Lab Anim Res.
2018 Sep;34(3):111-117. 10.5625/lar.2018.34.3.111.
Inhibitory effects of Pycnogenol®, a pine bark extract, in a rat model of testosterone propionate-induced benign prostatic hyperplasia
- Affiliations
-
- 1College of Veterinary Medicine (BK21 Plus Project Team), Chonnam National University, Gwangju, Korea. dvmmk79@gmail.com dhshin@jnu.ac.kr
- KMID: 2420823
- DOI: http://doi.org/10.5625/lar.2018.34.3.111
Abstract
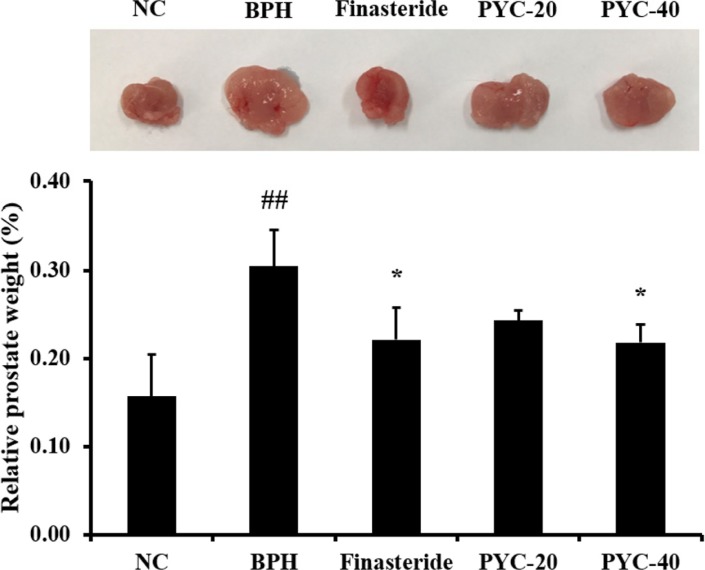
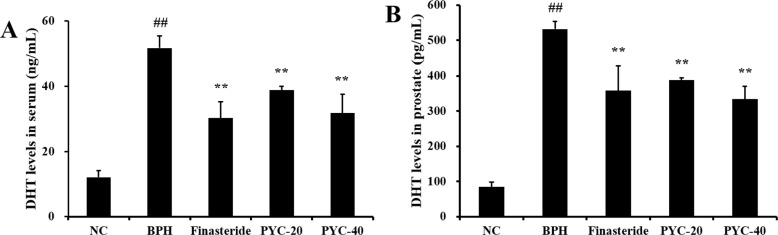
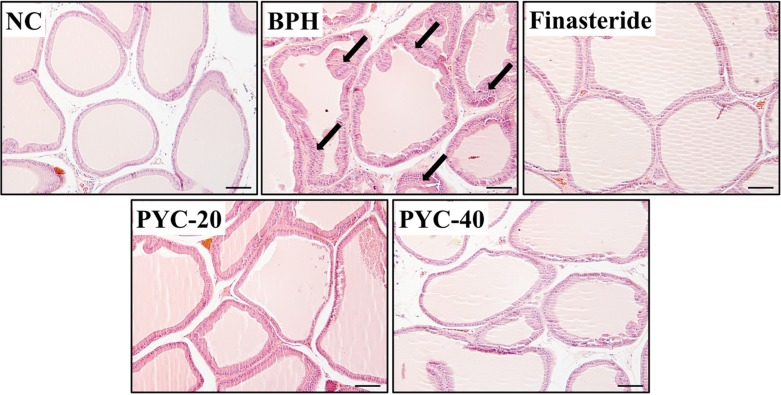
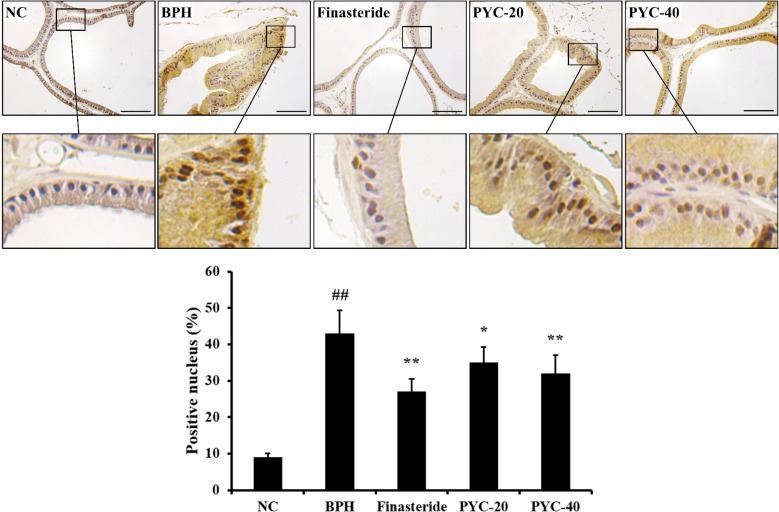
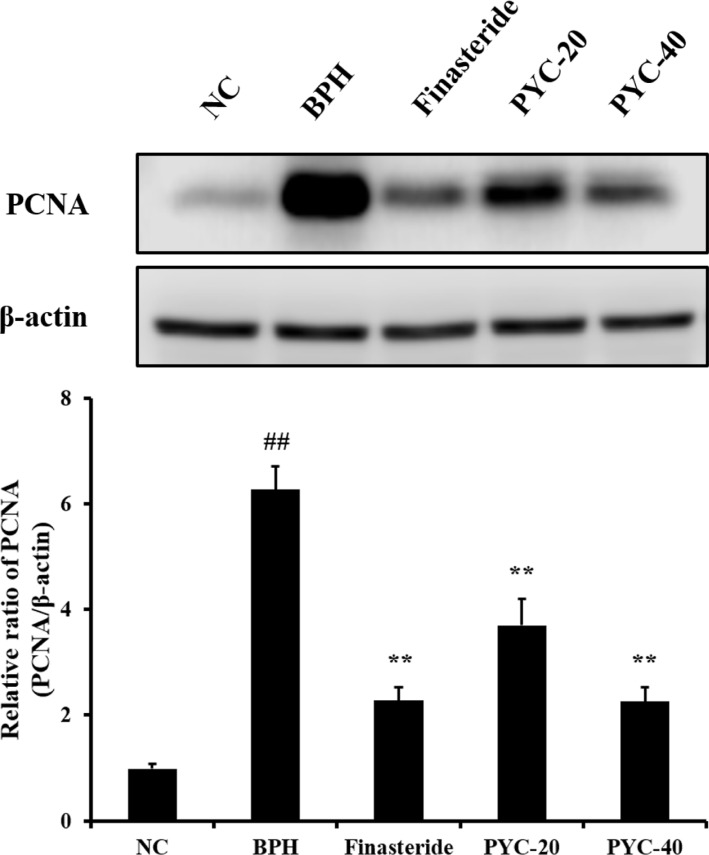
- Benign prostate hyperplasia (BPH) is a male reproductive disease that has gained increasing importance in recent years. The present study investigated whether Pycnogenol® (PYC), a standardized French maritime pine bark extract, could prevent BPH induced by testosterone propionate (TP) in rats. Male Sprague-Dawley rats were randomly divided into five groups of six rats. One group was used as a normal control rats and the other groups received subcutaneous injections of TP for 4 weeks to induce BPH. In the two treatment groups, PYC (20 or 40 mg/kg) was administered daily for 4 weeks by oral gavage concurrently with the induction of TP. All rats were sacrificed at the scheduled termination time, the prostates were weighed, and histopathologic examinations were conducted. Dihydrotestosterone (DHT) levels in serum and the prostate were measured, and the expression of proliferating cell nuclear antigen (PCNA) and Ki-67 proteins was investigated. BPH-treated animals showed increases in the relative weight of the prostate, higher concentrations of DHT in serum and the prostate, and higher expression of PCNA and Ki-67 in the prostate; in contrast, PYC-treated animals had significant reductions in these factors compared with the BPH animals. These findings indicated that PYC inhibited the development of BPH and that this was closely associated with a reduction in DHT concentration.
Keyword
MeSH Terms
Figure
Reference
-
1. Parsons JK. Benign Prostatic Hyperplasia and Male Lower Urinary Tract Symptoms: Epidemiology and Risk Factors. Curr Bladder Dysfunct Rep. 2010; 5(4):212–218. PMID: 21475707.
Article2. Roehrborn CG. Pathology of benign prostatic hyperplasia. Int J Impot Res. 2008; 20:S11–S18.
Article3. Zhang P, Hu WL, Cheng B, Zeng YJ, Wang XH, Liu TZ, Zhang WB. Which play a more important role in the development of large-sized prostates (80 ml), androgen receptors or oestrogen receptors? A comparative study. Int Urol Nephrol. 2016; 48(3):325–333. PMID: 26685888.4. Wang Z, Hu L, Salari K, Bechis SK, Ge R, Wu S, Rassoulian C, Pham J, Wu CL, Tabatabaei S, Strand DW, Olumi AF. Androgenic to oestrogenic switch in the human adult prostate gland is regulated by epigenetic silencing of steroid 5α-reductase 2. J Pathol. 2017; 243(4):457–467. PMID: 28940538.
Article5. Kiguradze T, Temps WH, Yarnold PR, Cashy J, Brannigan RE, Nardone B, Micali G, West DP, Belknap SM. Persistent erectile dysfunction in men exposed to the 5α-reductase inhibitors, finasteride, or dutasteride. PeerJ. 2017; 5:e3020. PMID: 28289563.
Article6. Canguven O, Burnett AL. The effect of 5 alpha-reductase inhibitors on erectile function. J Androl. 2008; 29(5):514–523. PMID: 18421068.7. Aggarwal S, Verma A, Shukla R, Batra S, Bhardwaj TR, Kumar M. An overview of phytotherapeutic approaches for the treatment of benign prostatic hyperplasia. Mini Rev Med Chem. 2014; 14(3):257–270. PMID: 24456273.
Article8. Ko JW, Shin NR, Park SH, Kim JS, Cho YK, Kim JC, Shin IS, Shin DH. Pine bark extract (Pycnogenol®) suppresses cigarette smoke induced fibrotic response via transforming growth factor-β1/Smad family member 2/3 signaling. Lab Anim Res. 2017; 33(2):76–83. PMID: 28747971.9. Bayomy NA, Abdelaziz EZ, Said MA, Badawi MS, El-Bakary RH. Effect of pycnogenol and spirulina on vancomycin-induced renal cortical oxidative stress, apoptosis, and autophagy in adult male albino rat. Can J Physiol Pharmacol. 2016; 94(8):838–848. PMID: 27203524.
Article10. Feng WH, Wei HL, Liu GT. Effect of PYCNOGENOL on the toxicity of heart, bone marrow and immune organs as induced by antitumor drugs. Phytomedicine. 2002; 9(5):414–418. PMID: 12222661.
Article11. Yang YS, Ahn TH, Lee JC, Moon CJ, Kim SH, Jun W, Park SC, Kim HC, Kim JC. Protective effects of Pycnogenol on carbon tetrachloride-induced hepatotoxicity in Sprague-Dawley rats. Food Chem Toxicol. 2008; 46(1):380–387. PMID: 17900780.
Article12. Eryilmaz A, Eliyatkin N, Demirci B, Basal Y, Kurt Omurlu I, Gunel C, Aktas S, Toka A, Basak S. Protective effect of Pycnogenol on cisplatin-induced ototoxicity in rats. Pharm Biol. 2016; 54(11):2777–2781. PMID: 27158843.
Article13. Huang WW, Yang JS, Lin CF, Ho WJ, Lee MR. Pycnogenol induces differentiation and apoptosis in human promyeloid leukemia HL-60 cells. Leuk Res. 2005; 29(6):685–692. PMID: 15863210.
Article14. Lee MY, Shin IS, Kyoung H, Seo CS, Son JK, Shin HK. α-spinasterol from Melandrium firmum attenuates benign prostatic hyperplasia in a rat model. Mol Med Rep. 2014; 9(6):2362–2366. PMID: 24682042.
Article15. Arivazhagan J, Nandeesha H, Dorairajan LN, Sreenivasulu K. Association of elevated interleukin-17 and angiopoietin-2 with prostate size in benign prostatic hyperplasia. Aging Male. 2017; 20(2):115–118. PMID: 28590830.
Article16. Bisson JF, Hidalgo S, Simons R, Verbruggen M. Preventive effects of lignan extract from flax hulls on experimentally induced benign prostate hyperplasia. J Med Food. 2014; 17(6):650–656. PMID: 24460407.
Article17. Kim CY, Chung KS, Cheon SY, Lee JH, Park YB, An HJ. Rice Hull Extract Suppresses Benign Prostate Hyperplasia by Decreasing Inflammation and Regulating Cell Proliferation in Rats. J Med Food. 2016; 19(8):746–754. PMID: 27441629.
Article18. Gratzke C, Bachmann A, Descazeaud A, Drake MJ, Madersbacher S, Mamoulakis C, Oelke M, Tikkinen KAO, Gravas S. EAU Guidelines on the Assessment of Non-neurogenic Male Lower Urinary Tract Symptoms including Benign Prostatic Obstruction. Eur Urol. 2015; 67(6):1099–1109. PMID: 25613154.
Article19. Harati K, Slodnik P, Chromik AM, Behr B, Goertz O, Hirsch T, Kapalschinski N, Klein-Hitpass L, Kolbenschlag J, Uhl W, Lehnhardt M, Daigeler A. Pro-apoptotic effects of pycnogenol on HT1080 human fibrosarcoma cells. Int J Oncol. 2015; 46(4):1629–1636. PMID: 25625225.
Article20. Romar GA, Kupper TS, Divito SJ. Research Techniques Made Simple: Techniques to Assess Cell Proliferation. J Invest Dermatol. 2016; 136(1):e1–e7. PMID: 26763463.
Article21. Vital P, Castro P, Ittmann M. Oxidative stress promotes benign prostatic hyperplasia. Prostate. 2016; 76(1):58–67. PMID: 26417670.
Article22. Cai HC, Song LB, Zhang GW, Qing XR, Mo DS, Liu W, Zhan XX, Huang YF, Shang XJ. Effects of Xialiqi Capsules on the expressions of PCNA and caspase-3 in rats with benign prostatic hyperplasia. Zhonghua Nan Ke Xue. 2017; 23(8):728–733. PMID: 29726649.23. Carson C, Rittmaster R. The role of dihydrotestosterone in benign prostatic hyperplasia. Urology. 2003; 61(4 Suppl 1):2–7.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Antihyperplastic Effect of Oral Catechin Ingestion in a Rat Model of Benign Prostatic Hyperplasia
- The Effects of Anthocyanin Extracted from Black Soybean on a Benign Prostatic Hyperplasia-induced Rat Model
- Ethanol Extract of Chaenomeles sinensis Inhibits the Development of Benign Prostatic Hyperplasia by Exhibiting Anti-oxidant and Anti-inflammatory Effects
- Effect of P. ginseng on the expression of c-Fos in the brain of Wistar rats with testosterone induced benign prostatic hyperplasia
- Influence of Sex Hormones on Citric Acid Concentration of the Prostatic Tissue of Male White Rats