Yonsei Med J.
2018 Jul;59(5):588-594. 10.3349/ymj.2018.59.5.588.
CpG Island Methylator Phenotype and Methylation of Wnt Pathway Genes Together Predict Survival in Patients with Colorectal Cancer
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea.
- 2Department of Medicine, Graduate School of Yonsei University, Seoul, Korea. vvswm513@yuhs.ac
- 3Department of Internal Medicine, Yonsei Cancer Center, Yonsei University College of Medicine, Seoul, Korea.
- 4Department of Surgery, Yonsei University College of Medicine, Seoul, Korea.
- KMID: 2412649
- DOI: http://doi.org/10.3349/ymj.2018.59.5.588
Abstract
- PURPOSE
Dysregulation of the Wnt pathway is a crucial step in the tumorigenesis of colorectal cancer (CRC). This study aimed to determine whether DNA methylation of Wnt pathway genes helps predict treatment response and survival in patients with metastatic or recurrent CRC.
MATERIALS AND METHODS
We retrospectively collected primary tumor tissues from 194 patients with metastatic or recurrent CRC. Pyrosequencing was used to examine the methylation of 10 CpG island loci in DNA extracted from formalin-fixed paraffin-embedded specimens. To elucidate the predictive role of DNA methylation markers, Kaplan-Meier survival estimation and Cox regression were performed for progression-free survival and overall survival (OS).
RESULTS
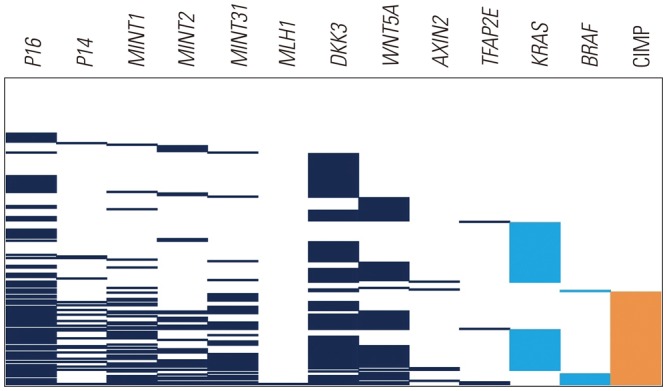
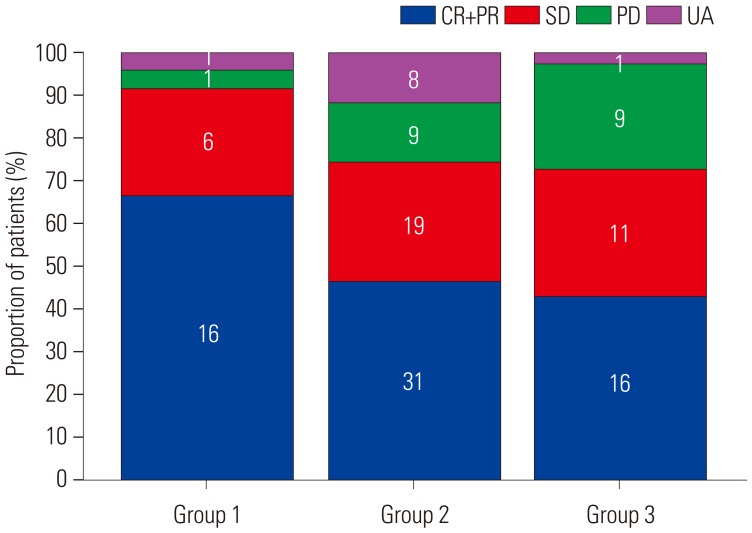
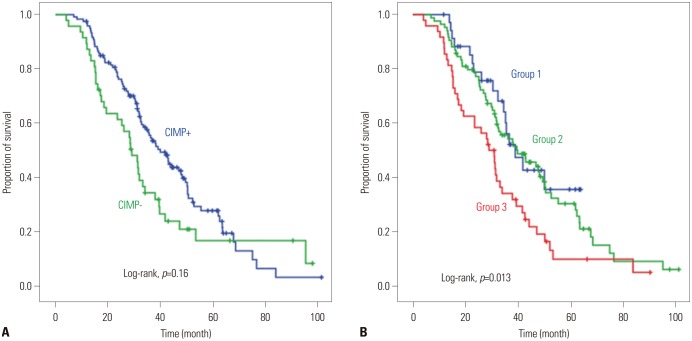
The methylation frequencies of the 10 genes analyzed (p16, p14, MINT1, MINT2, MINT31, hMLH1, DKK3, WNT5A, AXIN2, and TFAP2E) were 47.9%, 10.8%, 21.1%, 16.0%, 20.6%, 0.5%, 53.1%, 32.0%, 2.6%, and 2.1%, respectively. We divided patients into three groups based on the number of methylated genes (group 1, no methylation n=38; group 2, 1-2 methylations n=92; group 3, 3 or more methylations n=64). Among patients treated with palliative chemotherapy (n=167), median OSs of groups 1, 2, and 3 were 39.1, 39.7, and 29.1 months, respectively (log rank p=0.013). After adjustment, number of methylations was identified as an independent poor prognostic factor (0-2 methylated vs. ≥3 methylated: hazard ratio, 1.72; 95% confidence interval, 1.16-2.56, p=0.007).
CONCLUSION
This study suggests that methylation of Wnt pathway genes, in addition to known CpG island methylator phenotype markers, may help predict treatment outcome and survival in patients with CRC.
MeSH Terms
Figure
Cited by 1 articles
-
Regulation Mechanism of Long Noncoding RNAs in Colon Cancer Development and Progression
Xiaohuan Tang, Xiaofang Qiao, Chao Chen, Yuanda Liu, Jiaming Zhu, Jingjing Liu
Yonsei Med J. 2019;60(4):319-325. doi: 10.3349/ymj.2019.60.4.319.
Reference
-
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87–108. PMID: 25651787.
Article2. Jung KW, Won YJ, Oh CM, Kong HJ, Lee DH, Lee KH. Prediction of cancer incidence and mortality in Korea, 2017. Cancer Res Treat. 2017; 49:306–312. PMID: 28301926.
Article3. Cremolini C, Schirripa M, Antoniotti C, Moretto R, Salvatore L, Masi G, et al. First-line chemotherapy for mCRC—a review and evidence-based algorithm. Nat Rev Clin Oncol. 2015; 12:607–619. PMID: 26215044.
Article4. Toyota M, Ohe-Toyota M, Ahuja N, Issa JP. Distinct genetic profiles in colorectal tumors with or without the CpG island methylator phenotype. Proc Natl Acad Sci U S A. 2000; 97:710–715. PMID: 10639144.
Article5. Markowitz SD, Bertagnolli MM. Molecular origins of cancer: Molecular basis of colorectal cancer. N Engl J Med. 2009; 361:2449–2460. PMID: 20018966.6. Sambuudash O, Kim HS, Cho MY. Lack of aberrant methylation in an adjacent area of left-sided colorectal cancer. Yonsei Med J. 2017; 58:749–755. PMID: 28540987.
Article7. Juo YY, Johnston FM, Zhang DY, Juo HH, Wang H, Pappou EP, et al. Prognostic value of CpG island methylator phenotype among colorectal cancer patients: a systematic review and meta-analysis. Ann Oncol. 2014; 25:2314–2327. PMID: 24718889.
Article8. Ahn JB, Chung WB, Maeda O, Shin SJ, Kim HS, Chung HC, et al. DNA methylation predicts recurrence from resected stage III proximal colon cancer. Cancer. 2011; 117:1847–1854. PMID: 21509761.
Article9. Aguilera O, Fraga MF, Ballestar E, Paz MF, Herranz M, Espada J, et al. Epigenetic inactivation of the Wnt antagonist DICKKOPF-1 (DKK-1) gene in human colorectal cancer. Oncogene. 2006; 25:4116–4121. PMID: 16491118.
Article10. Koinuma K, Yamashita Y, Liu W, Hatanaka H, Kurashina K, Wada T, et al. Epigenetic silencing of AXIN2 in colorectal carcinoma with microsatellite instability. Oncogene. 2006; 25:139–146. PMID: 16247484.
Article11. de Sousa E Melo F, Colak S, Buikhuisen J, Koster J, Cameron K, de Jong JH, et al. Methylation of cancer-stem-cell-associated Wnt target genes predicts poor prognosis in colorectal cancer patients. Cell Stem Cell. 2011; 9:476–485. PMID: 22056143.
Article12. Ebert MP, Tänzer M, Balluff B, Burgermeister E, Kretzschmar AK, Hughes DJ, et al. TFAP2E-DKK4 and chemoresistance in colorectal cancer. N Engl J Med. 2012; 366:44–53. PMID: 22216841.13. Dejmek J, Dejmek A, Säfholm A, Sjölander A, Andersson T. Wnt-5a protein expression in primary dukes B colon cancers identifies a subgroup of patients with good prognosis. Cancer Res. 2005; 65:9142–9146. PMID: 16230369.
Article14. Kim SH, Park KH, Shin SJ, Lee KY, Kim TI, Kim NK, et al. p16 Hypermethylation and KRAS mutation are independent predictors of cetuximab plus FOLFIRI chemotherapy in patients with metastatic colorectal cancer. Cancer Res Treat. 2016; 48:208–215. PMID: 25943321.15. Yu J, Tao Q, Cheng YY, Lee KY, Ng SS, Cheung KF, et al. Promoter methylation of the Wnt/beta-catenin signaling antagonist Dkk-3 is associated with poor survival in gastric cancer. Cancer. 2009; 115:49–60. PMID: 19051296.16. Ying J, Li H, Yu J, Ng KM, Poon FF, Wong SC, et al. WNT5A exhibits tumor-suppressive activity through antagonizing the Wnt/beta-catenin signaling, and is frequently methylated in colorectal cancer. Clin Cancer Res. 2008; 14:55–61. PMID: 18172252.17. Rawson JB, Mrkonjic M, Daftary D, Dicks E, Buchanan DD, Younghusband HB, et al. Promoter methylation of Wnt5a is associated with microsatellite instability and BRAF V600E mutation in two large populations of colorectal cancer patients. Br J Cancer. 2011; 104:1906–1912. PMID: 21587258.
Article18. Yoon HH, Shi Q, Alberts SR, Goldberg RM, Thibodeau SN, Sargent DJ, et al. Racial differences in BRAF/KRAS mutation rates and survival in stage III colon cancer patients. J Natl Cancer Inst. 2015; 107:djv186. PMID: 26160882.19. Shaw RJ, Akufo-Tetteh EK, Risk JM, Field JK, Liloglou T. Methylation enrichment pyrosequencing: combining the specificity of MSP with validation by pyrosequencing. Nucleic Acids Res. 2006; 34:e78. PMID: 16807314.
Article20. Guinney J, Dienstmann R, Wang X, de Reyniès A, Schlicker A, Soneson C, et al. The consensus molecular subtypes of colorectal cancer. Nat Med. 2015; 21:1350–1356. PMID: 26457759.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Prognostic Significance of CpG Island Methylator Phenotype in Colorectal Cancer
- Multiple Methylator Phenotypes Involving Tumor Suppressor Genes in Ovarian Cancers
- Serrated neoplasia pathway as an alternative route of colorectal cancer carcinogenesis
- CpG Island Methylator Phenotype-High Colorectal Cancers and Their Prognostic Implications and Relationships with the Serrated Neoplasia Pathway
- Cancer Epigenetics