Korean J Physiol Pharmacol.
2019 Mar;23(2):103-111. 10.4196/kjpp.2019.23.2.103.
Beneficial effects of andrographolide in a rat model of autoimmune myocarditis and its effects on PI3K/Akt pathway
- Affiliations
-
- 1Department of Geriatrics, Anhui Provincial Hospital, The First Affiliated Hospital of University of Science and Technology of China, Anhui Institute of Cardiovascular Disease, Hefei 230001, China. yanguang399@sina.com
- KMID: 2438084
- DOI: http://doi.org/10.4196/kjpp.2019.23.2.103
Abstract
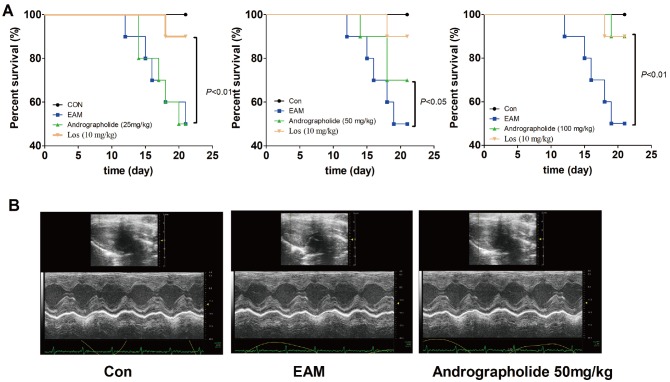
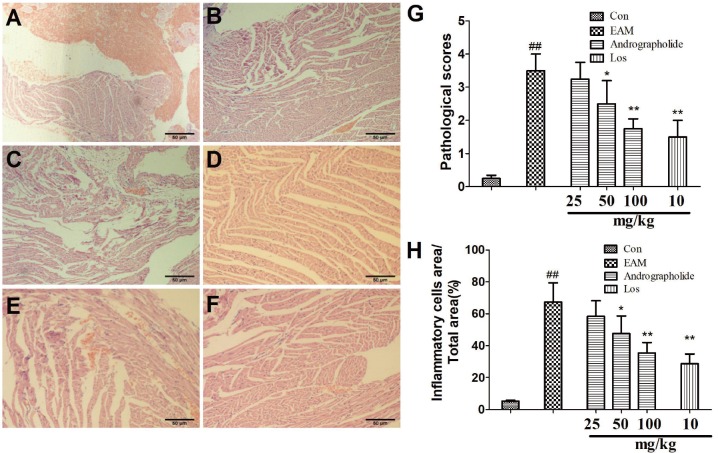
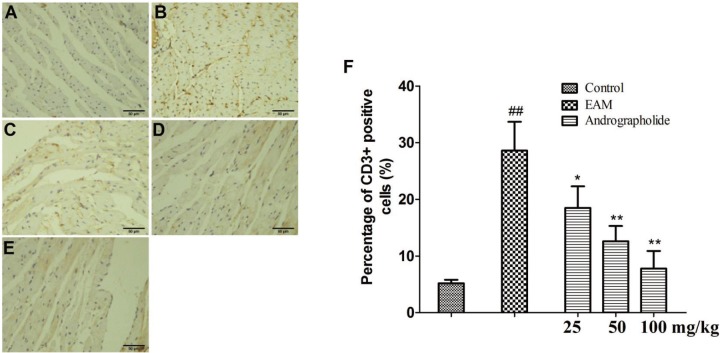
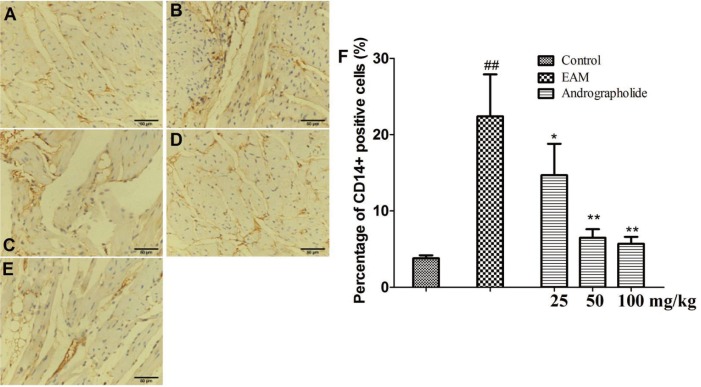
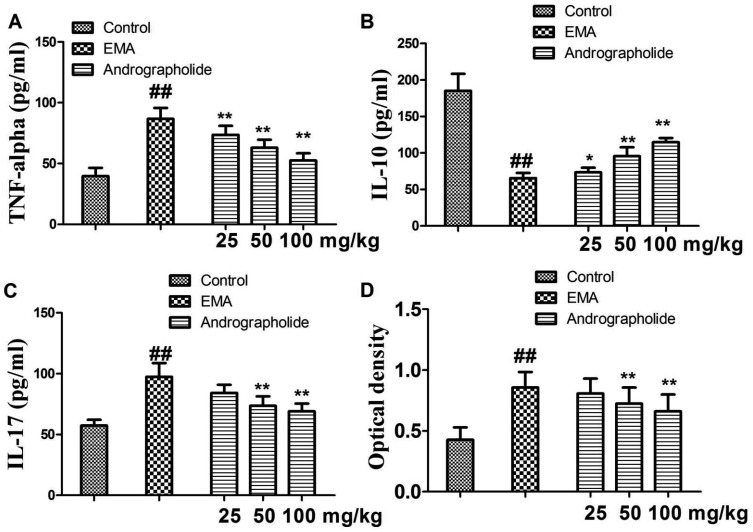
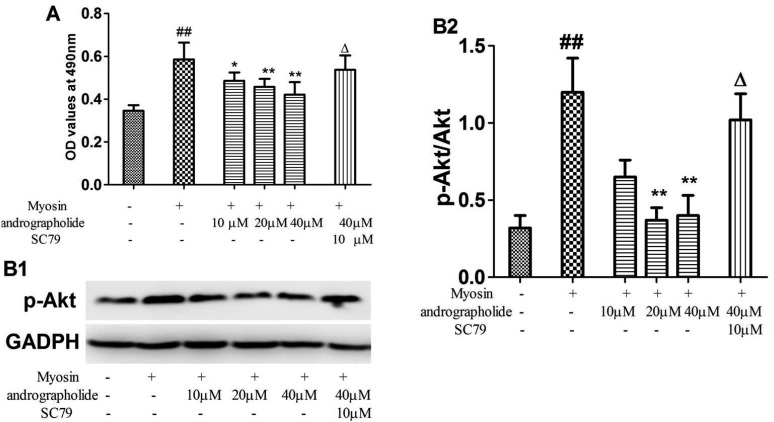
- The study is to investigate effects of andrographolide on experimental autoimmune myocarditis (EAM). Lewis rats were immunized on day 0 with porcine cardiac myosin to establish EAM. The EAM rats were treated with either andrographolide (25, 50, 100 mg/kg/day) or vehicle for 21 days. An antigen-specific splenocytes proliferation assay was performed by using the cells from control rats immunized with cardiac myosin. Survival rates, myocardial pathology and myocardial functional parameters (left ventricle end-diastolic pressure, ± dP/dt and left ventricular internal dimension) of EAM rats received andrographolide were significantly improved. Andrographolide treatment caused an decrease in the infiltration of CD3⺠and CD14⺠positive cells in myocardial tissue. Moreover, andrographolide treatment caused a reduction in the plasma levels of tumor necrosis factor-alpha, interleukin-17 (IL-17) and myosin-antibody, and an increase in the level of IL-10 in EAM rats. Oral administration of andrographolide resulted in the decreased expression of p-PI3K, p-Akt without any change of PI3K and Akt. Further results indicate andrographolide significantly inhibited myosin-induced proliferation in splenocytes, and this effect was inhibited by co-treatment of SC79 (Akt activator). Our data indicate andrographolide inhibits development of EAM, and this beneficial effect may be due to powerful anti-inflammatory activity and inhibitory effect on PI3K/Akt pathway.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Salvianolic acid B ameliorates psoriatic changes in imiquimod-induced psoriasis on BALB/c mice by inhibiting inflammatory and keratin markers
via altering phosphatidylinositol-3-kinase/protein kinase B signaling pathway
Shoufan Wang, Lihong Zhu, Yihou Xu, Zongbi Qin, Aiqin Xu
Korean J Physiol Pharmacol. 2020;24(3):213-221. doi: 10.4196/kjpp.2020.24.3.213.
Reference
-
1. Drory Y, Turetz Y, Hiss Y, Lev B, Fisman EZ, Pines A, Kramer MR. Sudden unexpected death in persons less than 40 years of age. Am J Cardiol. 1991; 68:1388–1392. PMID: 1951130.2. Cooper LT Jr, Keren A, Sliwa K, Matsumori A, Mensah GA. The global burden of myocarditis: part 1: a systematic literature review for the global burden of diseases, injuries, and risk factors 2010 study. Glob Heart. 2014; 9:121–129. PMID: 25432122.4. Elamm C, Fairweather D, Cooper LT. Pathogenesis and diagnosis of myocarditis. Heart. 2012; 11:835–840.
Article5. Ghigo A, Li M. Phosphoinositide 3-kinase: friend and foe in cardiovascular disease. Front Pharmacol. 2015; 6:169. PMID: 26321955.
Article6. Yu P, Zhang Y, Li C, Li Y, Jiang S, Zhang X, Ding Z, Tu F, Wu J, Gao X, Li L. Class III PI3K-mediated prolonged activation of autophagy plays a critical role in the transition of cardiac hypertrophy to heart failure. J Cell Mol Med. 2015; 19:1710–1719. PMID: 25851780.7. Liu HS, Zhang J, Guo JL, Lin CY, Wang ZW. Phosphoinositide 3-kinase inhibitor LY294002 ameliorates the severity of myosin-induced myocarditis in mice. Curr Res Transl Med. 2016; 64:21–27. PMID: 27140596.
Article8. Lim JC, Chan TK, Ng DS, Sagineedu SR, Stanslas J, Wong WS. Andrographolide and its analogues: versatile bioactive molecules for combating inflammation and cancer. Clin Exp Pharmacol Physiol. 2012; 39:300–310. PMID: 22017767.
Article9. He CL, Yi PF, Fan QJ, Shen HQ, Jiang XL, Qin QQ, Song Z, Zhang C, Wu SC, Wei XB, Li YL, Fu BD. Xiang-Qi-Tang and its active components exhibit anti-inflammatory and anticoagulant properties by inhibiting MAPK and NF-ĸB signaling pathways in LPS-treated rat cardiac microvascular endothelial cells. Immunopharmacol Immunotoxicol. 2013; 35:215–224. PMID: 23171279.
Article10. Hsieh YL, Shibu MA, Lii CK, Viswanadha VP, Lin YL, Lai CH, Chen YF, Lin KH, Kuo WW, Huang CY. Andrographis paniculata extract attenuates pathological cardiac hypertrophy and apoptosis in high-fat diet fed mice. J Ethnopharmacol. 2016; 192:170–177. PMID: 27401291.
Article11. Lee YC, Lin HH, Hsu CH, Wang CJ, Chiang TA, Chen JH. Inhibitory effects of andrographolide on migration and invasion in human non-small cell lung cancer A549 cells via down-regulation of PI3K/Akt signaling pathway. Eur J Pharmacol. 2010; 632:23–32. PMID: 20097193.
Article12. Wang W, Wang J, Dong SF, Liu CH, Italiani P, Sun SH, Xu J, Boraschi D, Ma SP, Qu D. Immunomodulatory activity of andrographolide on macrophage activation and specific antibody response. Acta Pharmacol Sin. 2010; 31:191–201. PMID: 20139902.
Article13. Veeraveedu PT, Watanabe K, Ma M, Thandavarayan RA, Palaniyandi SS, Yamaguchi K, Suzuki K, Kodama M, Aizawa Y. Comparative effects of torasemide and furosemide in rats with heart failure. Biochem Pharmacol. 2008; 75:649–659. PMID: 18001696.
Article14. Liu X, Zhang X, Ye L, Yuan H. Protective mechanisms of berberine against experimental autoimmune myocarditis in a rat model. Biomed Pharmacother. 2016; 79:222–230. PMID: 27044832.
Article15. Zhang J, Zhu D, Wang Y, Ju Y. Andrographolide attenuates LPS-induced cardiac malfunctions through inhibition of IĸB phosphorylation and apoptosis in mice. Cell Physiol Biochem. Cell Physiol Biochem. 2015; 37:1619–1628. PMID: 26536571.16. Woo AY, Waye MM, Tsui SK, Yeung ST, Cheng CH. Andrographolide up-regulates cellular-reduced glutathione level and protects cardiomyocytes against hypoxia/reoxygenation injury. J Pharmacol Exp Ther. 2008; 325:226–235. PMID: 18174384.
Article17. Lichtman AH. The heart of the matter: protection of the myocardium from T cells. J Autoimmun. 2013; 45:90–96. PMID: 23810579.
Article18. Myers JM, Cooper LT, Kem DC, Stavrakis S, Kosanke SD, Shevach EM, Fairweather D, Stoner JA, Cox CJ, Cunningham MW. Cardiac myosin-Th17 responses promote heart failure in human myocarditis. JCI Insight. 2016; 1(9):e85851.
Article19. Liu Y, Zhu H, Su Z, Sun C, Yin J, Yuan H, Sandoghchian S, Jiao Z, Wang S, Xu H. IL-17 contributes to cardiac fibrosis following experimental autoimmune myocarditis by a PKCβ/Erk1/2/NF-ĸB-dependent signaling pathway. Int Immunol. 2012; 24:605–612. PMID: 22531062.
Article20. Stumpf C, Seybold K, Petzi S, Wasmeier G, Raaz D, Yilmaz A, Anger T, Daniel WG, Garlichs CD. Interleukin-10 improves left ventricular function in rats with heart failure subsequent to myocardial infarction. Eur J Heart Fail. 2008; 10:733–739. PMID: 18599346.
Article21. Yu Y, Zhang ZH, Wei SG, Chu Y, Weiss RM, Heistad DD, Felder RB. Central gene transfer of interleukin-10 reduces hypothalamic inflammation and evidence of heart failure in rats after myocardial infarction. Circ Res. 2007; 101:304–312. PMID: 17569888.
Article22. Su SA, Yang D, Zhu W, Cai Z, Zhang N, Zhao L, Wang JA, Xiang M. Interleukin-17A mediates cardiomyocyte apoptosis through Stat3-iNOS pathway. Biochim Biophys Acta. 2016; 1863:2784–2794. PMID: 27566322.
Article23. Damås JK, Aukrust P, Ueland T, Odegaard A, Eiken HG, Gullestad L, Sejersted OM, Christensen G. Monocyte chemoattractant protein-1 enhances and interleukin-10 suppresses the production of inflammatory cytokines in adult rat cardiomyocytes. Basic Res Cardiol. 2001; 96:345–352. PMID: 11518190.
Article24. Chen ZW, Qian JY, Ma JY, Chang SF, Yun H, Jin H, Sun AJ, Zou YZ, Ge JB. TNF-α-induced cardiomyocyte apoptosis contributes to cardiac dysfunction after coronary microembolization in mini-pigs. J Cell Mol Med. 2014; 18:1953–1963. PMID: 25130514.25. Duerrschmid C, Trial J, Wang Y, Entman ML, Haudek SB. Tumor necrosis factor: a mechanistic link between angiotensin-II-induced cardiac inflammation and fibrosis. Circ Heart Fail. 2015; 8:352–361. PMID: 25550440.26. Guan SP, Kong LR, Cheng C, Lim JC, Wong WS. Protective role of 14-deoxy-11,12-didehydroandrographolide, a noncytotoxic analogue of andrographolide, in allergic airway inflammation. J Nat Prod. 2011; 74:1484–1490. PMID: 21598983.
Article27. Ji X, Li C, Ou Y, Li N, Yuan K, Yang G, Chen X, Yang Z, Liu B, Cheung WW, Wang L, Huang R, Lan T. Andrographolide ameliorates diabetic nephropathy by attenuating hyperglycemia-mediated renal oxidative stress and inflammation via Akt/NF-ĸB pathway. Mol Cell Endocrinol. 2016; 437:268–279. PMID: 27378149.
Article28. Yin JN, Li YN, Gao Y, Li SB, Li JD. Andrographolide plays an important role in bleomycin-induced pulmonary fibrosis treatment. Int J Clin Exp Med. 2015; 8:12374–12381. PMID: 26550147.29. Condorelli G, Drusco A, Stassi G, Bellacosa A, Roncarati R, Iaccarino G, Russo MA, Gu Y, Dalton N, Chung C, Latronico MV, Napoli C, Sadoshima J, Croce CM, Ross J Jr. Akt induces enhanced myocardial contractility and cell size in vivo in transgenic mice. Proc Natl Acad Sci U S A. 2002; 99:12333–12338. PMID: 12237475.30. DeBosch B, Treskov I, Lupu TS, Weinheimer C, Kovacs A, Courtois M, Muslin AJ. Akt1 is required for physiological cardiac growth. Circulation. 2006; 113:2097–2104. PMID: 16636172.
Article31. McMullen JR, Shioi T, Huang WY, Zhang L, Tarnavski O, Bisping E, Schinke M, Kong S, Sherwood MC, Brown J, Riggi L, Kang PM, Izumo S. The insulin-like growth factor 1 receptor induces physiological heart growth via the phosphoinositide 3-kinase(p110α) pathway. J Biol Chem. 2004; 279:4782–4793. PMID: 14597618.
Article32. Shioi T, Kang PM, Douglas PS, Hampe J, Yballe CM, Lawitts J, Cantley LC, Izumo S. The conserved phosphoinositide 3-kinase pathway determines heart size in mice. EMBO J. 2000; 19:2537–2548. PMID: 10835352.
Article33. Shiojima I, Sato K, Izumiya Y, Schiekofer S, Ito M, Liao R, Colucci WS, Walsh K. Disruption of coordinated cardiac hypertrophy and angiogenesis contributes to the transition to heart failure. J Clin Invest. 2005; 115:2108–2118. PMID: 16075055.
Article34. Chen HW, Lin AH, Chu HC, Li CC, Tsai CW, Chao CY, Wang CJ, Lii CK, Liu KL. Inhibition of TNF-α-Induced inflammation by andrographolide via down-regulation of the PI3K/Akt signaling pathway. J Nat Prod. 2011; 74:2408–2413. PMID: 22026410.
Article35. Lu CY, Yang YC, Li CC, Liu KL, Lii CK, Chen HW. Andrographolide inhibits TNFα-induced ICAM-1 expression via suppression of NADPH oxidase activation and induction of HO-1 and GCLM expression through the PI3K/Akt/Nrf2 and PI3K/Akt/AP-1 pathways in human endothelial cells. Biochem Pharmacol. 2014; 91:40–50. PMID: 24998495.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Developmental Exposure to Di-(2-ethylhexyl) Phthalate Induces Cerebellar Granule Cell Apoptosis via the PI3K/AKT Signaling Pathway
- Integrin β1 in Adipose-Derived Stem Cells Accelerates Wound Healing via Activating PI3K/AKT Pathway
- Irisin Enhances Angiogenesis of Mesenchymal Stem Cells to Promote Cardiac Function in Myocardial Infarction via PI3k/Akt Activation
- Analysis of the mechanism of fibrauretine alleviating Alzheimer's disease based on transcriptomics and proteomics
- The role of PI3K/AKT pathway and its therapeutic possibility in Alzheimer's disease