Investig Clin Urol.
2018 Jul;59(4):252-256. 10.4111/icu.2018.59.4.252.
Measuring urinary 8-hydroxy-2′-deoxyguanosine and malondialdehyde levels in women with overactive bladder
- Affiliations
-
- 1Department of Nutrition and Dietetics, Artvin Coruh University Faculty of Healthy Sciences, Artvin, Turkey. edadokumacioglu@yahoo.com
- 2Department of Urology, Hopa State Hospital, Artvin, Turkey.
- 3Department of Medical Biochemistry, Hopa State Hospital, Artvin, Turkey.
- 4Department of Physiology, Ordu University Faculty of Medicine, Ordu, Turkey.
- 5Department of Medical Biochemistry, Karadeniz Teknik University Faculty of Medicine, Trabzon, Turkey.
- 6Department of Biostatistics and Medical Informatics, Ordu University Faculty of Medicine, Ordu, Turkey.
- KMID: 2414758
- DOI: http://doi.org/10.4111/icu.2018.59.4.252
Abstract
- PURPOSE
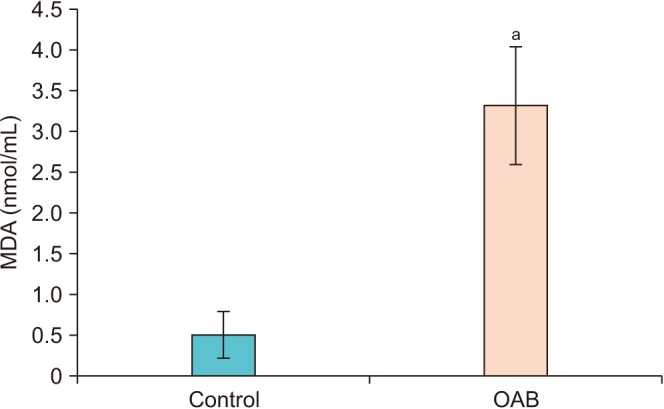
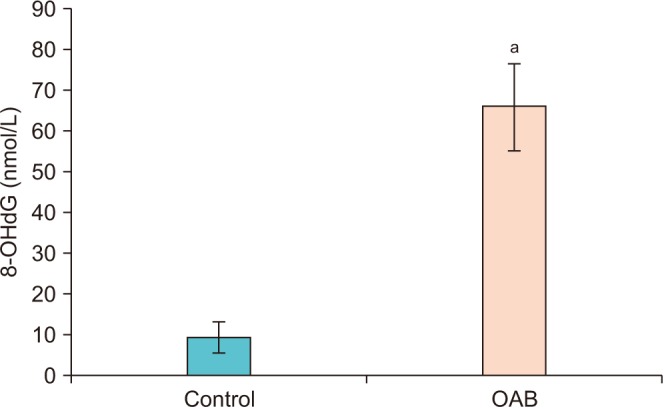
In this study, we aimed to explain the role of oxidative stress in women with overactive bladder (OAB) by investigating the levels of 8-hydroxy-2"²-deoxyguanosine (8-OHdG), a marker of oxidative DNA damage, and malondialdehyde (MDA), an indicator of lipid peroxidation.
MATERIALS AND METHODS
A total of 90 women were included in the study: 45 female patients diagnosed with OAB at Hopa State Hospital Urology Polyclinic and 45 healthy women without any metabolic or neurologic disease. Levels of MDA and 8-OHdG were measured in 24-hour urine samples for all subjects.
RESULTS
Urinary levels of MDA and 8-OHdG were significantly higher in the OAB group than in the control group (p < 0.001). A significant positive correlation (p<0.001) was found between the measurements of 8-OHdG and MDA.
CONCLUSIONS
Oxidative stress may be important in the pathophysiology of OAB, because levels of 8-OHdG and MDA are increased. Increased levels of 8-OHdG may be due to damaged nuclear and mitochondrial DNA as a result of oxidative attacks caused by free radicals. Nevertheless, further randomized and prospective studies with larger patient populations are needed.
MeSH Terms
Figure
Reference
-
1. Abrams P, Cardozo L, Fall M, Griffiths D, Rosier P, Ulmsten U, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Neurourol Urodyn. 2002; 21:167–178. PMID: 11857671.
Article2. Stewart WF, Van Rooyen JB, Cundiff GW, Abrams P, Herzog AR, Corey R, et al. Prevalence and burden of overactive bladder in the United States. World J Urol. 2003; 20:327–336. PMID: 12811491.
Article3. Steers WD. Pathophysiology of overactive bladder and urge urinary incontinence. Rev Urol. 2002; 4(Suppl 4):S7–S18. PMID: 16986023.4. Nitti VW, Blaivas JG. Urinary incontinence: epidemiology, pathophysiology, evaluation, and management overview. In : Wein AJ, Kavoussi LR, Novick AC, Partin AW, Peters CA, editors. Campbell-Walsh urology. 9th ed. Philadelphia: Saunders Elsevier;2007.5. Abrams P, Cardozo L, Khoury S, Wein A. Incontinence: The Third WHO International Consultation on Incontinence. Plymouth (England): Health Publications;2005. p. 26–32.6. Nielsen F, Mikkelsen BB, Nielsen JB, Andersen HR, Grandjean P. Plasma malondialdehyde as biomarker for oxidative stress: reference interval and effects of life-style factors. Clin Chem. 1997; 43:1209–1214. PMID: 9216458.
Article7. Özcan O, Erdal H, Çakırca G, Yönden Z. Oxidative stress and its impacts on intracellular lipids, proteins and DNA. J Clin Exp Invest. 2015; 6:331–336.
Article8. De Martinis BS, de Lourdes Pires Bianchi M. Methodology for urinary 8-hydroxy-2′-deoxyguanosine analysis by HPLC with electrochemical detection. Pharmacol Res. 2002; 46:129–131. PMID: 12220951.
Article9. Dizdaroglu M. Facts about the artifacts in the measurement of oxidative DNA base damage by gas chromatography-mass spectrometry. Free Radic Res. 1998; 29:551–563. PMID: 10098459.
Article10. Helbock HJ, Beckman KB, Ames BN. 8-Hydroxydeoxyguanosine and 8-hydroxyguanine as biomarkers of oxidative DNA damage. Methods Enzymol. 1999; 300:156–166. PMID: 9919519.
Article11. Williams GM, Jeffrey AM. Oxidative DNA damage: endogenous and chemically induced. Regul Toxicol Pharmacol. 2000; 32:283–292. PMID: 11162722.
Article12. Kasai H. Analysis of a form of oxidative DNA damage, 8-Hydroxy-2’ deoxyguanosine, as a marker of celluler cellular oxidative stress during carcinogensis. Mutat Res. 1997; 387:147–163. PMID: 9439711.13. Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979; 95:351–358. PMID: 36810.
Article14. Zhao Y, Levin SS, Wein AJ, Levin RM. Correlation of ischemia/reperfusion or partial outlet obstruction-induced spectrin proteolysis by calpain with contractile dysfunction in rabbit bladder. Urology. 1997; 49:293–300. PMID: 9037302.
Article15. Ouslander JG. Management of overactive bladder. N Engl J Med. 2004; 350:786–799. PMID: 14973214.
Article16. Teloken C, Caraver F, Weber FA, Teloken PE, Moraes JF, Sogari PR, et al. Overactive bladder: prevalence and implications in Brazil. Eur Urol. 2006; 49:1087–1092. PMID: 16497431.
Article17. Dambros M, Dambros MC, Lorenzetti F, Dassen EL, van Koeveringe GA. The use of hypochlorous acid as a model for investigating bladder overactivity. Int Braz J Urol. 2014; 40:408–413. PMID: 25010308.
Article18. Huang YB, Lin MW, Chao Y, Huang CT, Tsai YH, Wu PC. Anti-oxidant activity and attenuation of bladder hyperactivity by the flavonoid compound kaempferol. Int J Urol. 2014; 21:94–98. PMID: 23634640.
Article19. García JJ, Reiter RJ, Guerrero JM, Escames G, Yu BP, Oh CS, et al. Melatonin prevents changes in microsomal membrane fluidity during induced lipid peroxidation. FEBS Lett. 1997; 408:297–300. PMID: 9188780.20. Bisogni S, Ferreira FT, Amstalden Neto A, Chiarelli LO, Ortiz V. Influence of oxidative stress on inducing micturition dysfunction following chronic infravesical obstruction and the protective role of an antioxidant diet: association of in vivo and in vitro studies in rats. Int Braz J Urol. 2012; 38:552–560. PMID: 22951166.21. Alexandre EC, Calmasini FB, de Oliveira MG, Silva FH, da Silva CPV, André DM, et al. Chronic treatment with resveratrol improves overactive bladder in obese mice via antioxidant activity. Eur J Pharmacol. 2016; 788:29–36. PMID: 27316789.
Article22. Topol T, Schuler C, Leggett RE, Hydery T, Benyamin S, Levin RM. Effect of solifenacin plus and minus antioxidant supplements on the response to experimental outlet obstruction and overactive bladder dysfunction in rabbits: part 2. Urol Sci. 2011; 22:141–146.23. Kulaksız G, Sancar A. Nucleotide excision repair and cancer. Turk J Biochem. 2007; 32:104–111.24. Cooke MS, Evans MD, Dizdaroglu M, Lunec J. Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J. 2003; 17:1195–1214. PMID: 12832285.
Article25. Pilger A, Rüdiger HW. 8-Hydroxy-2′-deoxyguanosine as a marker of oxidative DNA damage related to occupational and environmental exposures. Int Arch Occup Environ Health. 2006; 80:1–15. PMID: 16685565.
Article26. Yang JH, Siroky MB, Yalla SV, Azadzoi KM. Mitochondrial stress and activation of PI3K and Akt survival pathway in bladder ischemia. Res Rep Urol. 2017; 9:93–100. PMID: 28652996.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Development of an Improved Animal Model of Overactive Bladder: Transperineal Ligation versus Transperitoneal Ligation in Male Rats
- Association of Urinary Urgency and Delay Time of Micturition in Women With Overactive Bladder
- Overactive Bladder and Urinary Incontinence in Adult Women: Prevalence and Effects on Daily Life and Sexual Activity
- Overactive Bladder
- Age Related Changes of Voiding Patterns in Women with Overactive Bladder