Clin Exp Otorhinolaryngol.
2019 Feb;12(1):86-94. 10.21053/ceo.2018.00486.
Decellularization of Trachea With Combined Techniques for Tissue-Engineered Trachea Transplantation
- Affiliations
-
- 1Department of Otolaryngology, Head and Neck Surgery, Istanbul University Cerrahpasa Medicine Faculty, Istanbul, Turkey. batioglu@yahoo.com
- 2Acibadem Labcell Laboratories, Istanbul, Turkey.
- 3Department of Plastic and Reconstructive Surgery, Acibadem University School of Medicine, Istanbul, Turkey.
- 4Department of Pathology, Istanbul Education and Research Hospital, Istanbul, Turkey.
- 5Department of Histology and Embryology, Akdeniz University Medicine Faculty, Antalya, Turkey.
- 6Faculty of Mechanical Engineering, Department of Mechanical Engineering, Istanbul Technical University, Istanbul, Turkey.
- 7Department of Otolaryngology, Head and Neck Surgery, Istanbul Education and Research Hospital, Istanbul, Turkey.
- KMID: 2437496
- DOI: http://doi.org/10.21053/ceo.2018.00486
Abstract
OBJECTIVES
The purpose of this study is to shorten the decellularization time of trachea by using combination of physical, chemical, and enzymatic techniques.
METHODS
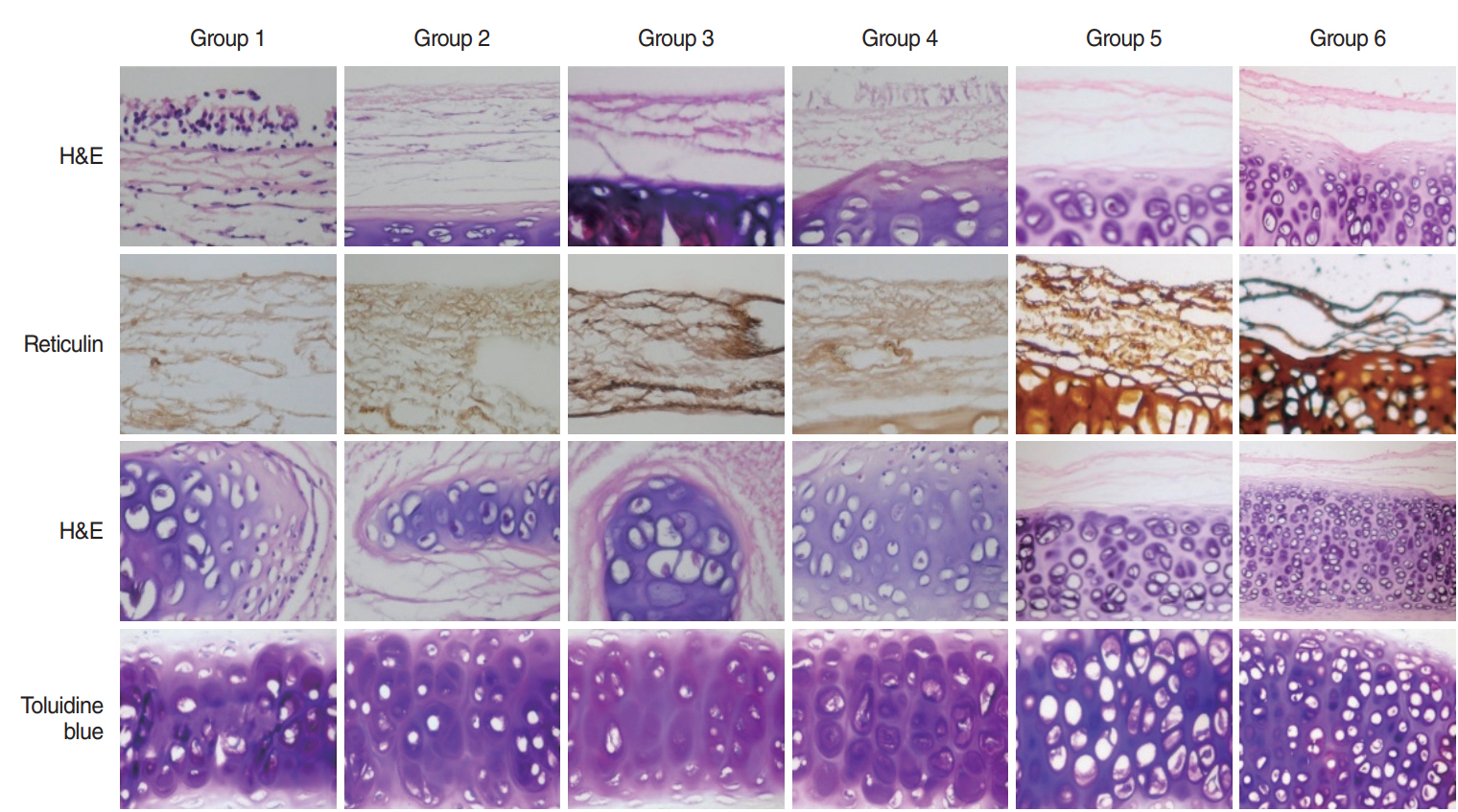
Approximately 3.5-cm-long tracheal segments from 42 New Zealand rabbits (3.5±0.5 kg) were separated into seven groups according to decellularization protocols. After decellularization, cellular regions, matrix and strength and endurance of the scaffold were followed up.
RESULTS
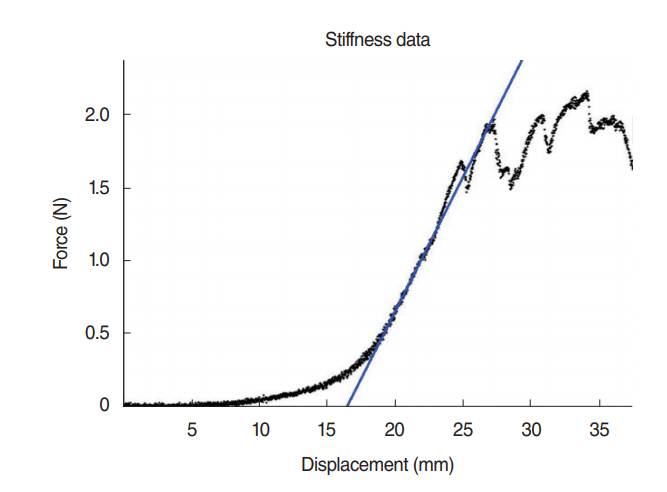
DNA content in all groups was measured under 50 ng/mg and there was no significant difference for the glycosaminoglycan content between group 3 (lyophilization+deoxycholic acid+de-oxyribonuclease method) and control group (P=0.46). None of the decellularized groups was different than the normal trachea in tensile stress values (P>0.05). Glucose consumption and lactic acid levels measured from supernatants of all decellularized groups were close to group with cells only (76 mg/dL and 53 mg/L).
CONCLUSION
Using combination methods may reduce exposure to chemicals, prevent the excessive influence of the matrix, and shorten the decellularization time.
Keyword
MeSH Terms
Figure
Reference
-
1. Zang M, Zhang Q, Chang EI, Mathur AB, Yu P. Decellularized tracheal matrix scaffold for tissue engineering. Plast Reconstr Surg. 2012; Sep. 130(3):532–40.
Article2. Macchiarini P, Walles T, Biancosino C, Mertsching H. First human transplantation of a bioengineered airway tissue. J Thorac Cardiovasc Surg. 2004; Oct. 128(4):638–41.
Article3. Go T, Jungebluth P, Baiguero S, Asnaghi A, Martorell J, Ostertag H, et al. Both epithelial cells and mesenchymal stem cell-derived chondrocytes contribute to the survival of tissue-engineered airway transplants in pigs. J Thorac Cardiovasc Surg. 2010; Feb. 139(2):437–43.
Article4. Jungebluth P, Moll G, Baiguera S, Macchiarini P. Tissue-engineered airway: a regenerative solution. Clin Pharmacol Ther. 2012; Jan. 91(1):81–93.
Article5. Benders KE, van Weeren PR, Badylak SF, Saris DB, Dhert WJ, Malda J. Extracellular matrix scaffolds for cartilage and bone regeneration. Trends Biotechnol. 2013; Mar. 31(3):169–76.
Article6. Schwarz S, Elsaesser AF, Koerber L, Goldberg-Bockhorn E, Seitz AM, Bermueller C, et al. Processed xenogenic cartilage as innovative biomatrix for cartilage tissue engineering: effects on chondrocyte differentiation and function. J Tissue Eng Regen Med. 2015; Dec. 9(12):E239–51.
Article7. Schwarz S, Koerber L, Elsaesser AF, Goldberg-Bockhorn E, Seitz AM, Dürselen L, et al. Decellularized cartilage matrix as a novel biomatrix for cartilage tissue-engineering applications. Tissue Eng Part A. 2012; Nov. 18(21-22):2195–209.
Article8. Yang Z, Shi Y, Wei X, He J, Yang S, Dickson G, et al. Fabrication and repair of cartilage defects with a novel acellular cartilage matrix scaffold. Tissue Eng Part C Methods. 2010; Oct. 16(5):865–76.
Article9. Kalathur M, Baiguera S, Macchiarini P. Translating tissue-engineered tracheal replacement from bench to bedside. Cell Mol Life Sci. 2010; Dec. 67(24):4185–96.
Article10. Sutherland AJ, Beck EC, Dennis SC, Converse GL, Hopkins RA, Berkland CJ, et al. Decellularized cartilage may be a chondroinductive material for osteochondral tissue engineering. PLoS One. 2015; May. 10(5):e0121966.
Article11. Baiguera S, Jungebluth P, Burns A, Mavilia C, Haag J, De Coppi P, et al. Tissue engineered human tracheas for in vivo implantation. Biomaterials. 2010; Dec. 31(34):8931–8.
Article12. Jungebluth P, Go T, Asnaghi A, Bellini S, Martorell J, Calore C, et al. Structural and morphologic evaluation of a novel detergent-enzymatic tissue-engineered tracheal tubular matrix. J Thorac Cardiovasc Surg. 2009; Sep. 138(3):586–93.
Article13. Remlinger NT, Czajka CA, Juhas ME, Vorp DA, Stolz DB, Badylak SF, et al. Hydrated xenogeneic decellularized tracheal matrix as a scaffold for tracheal reconstruction. Biomaterials. 2010; May. 31(13):3520–6.
Article14. Gilbert TW. Strategies for tissue and organ decellularization. J Cell Biochem. 2012; Jul. 113(7):2217–22.
Article15. Suzuki T, Kobayashi K, Tada Y, Suzuki Y, Wada I, Nakamura T, et al. Regeneration of the trachea using a bioengineered scaffold with adipose-derived stem cells. Ann Otol Rhinol Laryngol. 2008; Jun. 117(6):453–63.
Article16. Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997; Apr. 276(5309):71–4.
Article17. Kafienah W, Mistry S, Williams C, Hollander AP. Nucleostemin is a marker of proliferating stromal stem cells in adult human bone marrow. Stem Cells. 2006; Apr. 24(4):1113–20.
Article18. Tsutsumi S, Shimazu A, Miyazaki K, Pan H, Koike C, Yoshida E, et al. Retention of multilineage differentiation potential of mesenchymal cells during proliferation in response to FGF. Biochem Biophys Res Commun. 2001; Oct. 288(2):413–9.
Article19. Gilbert TW, Sellaro TL, Badylak SF. Decellularization of tissues and organs. Biomaterials. 2006; Jul. 27(19):3675–83.
Article20. Crapo PM, Gilbert TW, Badylak SF. An overview of tissue and whole organ decellularization processes. Biomaterials. 2011; Apr. 32(12):3233–43.
Article21. Hodde J, Hiles M. Virus safety of a porcine-derived medical device: evaluation of a viral inactivation method. Biotechnol Bioeng. 2002; Jul. 79(2):211–6.
Article22. Yang M, Chen CZ, Wang XN, Zhu YB, Gu YJ. Favorable effects of the detergent and enzyme extraction method for preparing decellularized bovine pericardium scaffold for tissue engineered heart valves. J Biomed Mater Res B Appl Biomater. 2009; Oct. 91(1):354–61.
Article23. Meezan E, Hjelle JT, Brendel K, Carlson EC. A simple, versatile, nondisruptive method for the isolation of morphologically and chemically pure basement membranes from several tissues. Life Sci. 1975; Dec. 17(11):1721–32.
Article24. Batioglu-Karaaltin A, Karaaltin MV, Ovali E, Yigit O, Kongur M, Inan O, et al. In vivo tissue-engineered allogenic trachea transplantation in rabbits: a preliminary report. Stem Cell Rev. 2015; Apr. 11(2):347–56.
Article25. Nireesha GR, Divya L, Sowmya C, Venkateshan N, Babu MN, Lavakumar V. Lyophilization/freeze drying: an review. Int J Nov Trends Pharm Sci. 2013; Oct. 3(4):87–98.26. Wang S, Goecke T, Meixner C, Haverich A, Hilfiker A, Wolkers WF. Freeze-dried heart valve scaffolds. Tissue Eng Part C Methods. 2012; Jul. 18(7):517–25.
Article27. Hung SH, Su CH, Lee FP, Tseng H. Larynx decellularization: combining freeze-drying and sonication as an effective method. J Voice. 2013; May. 27(3):289–94.
Article28. Ingavle GC, Frei AW, Gehrke SH, Detamore MS. Incorporation of aggrecan in interpenetrating network hydrogels to improve cellular performance for cartilage tissue engineering. Tissue Eng Part A. 2013; Jun. 19(11-12):1349–59.
Article29. Roberts CR, Rains JK, Pare PD, Walker DC, Wiggs B, Bert JL. Ultrastructure and tensile properties of human tracheal cartilage. J Biomech. 1998; Jan. 31(1):81–6.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Comparative Study of the Effects of Different Decellularization Methods and Genipin-Cross-Linking on the Properties of Tracheal Matrices
- Decellularlized Matrix in Organ Transplantation
- Tissue-Engineered Tracheal Regeneration: Current Status
- Benign Neoplasms of the Trachea: Case Reports
- Surgery of the Trachea