Korean J Physiol Pharmacol.
2018 Sep;22(5):493-501. 10.4196/kjpp.2018.22.5.493.
The combination of canagliflozin and omega-3 fatty acid ameliorates insulin resistance and cardiac biomarkers via modulation of inflammatory cytokines in type 2 diabetic rats
- Affiliations
-
- 1Department of Pharmacology, College of Pharmacy, Jazan University, Gizan 45142, Saudi Arabia. msafhi@jazanu.edu.sa
- 2Department of Pharmaceutics, College of Pharmacy, Jazan University, Gizan 45142, Saudi Arabia.
- KMID: 2418999
- DOI: http://doi.org/10.4196/kjpp.2018.22.5.493
Abstract
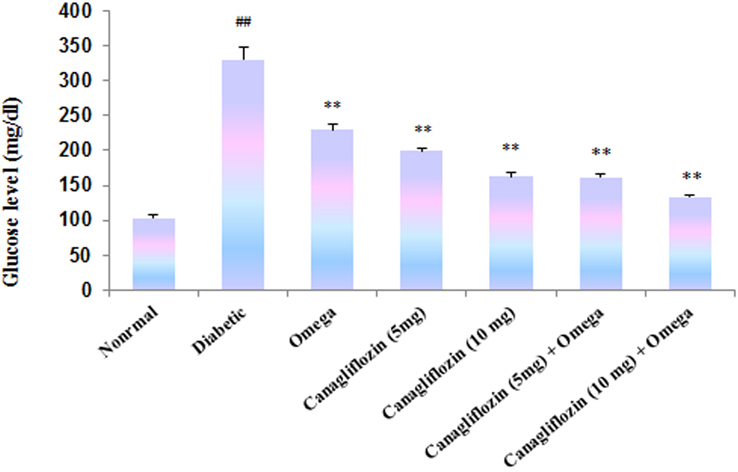
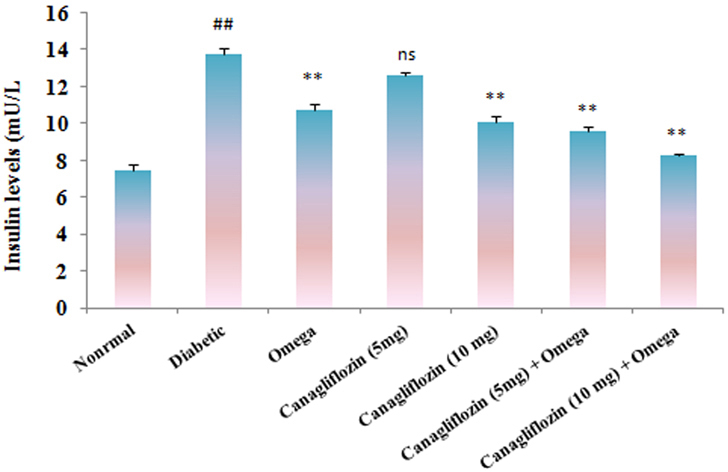
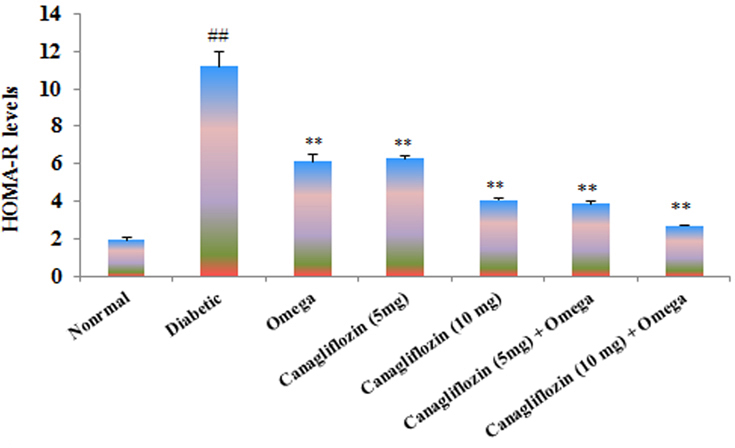
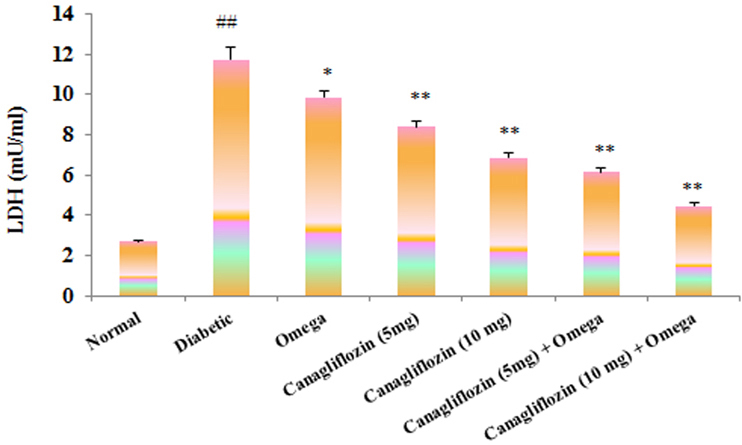
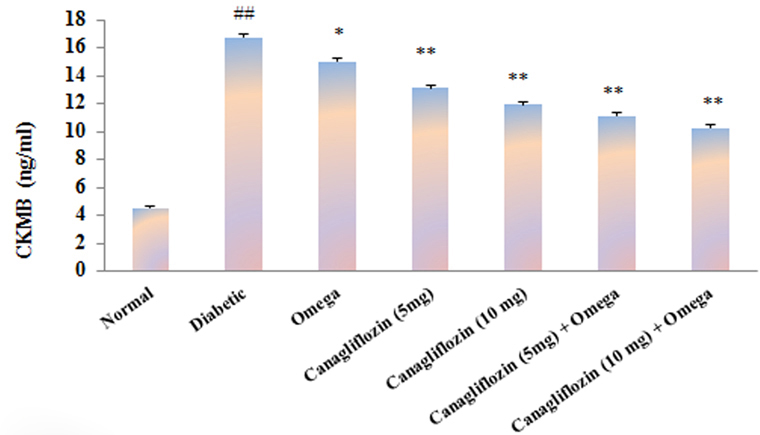
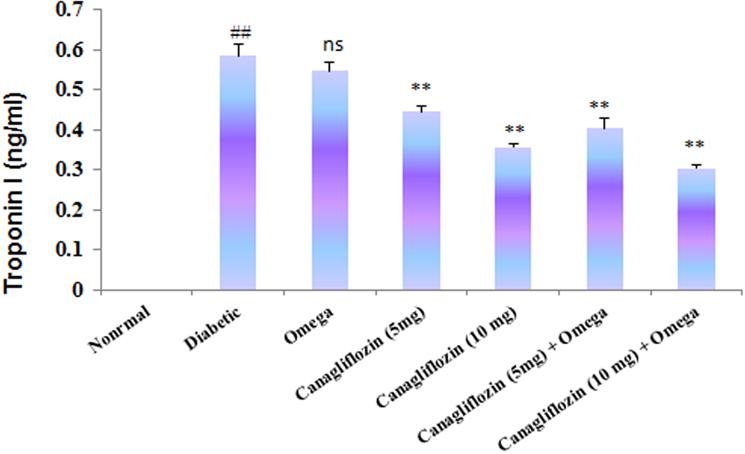
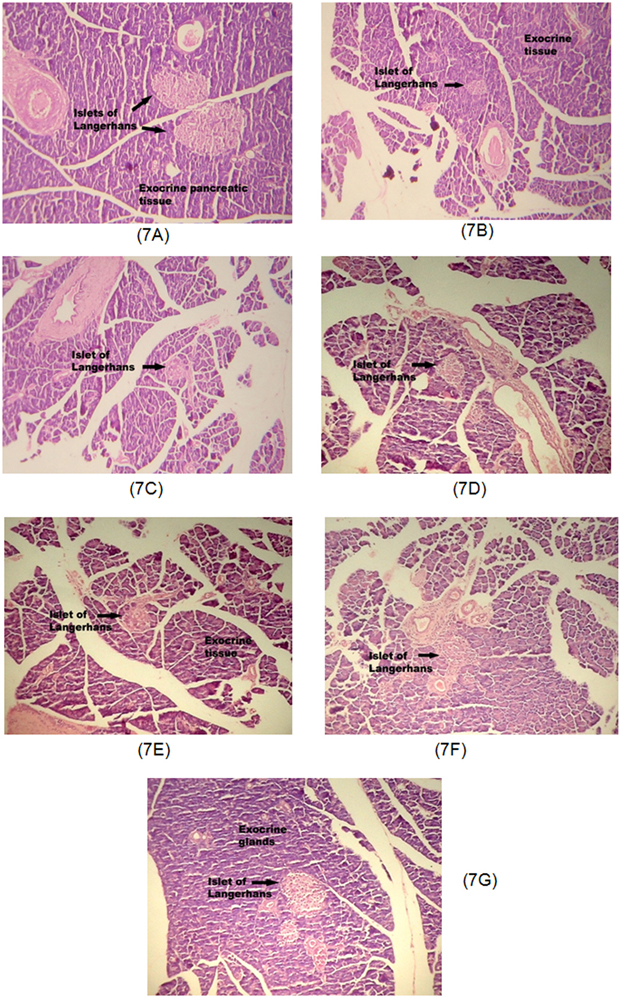
- The present study was carried out with the hypothesis that combination of canagliflozin and omega-3 fatty acid may have potential effect on insulin level, insulin resistance, cardiac biomarkers, inflammatory cytokines and histological studies in type 2 diabetes mellitus (DM). Type 2 DM was induced by injecting nicotinamide (120 mg/kg, i.p.) 15 min before STZ (60 mg/kg) injection. Canagliflozin (5 and 10 mg/kg) and omega-3 fatty acid (300 mg/kg) were given for 28 days after confirmation of diabetes. Biochemical estimations revealed elevated levels of glucose, insulin, HOMA-R and inflammatory cytokines in diabetic group. Daily dosing of alone canagliflozin and omega-3 fatty acid slightly reduced elevated levels of glucose, insulin, HOMA-R and inflammatory cytokines (IL-1β, IL-2, and TNFα), whereas canagliflozin and omega-3 fatty acid combination has reduced these biochemical parameters significantly when compared with diabetic group. Similarly in diabetic group the levels of cardiac biomarkers such as lipid profile, LDH, CKMB and troponin were significantly increased. Elevated levels of cardiac biomarkers were significantly reduced after daily dosing of alone canagliflozin and omega-3 fatty acid. Canagliflozin and omega-3 fatty acid combination has offered better improvement in cardiac biomarkers compared to alone canagliflozin and omega-3 fatty acid. Histopathological analysis also supported the above hypothesis that combination therapy (canagliflozin and omega-3 fatty acid) offered better protection against degenerative changes in β-cells of pancreas as compared to alone treatment with these drugs. Thus the present study revealed that canagliflozin and omega-3 fatty acid can be used as potential combination therapy in type 2 DM along with cardiac complication.
Keyword
MeSH Terms
Figure
Reference
-
1. American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2014; 37:Suppl 1. S81–S90.2. IDF Diabetes Atlas 5th edition [Internet]. International Diabetes Federation;2011. p-7 https://www.idf.org/e-library/epidemiology-research/diabetes-atlas/20-atlas-5th-edition.html.3. Alqurashi KA, Aljabri KS, Bokhari SA. Prevalence of diabetes mellitus in a Saudi community. Ann Saudi Med. 2011; 31:19–23.
Article4. Zimmet P, Thomas CR. Genotype, obesity and cardiovascular disease-has technical and social advancement outstripped evolution? J Intern Med. 2003; 254:114–125.5. Barrett-Connor EL, Cohn BA, Wingard DL, Edelstein SL. Why is diabetes mellitus a stronger risk factor for fatal ischemic heart disease in women than in men? The Rancho Bernardo Study. JAMA. 1991; 265:627–631.
Article6. Trost S, LeWinter M. Diabetic cardiomyopathy. Curr Treat Options Cardiovasc Med. 2001; 3:481–492.
Article7. Alexandraki K, Piperi C, Kalofoutis C, Singh J, Alaveras A, Kalofoutis A. Inflammatory process in type 2 diabetes: The role of cytokines. Ann N Y Acad Sci. 2006; 1084:89–117.
Article8. Banerjee M, Saxena M. Interleukin-1 (IL-1) family of cytokines: role in type 2 diabetes. Clin Chim Acta. 2012; 413:1163–1170.
Article9. Dandona P, Aljada A, Bandyopadhyay A. Inflammation: the link between insulin resistance, obesity and diabetes. Trends Immunol. 2004; 25:4–7.
Article10. Mahmoud F, Al-Ozairi E. Inflammatory cytokines and the risk of cardiovascular complications in type 2 diabetes. Dis Markers. 2013; 35:235–241.
Article11. Lee MW, Park JK, Hong JW, Kim KJ, Shin DY, Ahn CW, Song YD, Cho HK, Park SW, Lee EJ. Beneficial effects of omega-3 fatty acids on low density lipoprotein particle size in patients with type 2 diabetes already under statin therapy. Diabetes Metab J. 2013; 37:207–211.
Article12. Eissa LA, Abdel-Rahman N, Eraky SM. Effects of omega-3 fatty acids and pioglitazone combination on insulin resistance through fibroblast growth factor 21 in type 2 diabetes mellitus. Egypt J Basic Appl Sci. 2015; 2:75–86.
Article13. Hasan FM, Alsahli M, Gerich JE. SGLT inhibitors in the treatment of type 2 diabetes. Diabetes Res Clin Pract. 2014; 104:297–322.14. NRCC (National Research Council). Guide for the care and use of laboratory animals. 7th ed. Washington DC: National Academy Press;1996.15. Shirwaikar A, Rajendran K, Punitha IS. Antidiabetic activity of alcoholic stem extract of Coscinium fenestratum in streptozotocin-nicotinamide induced type 2 diabetic rats. J Ethnopharmacol. 2005; 97:369–374.
Article16. Barham D, Trinder P. An improved colour reagent for the determination of blood glucose by the oxidase system. Analyst. 1972; 97:142–145.
Article17. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985; 28:412–419.18. Bucolo G, David H. Quantitative determination of serum triglycerides by the use of enzymes. Clin Chem. 1973; 19:476–482.
Article19. Wybenga DR, Pileggi VJ, Dirstine PH, Di Giorgio J. Direct manual determination of serum total cholesterol with a single stable reagent. Clin Chem. 1970; 16:980–984.
Article20. Warnick GR, Nguyen T, Albers AA. Comparison of improved precipitation methods for quantification of high-density lipoprotein cholesterol. Clin Chem. 1985; 31:217–222.
Article21. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972; 18:499–502.
Article22. Vanderlinde RE. Measurement of total lactate dehydrogenase activity. Ann Clin Lab Sci. 1985; 15:13–31.23. Yki-Järvinen H. Management of type 2 diabetes mellitus and cardiovascular risk: lessons from intervention trials. Drugs. 2000; 60:975–983.24. Goldstein BJ. Insulin resistance as the core defect in type 2 diabetes mellitus. Am J Cardiol. 2002; 90:3G–10G.
Article25. Bonora E, Zavaroni I, Coscelli C, Butturini U. Decreased hepatic insulin extraction in subjects with mild glucose intolerance. Metabolism. 1983; 32:438–446.
Article26. Olefsky JM, Revers RR, Prince M, Henry RR, Garvey WT, Scarlett JA, Kolterman OG. Insulin resistance in non-insulin dependent (type II) and insulin dependent (type I) diabetes mellitus. Adv Exp Med Biol. 1985; 189:176–205.
Article27. Lombardo YB, Chicco AG. Effects of dietary polyunsaturated n-3 fatty acids on dyslipidemia and insulin resistance in rodents and humans. A review. J Nutr Biochem. 2006; 17:1–13.
Article28. Fasshauer M, Paschke R. Regulation of adipocytokines and insulin resistance. Diabetologia. 2003; 46:1594–1603.
Article29. McArdle MA, Finucane OM, Connaughton RM, McMorrow AM, Roche HM. Mechanisms of obesity-induced inflammation and insulin resistance: insights into the emerging role of nutritional strategies. Front Endocrinol (Lausanne). 2013; 4:52.
Article30. Boyman O, Sprent J. The role of interleukin-2 during homeostasis and activation of the immune system. Nat Rev Immunol. 2012; 12:180–190.
Article31. Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW Jr. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003; 112:1796–1808.
Article32. Endo J, Arita M. Cardioprotective mechanism of omega-3 polyunsaturated fatty acids. J Cardiol. 2016; 67:22–27.
Article33. De Caterina R, Madonna R, Bertolotto A, Schmidt EB. n-3 fatty acids in the treatment of diabetic patients: biological rationale and clinical data. Diabetes Care. 2007; 30:1012–1026.
Article34. Bode B, Stenlöf K, Sullivan D, Fung A, Usiskin K. Efficacy and safety of canagliflozin treatment in older subjects with type 2 diabetes mellitus: a randomized trial. Hosp Pract (1995). 2013; 41:72–84.
Article35. Jonkers IJ, Mohrschladt MF, Westendorp RG, van der Laarse A, Smelt AH. Severe hypertriglyceridemia with insulin resistance is associated with systemic inflammation: reversal with bezafibrate therapy in a randomized controlled trial. Am J Med. 2002; 112:275–280.
Article36. Feingold KR, Grunfeld C. Tumor necrosis factor-alpha stimulates hepatic lipogenesis in the rat in vivo. J Clin Invest. 1987; 80:184–190.
Article37. Ali F, Naqvi SA, Bismillah M, Wajid N. Comparative analysis of biochemical parameters in diabetic and non-diabetic acute myocardial infarction patients. Indian Heart J. 2016; 68:325–331.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Rosiglitazone on Inflammation in Otsuka Long-Evans Tokushima Fatty Rats
- Omega-3 Index as a Risk Factor for Cardiovascular Disease and Its Application to Korean Population
- The Role of Inflammatory Mediators in the Pathogenesis of Nonalcoholic Fatty Liver Disease
- Evaluation of fish oil-rich in MUFAs for anti-diabetic and anti-inflammation potential in experimental type 2 diabetic rats
- Effects of phlorizin and acipimox on insulin resistance in STZ-diabetic rats