictors of plasma S1P concentration. A significant increase in plasma SFA concentration in response to allergen challenge was seen only in patients who did not develop asthmatic response.
CONCLUSIONS
Altered sphingolipid metabolism, with augmented synthesis of S1P and impaired de novo sphingolipid synthesis in response to allergen challenge, may participate in the development of asthma phenotype in HDM-APs.
Keyword
MeSH Terms
Figure
Reference
-
1. Lötvall J, Akdis CA, Bacharier LB, Bjermer L, Casale TB, Custovic A, et al. Asthma endotypes: a new approach to classification of disease entities within the asthma syndrome. J Allergy Clin Immunol. 2011; 127:355–360.2. Nelson RP Jr, DiNicolo R, Fernández-Caldas E, Seleznick MJ, Lockey RF, Good RA. Allergen-specific IgE levels and mite allergen exposure in children with acute asthma first seen in an emergency department and in nonasthmatic control subjects. J Allergy Clin Immunol. 1996; 98:258–263.
Article3. Wong GW, Li ST, Hui DS, Fok TF, Zhong NS, Chen YZ, et al. Individual allergens as risk factors for asthma and bronchial hyperresponsiveness in Chinese children. Eur Respir J. 2002; 19:288–293.
Article4. Obase Y, Shimoda T, Mitsuta K, Matsuo N, Matsuse H, Kohno S. Sensitivity to the house dust mite and airway hyperresponsiveness in a young adult population. Ann Allergy Asthma Immunol. 1999; 83:305–310.
Article5. Kowal K, Møller HJ, Dubuske LM, Moestrup SK, Bodzenta-Lukaszyk A. Differential expression of monocyte CD163 in single- and dual-asthmatic responders during allergen-induced bronchoconstriction. Clin Exp Allergy. 2006; 36:1584–1591.6. Kowal K, Bodzenta-Lukaszyk A, Zukowski S. Exhaled nitric oxide in evaluation of young adults with chronic cough. J Asthma. 2009; 46:692–698.
Article7. Hall S, Agrawal DK. Key mediators in the immunopathogenesis of allergic asthma. Int Immunopharmacol. 2014; 23:316–329.
Article8. Chapman DG, Irvin CG. Mechanisms of airway hyper-responsiveness in asthma: the past, present and yet to come. Clin Exp Allergy. 2015; 45:706–719.
Article9. Jolly PS, Rosenfeldt HM, Milstien S, Spiegel S. The roles of sphingosine-1-phosphate in asthma. Mol Immunol. 2002; 38:1239–1245.
Article10. Lai WQ, Wong WS, Leung BP. Sphingosine kinase and sphingosine 1-phosphate in asthma. Biosci Rep. 2011; 31:145–150.
Article11. Schauberger E, Peinhaupt M, Cazares T, Lindsley AW. Lipid mediators of allergic disease: pathways, treatments, and emerging therapeutic targets. Curr Allergy Asthma Rep. 2016; 16:48.
Article12. Ono JG, Worgall TS, Worgall S. Airway reactivity and sphingolipids-implications for childhood asthma. Mol Cell Pediatr. 2015; 2:13.13. Yang Y, Uhlig S. The role of sphingolipids in respiratory disease. Ther Adv Respir Dis. 2011; 5:325–344.
Article14. Nixon GF. Sphingolipids in inflammation: pathological implications and potential therapeutic targets. Br J Pharmacol. 2009; 158:982–993.
Article15. Roviezzo F, Sorrentino R, Bertolino A, De Gruttola L, Terlizzi M, Pinto A, et al. S1P-induced airway smooth muscle hyperresponsiveness and lung inflammation in vivo: molecular and cellular mechanisms. Br J Pharmacol. 2015; 172:1882–1893.16. Chiba Y, Suzuki K, Kurihara E, Uechi M, Sakai H, Misawa M. Sphingosine-1-phosphate aggravates antigen-induced airway inflammation in mice. Open Respir Med J. 2010; 4:82–85.
Article17. Price MM, Oskeritzian CA, Falanga YT, Harikumar KB, Allegood JC, Alvarez SE, et al. A specific sphingosine kinase 1 inhibitor attenuates airway hyperresponsiveness and inflammation in a mast cell-dependent murine model of allergic asthma. J Allergy Clin Immunol. 2013; 131:501–511.e1.
Article18. Karmouty-Quintana H, Siddiqui S, Hassan M, Tsuchiya K, Risse PA, Xicota-Vila L, et al. Treatment with a sphingosine-1-phosphate analog inhibits airway remodeling following repeated allergen exposure. Am J Physiol Lung Cell Mol Physiol. 2012; 302:L736–45.
Article19. Ammit AJ, Hastie AT, Edsall LC, Hoffman RK, Amrani Y, Krymskaya VP, et al. Sphingosine 1-phosphate modulates human airway smooth muscle cell functions that promote inflammation and airway remodeling in asthma. FASEB J. 2001; 15:1212–1214.
Article20. Fuerst E, Foster HR, Ward JP, Corrigan CJ, Cousins DJ, Woszczek G. Sphingosine-1-phosphate induces pro-remodeling response in airway smooth muscle cells. Allergy. 2014; 69:1531–1539.21. Książek M, Chacińska M, Chabowski A, Baranowski M. Sources, metabolism, and regulation of circulating sphingosine-1-phosphate. J Lipid Res. 2015; 56:1271–1281.
Article22. Kowal K, Pampuch A, Kowal-Bielecka O, DuBuske LM, Bodzenta-Łukaszyk A. Platelet activation in allergic asthma patients during allergen challenge with Dermatophagoides pteronyssinus . Clin Exp Allergy. 2006; 36:426–432.23. Baranowski M, Charmas M, Długołęcka B, Górski J. Exercise increases plasma levels of sphingoid base-1 phosphates in humans. Acta Physiol (Oxf). 2011; 203:373–380.
Article24. Knapp M, Lisowska A, Knapp P, Baranowski M. Dose-dependent effect of aspirin on the level of sphingolipids in human blood. Adv Med Sci. 2013; 58:274–281.
Article25. Trinh HK, Kim SC, Cho K, Kim SJ, Ban GY, Yoo HJ, et al. Exploration of the sphingolipid metabolite, sphingosine-1-phosphate and sphingosine, as novel biomarkers for aspirin-exacerbated respiratory disease. Sci Rep. 2016; 6:36599.
Article26. Chiba Y, Takeuchi H, Sakai H, Misawa M. SKI-II, an inhibitor of sphingosine kinase, ameliorates antigen-induced bronchial smooth muscle hyperresponsiveness, but not airway inflammation, in mice. J Pharmacol Sci. 2010; 114:304–310.
Article27. Sawicka E, Zuany-Amorim C, Manlius C, Trifilieff A, Brinkmann V, Kemeny DM, et al. Inhibition of Th1- and Th2-mediated airway inflammation by the sphingosine 1-phosphate receptor agonist FTY720. J Immunol. 2003; 171:6206–6214.
Article28. Rumzhum NN, Rahman MM, Oliver BG, Ammit AJ. Effect of sphingosine 1-phosphate on cyclo-oxygenase-2 expression, prostaglandin E2 secretion, and β2-adrenergic receptor desensitization. Am J Respir Cell Mol Biol. 2016; 54:128–135.29. Roviezzo F, Del Galdo F, Abbate G, Bucci M, D'Agostino B, Antunes E, et al. Human eosinophil chemotaxis and selective in vivo recruitment by sphingosine 1-phosphate. Proc Natl Acad Sci U S A. 2004; 101:11170–11175.30. Nussbaum C, Bannenberg S, Keul P, Gräler MH, Gonçalves-de-Albuquerque CF, Korhonen H, et al. Sphingosine-1-phosphate receptor 3 promotes leukocyte rolling by mobilizing endothelial P-selectin. Nat Commun. 2015; 6:6416.
Article31. Olivera A, Urtz N, Mizugishi K, Yamashita Y, Gilfillan AM, Furumoto Y, et al. IgE-dependent activation of sphingosine kinases 1 and 2 and secretion of sphingosine 1-phosphate requires Fyn kinase and contributes to mast cell responses. J Biol Chem. 2006; 281:2515–2525.
Article32. Roviezzo F, Sorrentino R, Iacono VM, Brancaleone V, Terlizzi M, Riemma MA, et al. Disodium cromoglycate inhibits asthma-like features induced by sphingosine-1-phosphate. Pharmacol Res. 2016; 113:626–635.
Article33. Moffatt MF, Kabesch M, Liang L, Dixon AL, Strachan D, Heath S, et al. Genetic variants regulating ORMDL3 expression contribute to the risk of childhood asthma. Nature. 2007; 448:470–473.
Article34. Levy BD. Sphingolipids and susceptibility to asthma. N Engl J Med. 2013; 369:976–978.
Article35. Miller M, Rosenthal P, Beppu A, Gordillo R, Broide DH. Oroscomucoid like protein 3 (ORMDL3) transgenic mice have reduced levels of sphingolipids including sphingosine-1-phosphate and ceramide. J Allergy Clin Immunol. 2017; 139:1373–1376.e4.
Article36. Miller M, Tam AB, Mueller JL, Rosenthal P, Beppu A, Gordillo R, et al. Cutting edge: targeting epithelial ORMDL3 increases, rather than reduces, airway responsiveness and is associated with increased sphingosine-1-phosphate. J Immunol. 2017; 198:3017–3022.
Article37. Ferrando M, Bagnasco D, Varricchi G, Bernardi S, Bragantini A, Passalacqua G, et al. Personalized medicine in allergy. Allergy Asthma Immunol Res. 2017; 9:15–24.
Article38. Braido F, Tiotiu A, Kowal K, Mihaicuta S, Novakova P, Oguzulgen IK. Phenotypes/endotypes-driven treatment in asthma. Curr Opin Allergy Clin Immunol. 2018; 18:184–189.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Asthma Management Educational Program on The Disease Related Knowledge, Stress, and Self-efficacy of Asthmatics Allergic to House Dust Mite
- Distribution of House Dust Mites in the Bedroom of Patients with Allergic Rhinitis in Pusan Area
- House dust mite dose not directly activate the human peripheral blood eosinophils in house dust mite-sensitized asthmatics
- Effect of Asthma Management Education Program on Stress and Compliance of Patients with Allergic Asthma to House Dust Mite
- Repellent effect of Mate tea and Jasmine tea against house dust mites (Dermatophagoides farinae and D. pteronyssinus)