Lab Anim Res.
2018 Jun;34(2):58-64. 10.5625/lar.2018.34.2.58.
Decrease in glucose transporter 1 levels and translocation of glucose transporter 3 in the dentate gyrus of C57BL/6 mice and gerbils with aging
- Affiliations
-
- 1Department of Anatomy, College of Veterinary Medicine and Institute of Veterinary Science, Kangwon National University, Chuncheon, Korea. jhchoi@kangwon.ac.kr
- 2Department of Anatomy and Cell Biology, College of Veterinary Medicine, and Research Institute for Veterinary Science, Seoul National University, Seoul, Korea.
- 3Undergraduate School, College of Veterinary Medicine, Seoul National University, Seoul, Korea.
- 4Department of Biochemistry and Molecular Biology, Research Institute of Oral Sciences, College of Dentistry, Gangneung-Wonju National University, Gangneung, Korea.
- 5Department of Veterinary Internal Medicine and Geriatrics, College of Veterinary Medicine and Institute of Veterinary Science, Kangwon National University, Chuncheon, Korea.
- 6Department of Medical Education, Kangwon National University School of Medicine, Chuncheon, Korea.
- KMID: 2413844
- DOI: http://doi.org/10.5625/lar.2018.34.2.58
Abstract
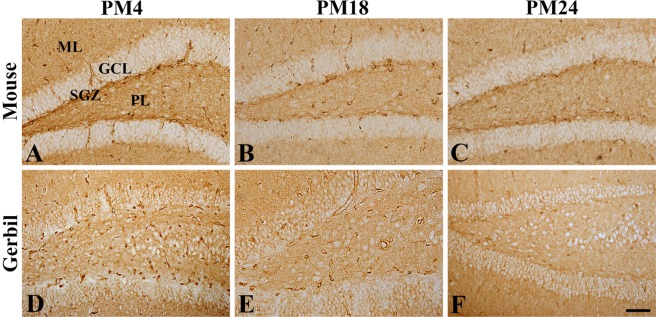
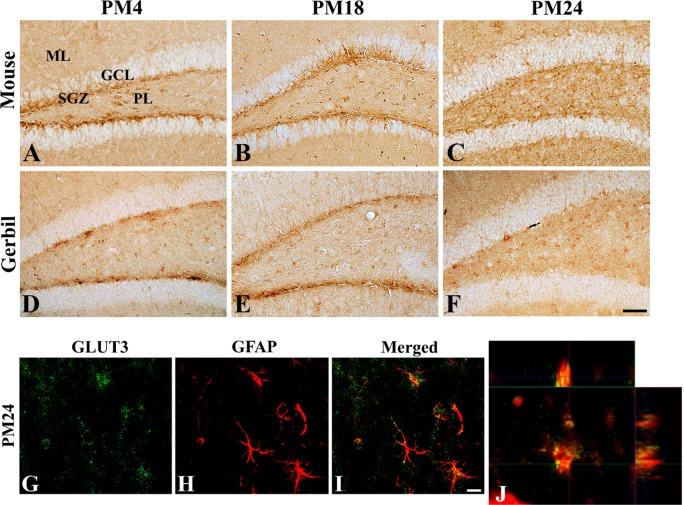
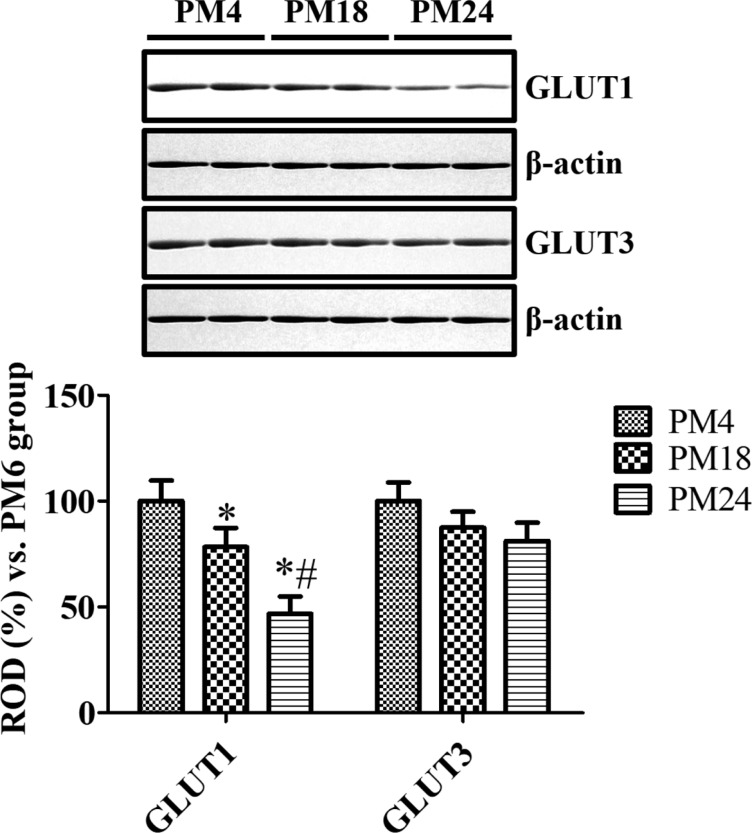
- In the present study, we compared the cell-specific expression and changes protein levels in the glucose transporters (GLUTs) 1 and 3, the major GLUTs in the mouse and gerbil brains using immunohistochemistry and Western blot analysis. In both mouse and gerbils, GLUT1 immunoreactivity was mainly found in the blood vessels in the dentate gyrus, while GLUT3 immunoreactivity was detected in the subgranular zone and the molecular layer of the dentate gyrus. GLUT1-immunoreactivity in blood vessels and GLUT1 protein levels were significantly decreased with age in the mice and gerbils, respectively. In addition, few GLUT3-immunoreactive cells were found in the subgranular zone in aged mice and gerbils, but GLUT3-immunoreactivity was abundantly found in the polymorphic layer of dentate gyrus in mice and gerbils with a dot-like pattern. Based on the double immunofluorescence study, GLUT3-immunoreactive structures in gerbils were localized in the glial fibrillary acidic protein-immunoreactive astrocytes in the dentate gyrus. Western blot analysis showed that GLUT3 expression in the hippocampal homogenates was slightly, although not significantly, decreased with age in mice and gerbils, respectively. These results indicate that the reduction in GLUT1 in the blood vessels of dentate gyrus and GLUT3 in the subgranular zone of dentate gyrus may be associated with the decrease in uptake of glucose into brain and neuroblasts in the dentate gyrus. In addition, the expression of GLUT3 in the astrocytes in polymorphic layer of dentate gyrus may be associated with metabolic changes in glucose in aged hippocampus.
Keyword
MeSH Terms
Figure
Reference
-
1. Clark DD, Sokoloff L. Circulation and energy metabolism of the brain. In : Siegel GJ, Agranoff BW, Albers RW, Fisher SK, Uhler MD, editors. Basic neurochemistry: molecular, cellular and medical aspects. 6th ed. Philadelphia: Lippincott-Raven;1999. p. 637–670.2. Flier JS, Mueckler M, McCall AL, Lodish HF. Distribution of glucose transporter messenger RNA transcripts in tissues of rat and man. J Clin Invest. 1987; 79(2):657–661. PMID: 3027132.
Article3. Yano H, Seino Y, Inagaki N, Hinokio Y, Yamamoto T, Yasuda K, Masuda K, Someya Y, Imura H. Tissue distribution and species difference of the brain type glucose transporter (GLUT3). Biochem Biophys Res Commun. 1991; 174(2):470–477. PMID: 1704223.
Article4. Nagamatsu S, Kornhauser JM, Burant CF, Seino S, Mayo KE, Bell GI. Glucose transporter expression in brain. cDNA sequence of mouse GLUT3, the brain facilitative glucose transporter isoform, and identification of sites of expression by in situ hybridization. J Biol Chem. 1992; 267(1):467–472. PMID: 1730609.
Article5. Gerhart DZ, LeVasseur RJ, Broderius MA, Drewes LR. Glucose transporter localization in brain using light and electron immunocytochemistry. J Neurosci Res. 1989; 22(4):464–472. PMID: 2668543.
Article6. Pardridge WM, Boado RJ, Farrell CR. Brain-type glucose transporter (GLUT-1) is selectively localized to the blood-brain barrier. Studies with quantitative western blotting and in situ hybridization. J Biol Chem. 1990; 265(29):18035–18040. PMID: 2211679.
Article7. Dick AP, Harik SI, Klip A, Walker DM. Identification and characterization of the glucose transporter of the blood-brain barrier by cytochalasin B binding and immunological reactivity. Proc Natl Acad Sci USA. 1984; 81(22):7233–7237. PMID: 6150484.
Article8. Heilig CW, Saunders T, Brosius FC 3rd, Moley K, Heilig K, Baggs R, Guo L, Conner D. Glucose transporter-1-deficient mice exhibit impaired development and deformities that are similar to diabetic embryopathy. Proc Natl Acad Sci USA. 2003; 100(26):15613–15618. PMID: 14673082.
Article9. Jensen PJ, Gitlin JD, Carayannopoulos MO. GLUT1 deficiency links nutrient availability and apoptosis during embryonic development. J Biol Chem. 2006; 281(19):13382–13387. PMID: 16543226.
Article10. Zhao Y, Fung C, Shin D, Shin BC, Thamotharan S, Sankar R, Ehninger D, Silva A, Devaskar SU. Neuronal glucose transporter isoform 3 deficient mice demonstrate features of autism spectrum disorders. Mol Psychiatry. 2010; 15(3):286–299. PMID: 19506559.
Article11. Willis MW, Ketter TA, Kimbrell TA, George MS, Herscovitch P, Danielson AL, Benson BE, Post RM. Age, sex and laterality effects on cerebral glucose metabolism in healthy adults. Psychiatry Res. 2002; 114(1):23–37. PMID: 11864807.
Article12. Zhang X, Li G, Guo L, Nie K, Jia Y, Zhao L, Yu J. Age-related alteration in cerebral blood flow and energy failure is correlated with cognitive impairment in the senescence-accelerated prone mouse strain 8 (SAMP8). Neurol Sci. 2013; 34(11):1917–1924. PMID: 23563860.
Article13. Dornbos D 3rd, Zwagerman N, Guo M, Ding JY, Peng C, Esmail F, Sikharam C, Geng X, Guthikonda M, Ding Y. Preischemic exercise reduces brain damage by ameliorating metabolic disorder in ischemia/reperfusion injury. J Neurosci Res. 2013; 91(6):818–827. PMID: 23553672.
Article14. Iwabuchi S, Kawahara K, Harata NC. Effects of pharmacological inhibition of AMP-activated protein kinase on GLUT3 expression and the development of ischemic tolerance in astrocytes. Neurosci Res. 2014; 84:68–71. PMID: 24815515.
Article15. Iwabuchi S, Kawahara K. Inducible astrocytic glucose transporter-3 contributes to the enhanced storage of intracellular glycogen during reperfusion after ischemia. Neurochem Int. 2011; 59(2):319–325. PMID: 21703319.
Article16. Choeiri C, Staines W, Messier C. Immunohistochemical localization and quantification of glucose transporters in the mouse brain. Neuroscience. 2002; 111(1):19–34. PMID: 11955709.
Article17. Fattoretti P, Bertoni-Freddari C, Casoli T, Di Stefano G, Solazzi M, Giorgetti B. Decreased expression of glucose transport protein (Glut3) in aging and vitamin E deficiency. Ann N Y Acad Sci. 2002; 973:293–296. PMID: 12485881.
Article18. Jung HY, Yim HS, Yoo DY, Kim JW, Chung JY, Seong JK, Yoon YS, Kim DW, Hwang IK. Postnatal changes in glucose transporter 3 expression in the dentate gyrus of the C57BL/6 mouse model. Lab Anim Res. 2016; 32(1):1–7. PMID: 27051437.
Article19. Fox JG, Barthold SW, Davisson MT, Newcomer CE, Quimby FW, Smith AL. The Mouse in Biomedical Research: Normative Biology, Husbandry, and Models. 2nd ed. Burlington: American College of Laboratory Animal Medicine (Elsevier);2007. p. 1–759.20. Franklin KBJ, Paxinos G. The Mouse Brain in Stereotaxic Coordinates. San Diego: Academic Press;1997. p. 1–190.21. Loskota WJ, Lomax P, Verity MA. A Stereotaxic Atlas of the Mongolian Gerbil Brain (Meriones unguiculatus). Ann Arbor: Ann Arbor Science;1974. p. 1–157.22. Hahn KR, Jung HY, Yoo DY, Kim JW, Kim YH, Jo YK, Kim GA, Chung JY, Choi JH, Hwang IK, Jang G, Yoon YS. Immunohistochemical localization of glucose transporter 1 and 3 in the scrotal and abdominal testes of a dog. Lab Anim Res. 2017; 33(2):114–118. PMID: 28747976.
Article23. Deary IJ, Sommerfield AJ, McAulay V, Frier BM. Moderate hypoglycaemia obliterates working memory in humans with and without insulin treated diabetes. J Neurol Neurosurg Psychiatry. 2003; 74(2):278–279.
Article24. McNay EC. The impact of recurrent hypoglycemia on cognitive function in aging. Neurobiol Aging. 2005; 26(Suppl 1):76–79. PMID: 16236383.
Article25. Meier-Ruge W, Kolbe M, Sattler J. Investigations of the cholinergic deficit hypothesis in the hippocampus of the aged rat brain with physostigmine and scopolamine. Arch Gerontol Geriatr. 1991; 12(2-3):239–251. PMID: 15374451.
Article26. McNay EC, Gold PE. Age-related differences in hippocampal extracellular fluid glucose concentration during behavioral testing and following systemic glucose administration. J Gerontol A Biol Sci Med Sci. 2001; 56(2):B66–B71. PMID: 11213269.
Article27. Horwood N, Davies DC. Immunolabelling of hippocampal microvessel glucose transporter protein is reduced in Alzheimer's disease. Virchows Arch. 1994; 425(1):69–72. PMID: 7921416.
Article28. Kalaria RN, Harik SI. Reduced glucose transporter at the blood-brain barrier and in cerebral cortex in Alzheimer disease. J Neurochem. 1989; 53(4):1083–1088. PMID: 2769254.
Article29. Simpson IA, Chundu KR, Davies-Hill T, Honer WG, Davies P. Decreased concentrations of GLUT1 and GLUT3 glucose transporters in the brains of patients with Alzheimer's disease. Ann Neurol. 1994; 35(5):546–551. PMID: 8179300.
Article30. Hooijmans CR, Graven C, Dederen PJ, Tanila H, van Groen T, Kiliaan AJ. Amyloid beta deposition is related to decreased glucose transporter-1 levels and hippocampal atrophy in brains of aged APP/PS1 mice. Brain Res. 2007; 1181:93–103. PMID: 17916337.
Article31. Yoo DY, Lee KY, Park JH, Jung HY, Kim JW, Yoon YS, Won MH, Choi JH, Hwang IK. Glucose metabolism and neurogenesis in the gerbil hippocampus after transient forebrain ischemia. Neural Regen Res. 2016; 11(8):1254–1259. PMID: 27651772.
Article32. Rajakumar A, Thamotharan S, Raychaudhuri N, Menon RK, Devaskar SU. Trans-activators regulating neuronal glucose transporter isoform-3 gene expression in mammalian neurons. J Biol Chem. 2004; 279(25):26768–26779. PMID: 15054091.
Article33. Thoenen H. Neurotrophins and neuronal plasticity. Science. 1995; 270(5236):593–598. PMID: 7570017.
Article34. Finkbeiner S, Tavazoie SF, Maloratsky A, Jacobs KM, Harris KM, Greenberg ME. CREB: a major mediator of neuronal neurotrophin responses. Neuron. 1997; 19(5):1031–1047. PMID: 9390517.
Article35. Wang BW, Hok V, Della-Chiesa A, Callaghan C, Barlow S, Tsanov M, Bechara R, Irving E, Virley DJ, Upton N, O'Mara SM. Rosiglitazone enhances learning, place cell activity, and synaptic plasticity in middle-aged rats. Neurobiol Aging. 2012; 33(4):835.e13–835.e30.
Article36. Wu Y, Zhang AQ, Yew DT. Age related changes of various markers of astrocytes in senescence-accelerated mice hippocampus. Neurochem Int. 2005; 46(7):565–574. PMID: 15843051.
Article37. Lee CY, Dallérac G, Ezan P, Anderova M, Rouach N. Glucose tightly controls morphological and functional properties of astrocytes. Front Aging Neurosci. 2016; 8:82. PMID: 27148048.
Article38. Yu S, Zhao T, Guo M, Fang H, Ma J, Ding A, Wang F, Chan P, Fan M. Hypoxic preconditioning up-regulates glucose transport activity and glucose transporter (GLUT1 and GLUT3) gene expression after acute anoxic exposure in the cultured rat hippocampal neurons and astrocytes. Brain Res. 2008; 1211:22–29. PMID: 18474279.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Postnatal changes in glucose transporter 3 expression in the dentate gyrus of the C57BL/6 mouse model
- Study on the Mechanism of the Translocation of Glucose Transporter by Insulin
- Response: Predictors of the Therapeutic Efficacy and Consideration of the Best Combination Therapy of Sodium-Glucose Co-transporter 2 Inhibitors (Diabetes Metab J 2019;43:158–73)
- Letter: Predictors of the Therapeutic Efficacy and Consideration of the Best Combination Therapy of Sodium-Glucose Co-transporter 2 Inhibitors (Diabetes Metab J 2019;43:158–73)
- Dietary zinc supplementation in highfat diet-induced obese mice: Effects on the skeletal muscle ZIP7 expression and blood glucose regulation