Korean J Physiol Pharmacol.
2018 Sep;22(5):539-546. 10.4196/kjpp.2018.22.5.539.
Botulinum toxin type A enhances the inhibitory spontaneous postsynaptic currents on the substantia gelatinosa neurons of the subnucleus caudalis in immature mice
- Affiliations
-
- 1Department of Oral Physiology, School of Dentistry & Institute of Oral Bioscience, Chonbuk National University, Jeonju 54896, Korea. skhan@jbnu.ac.kr
- 2Research and Development Division, Hugel Inc., Chuncheon 24206, Korea.
- 3Department of Oral Physiology, School of Dentistry, Kyungpook National University, Daegu 41940, Korea. dkahn@knu.ac.kr
- KMID: 2419003
- DOI: http://doi.org/10.4196/kjpp.2018.22.5.539
Abstract
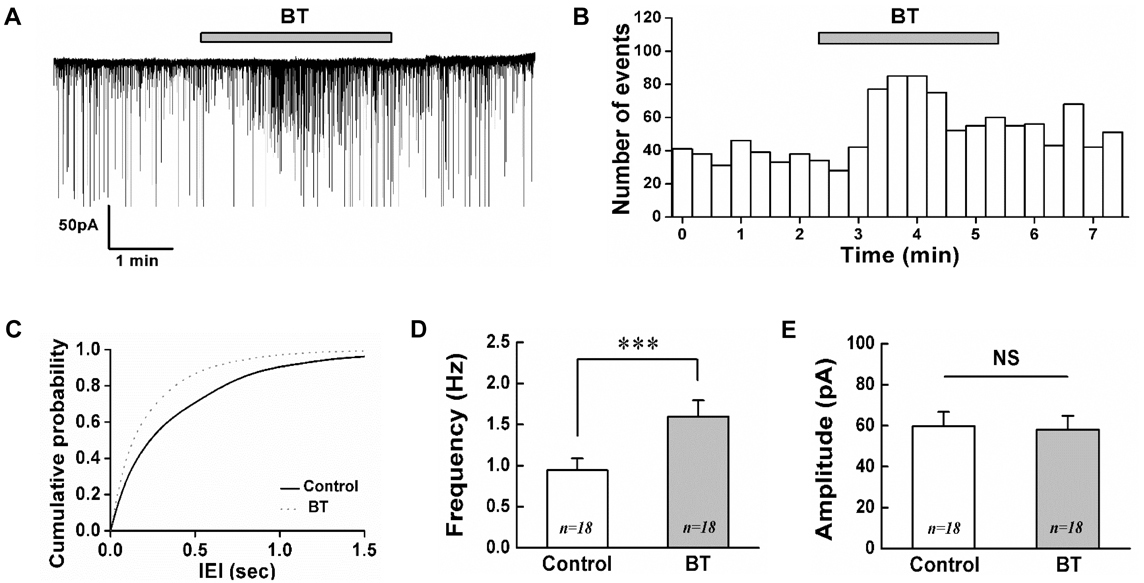
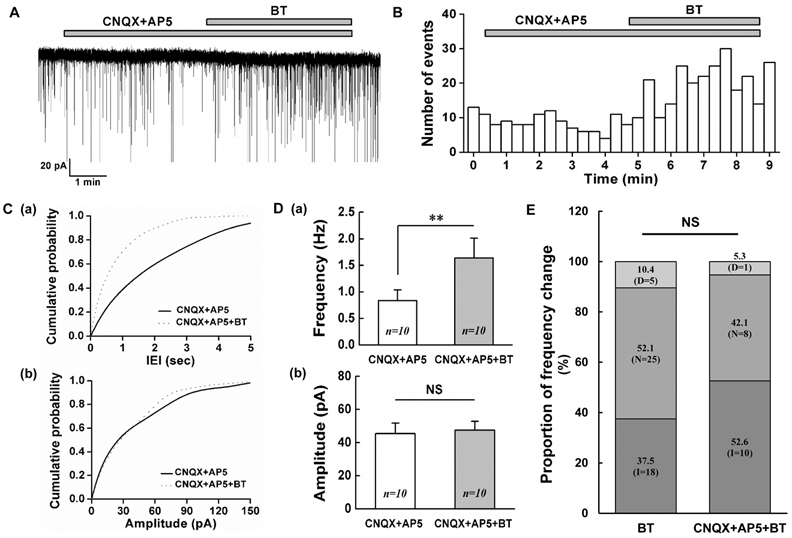
- Botulinum toxin type A (BoNT/A) has been used therapeutically for various conditions including dystonia, cerebral palsy, wrinkle, hyperhidrosis and pain control. The substantia gelatinosa (SG) neurons of the trigeminal subnucleus caudalis (Vc) receive orofacial nociceptive information from primary afferents and transmit the information to higher brain center. Although many studies have shown the analgesic effects of BoNT/A, the effects of BoNT/A at the central nervous system and the action mechanism are not well understood. Therefore, the effects of BoNT/A on the spontaneous postsynaptic currents (sPSCs) in the SG neurons were investigated. In whole cell voltage clamp mode, the frequency of sPSCs was increased in 18 (37.5%) neurons, decreased in 5 (10.4%) neurons and not affected in 25 (52.1%) of 48 neurons tested by BoNT/A (3 nM). Similar proportions of frequency variation of sPSCs were observed in 1 and 10 nM BoNT/A and no significant differences were observed in the relative mean frequencies of sPSCs among 1-10 nM BoNT/A. BoNT/A-induced frequency increase of sPSCs was not affected by pretreated tetrodotoxin (0.5 µM). In addition, the frequency of sIPSCs in the presence of CNQX (10 µM) and AP5 (20 µM) was increased in 10 (53%) neurons, decreased in 1 (5%) neuron and not affected in 8 (42%) of 19 neurons tested by BoNT/A (3 nM). These results demonstrate that BoNT/A increases the frequency of sIPSCs on SG neurons of the Vc at least partly and can provide an evidence for rapid action of BoNT/A at the central nervous system.
Keyword
MeSH Terms
-
6-Cyano-7-nitroquinoxaline-2,3-dione
Animals
Botulinum Toxins*
Botulinum Toxins, Type A*
Brain
Central Nervous System
Cerebral Palsy
Dystonia
Hyperhidrosis
Mice*
Neurons*
Substantia Gelatinosa*
Synaptic Potentials*
Tetrodotoxin
6-Cyano-7-nitroquinoxaline-2,3-dione
Botulinum Toxins
Botulinum Toxins, Type A
Tetrodotoxin
Figure
Reference
-
1. Huang W, Foster JA, Rogachefsky AS. Pharmacology of botulinum toxin. J Am Acad Dermatol. 2000; 43:249–259.
Article2. Pellizzari R, Rossetto O, Schiavo G, Montecucco C. Tetanus and botulinum neurotoxins: mechanism of action and therapeutic uses. Philos Trans R Soc Lond B Biol Sci. 1999; 354:259–268.
Article3. Simpson LL. The origin, structure, and pharmacological activity of botulinum toxin. Pharmacol Rev. 1981; 33:155–188.4. Peng Chen Z, Morris JG Jr, Rodriguez RL, Shukla AW, Tapia-Núñez J, Okun MS. Emerging opportunities for serotypes of botulinum neurotoxins. Toxins (Basel). 2012; 4:1196–1222.
Article5. Schiavo G, Matteoli M, Montecucco C. Neurotoxins affecting neuroexocytosis. Physiol Rev. 2000; 80:717–766.
Article6. Kim DW, Lee SK, Ahnn J. Botulinum toxin as a pain killer: players and actions in antinociception. Toxins (Basel). 2015; 7:2435–2453.7. Pavone F, Luvisetto S. Botulinum neurotoxin for pain management: insights from animal models. Toxins (Basel). 2010; 2:2890–2913.
Article8. Brin MF, Binder WJ, Blitzer A, Schenrock L, Pogoda JM. Botulinum toxin type A for pain and headache. In : Brin MF, Hallett M, Jankovic J, editors. Scientific and therapeutic aspects of botulinum toxin. 1st ed. Philadelphia: Lippincott Williams & Wilkins;2002. p. 233–250.9. Wheeler AH. Therapeutic uses of botulinum toxin. Am Fam Physician. 1997; 55:541–545.10. Truong DD, Jost WH. Botulinum toxin: clinical use. Parkinsonism Relat Disord. 2006; 12:331–355.
Article11. Bach-Rojecky L, Lacković Z. Antinociceptive effect of botulinum toxin type a in rat model of carrageenan and capsaicin induced pain. Croat Med J. 2005; 46:201–208.12. Cui M, Khanijou S, Rubino J, Aoki KR. Subcutaneous administration of botulinum toxin A reduces formalin-induced pain. Pain. 2004; 107:125–133.
Article13. Luvisetto S, Marinelli S, Lucchetti F, Marchi F, Cobianchi S, Rossetto O, Montecucco C, Pavone F. Botulinum neurotoxins and formalin-induced pain: central vs. peripheral effects in mice. Brain Res. 2006; 1082:124–131.
Article14. Lee WH, Shin TJ, Kim HJ, Lee JK, Suh HW, Lee SC, Seo K. Intrathecal administration of botulinum neurotoxin type A attenuates formalin-induced nociceptive responses in mice. Anesth Analg. 2011; 112:228–235.
Article15. Bach-Rojecky L, Lacković Z. Central origin of the antinociceptive action of botulinum toxin type A. Pharmacol Biochem Behav. 2009; 94:234–238.
Article16. Sessle BJ. Acute and chronic craniofacial pain: brainstem mechanisms of nociceptive transmission and neuroplasticity, and their clinical correlates. Crit Rev Oral Biol Med. 2000; 11:57–91.
Article17. Cervero F, Iggo A. The substantia gelatinosa of the spinal cord: a critical review. Brain. 1980; 103:717–772.18. Todd AJ. Neuronal circuitry for pain processing in the dorsal horn. Nat Rev Neurosci. 2010; 11:823–836.
Article19. Matak I, Lacković Z. Botulinum toxin A, brain and pain. Prog Neurobiol. 2014; 119–120:39–59.
Article20. Wheeler A, Smith HS. Botulinum toxins: mechanisms of action, antinociception and clinical applications. Toxicology. 2013; 306:124–146.
Article21. Dressler D. Botulinum toxin therapy: its use for neurological disorders of the autonomic nervous system. J Neurol. 2013; 260:701–713.
Article22. Antonucci F, Rossi C, Gianfranceschi L, Rossetto O, Caleo M. Long-distance retrograde effects of botulinum neurotoxin A. J Neurosci. 2008; 28:3689–3696.
Article23. Matak I, Bach-Rojecky L, Filipović B, Lacković Z. Behavioral and immunohistochemical evidence for central antinociceptive activity of botulinum toxin A. Neuroscience. 2011; 186:201–207.
Article24. Filipović B, Matak I, Bach-Rojecky L, Lacković Z. Central action of peripherally applied botulinum toxin type A on pain and dural protein extravasation in rat model of trigeminal neuropathy. PLoS One. 2012; 7:e29803.
Article25. Kim HJ, Lee GW, Kim MJ, Yang KY, Kim ST, Bae YC, Ahn DK. Antinociceptive effects of transcytosed botulinum neurotoxin type A on trigeminal nociception in rats. Korean J Physiol Pharmacol. 2015; 19:349–355.
Article26. Restani L, Antonucci F, Gianfranceschi L, Rossi C, Rossetto O, Caleo M. Evidence for anterograde transport and transcytosis of botulinum neurotoxin A (BoNT/A). J Neurosci. 2011; 31:15650–15659.
Article27. Guy N, Chalus M, Dallel R, Voisin DL. Both oral and caudal parts of the spinal trigeminal nucleus project to the somatosensory thalamus in the rat. Eur J Neurosci. 2005; 21:741–754.
Article28. Davies AJ, North RA. Electrophysiological and morphological properties of neurons in the substantia gelatinosa of the mouse trigeminal subnucleus caudalis. Pain. 2009; 146:214–221.
Article29. McMahon HT, Foran P, Dolly JO, Verhage M, Wiegant VM, Nicholls DG. Tetanus toxin and botulinum toxins type A and B inhibit glutamate, gamma-aminobutyric acid, aspartate, and met-enkephalin release from synaptosomes. Clues to the locus of action. J Biol Chem. 1992; 267:21338–21343.
Article30. Durham PL, Cady R, Cady R. Regulation of calcitonin gene-related peptide secretion from trigeminal nerve cells by botulinum toxin type A: implications for migraine therapy. Headache. 2004; 44:35–42.
Article31. Morris JL, Jobling P, Gibbins IL. Botulinum neurotoxin A attenuates release of norepinephrine but not NPY from vasoconstrictor neurons. Am J Physiol Heart Circ Physiol. 2002; 283:H2627–H2635.32. Nakov R, Habermann E, Hertting G, Wurster S, Allgaier C. Effects of botulinum A toxin on presynaptic modulation of evoked transmitter release. Eur J Pharmacol. 1989; 164:45–53.
Article33. Beske PH, Scheeler SM, Adler M, McNutt PM. Accelerated intoxication of GABAergic synapses by botulinum neurotoxin A disinhibits stem cell-derived neuron networks prior to network silencing. Front Cell Neurosci. 2015; 9:159.
Article34. Akaike N, Ito Y, Shin MC, Nonaka K, Torii Y, Harakawa T, Ginnaga A, Kozaki S, Kaji R. Effects of A2 type botulinum toxin on spontaneous miniature and evoked transmitter release from the rat spinal excitatory and inhibitory synapses. Toxicon. 2010; 56:1315–1326.
Article35. Drinovac V, Bach-Rojecky L, Lacković Z. Association of antinociceptive action of botulinum toxin type A with GABA-A receptor. J Neural Transm (Vienna). 2014; 121:665–669.
Article36. Matteoli M, Pozzi D, Grumelli C, Condliffe SB, Frassoni C, Harkany T, Verderio C. The synaptic split of SNAP-25: different roles in glutamatergic and GABAergic neurons? Neuroscience. 2009; 158:223–230.
Article37. Waldenström A, Thelin J, Thimansson E, Levinsson A, Schouenborg J. Developmental learning in a pain-related system: evidence for a cross-modality mechanism. J Neurosci. 2003; 23:7719–7725.
Article38. Ruiz-Medina J, Baulies A, Bura SA, Valverde O. Paclitaxel-induced neuropathic pain is age dependent and devolves on glial response. Eur J Pain. 2013; 17:75–85.
Article39. Park SA, Yang EJ, Han SK, Park SJ. Age-related changes in the effects of 5-hydroxytryptamine on substantia gelatinosa neurons of the trigeminal subnucleus caudalis. Neurosci Lett. 2012; 510:78–81.
Article40. Baccei ML, Fitzgerald M. Development of GABAergic and glycinergic transmission in the neonatal rat dorsal horn. J Neurosci. 2004; 24:4749–4757.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inhibitory actions of borneol on the substantia gelatinosa neurons of the trigeminal subnucleus caudalis in mice
- Peripheral Nerve Injury Alters Excitatory and Inhibitory Synaptic Transmission in Rat Spinal Cord Substantia Gelatinosa
- Naringenin modulates GABA mediated response in a sexdependent manner in substantia gelatinosa neurons of trigeminal subnucleus caudalis in immature mice
- Expression of KA1 kainate receptor subunit in the substantia gelatinosa of the trigeminal subnucleus caudalis in mice
- Glycine- and GABA-mimetic Actions of Shilajit on the Substantia Gelatinosa Neurons of the Trigeminal Subnucleus Caudalis in Mice