J Vet Sci.
2016 Dec;17(4):445-451. 10.4142/jvs.2016.17.4.445.
Claudin-1, -2, -4, and -5: comparison of expression levels and distribution in equine tissues
- Affiliations
-
- 1Laboratory of Veterinary Biochemistry and Molecular Biology, College of Veterinary Medicine, Chungbuk National University, Cheongju 28644, Korea. ebjeung@chungbuk.ac.kr
- KMID: 2412599
- DOI: http://doi.org/10.4142/jvs.2016.17.4.445
Abstract
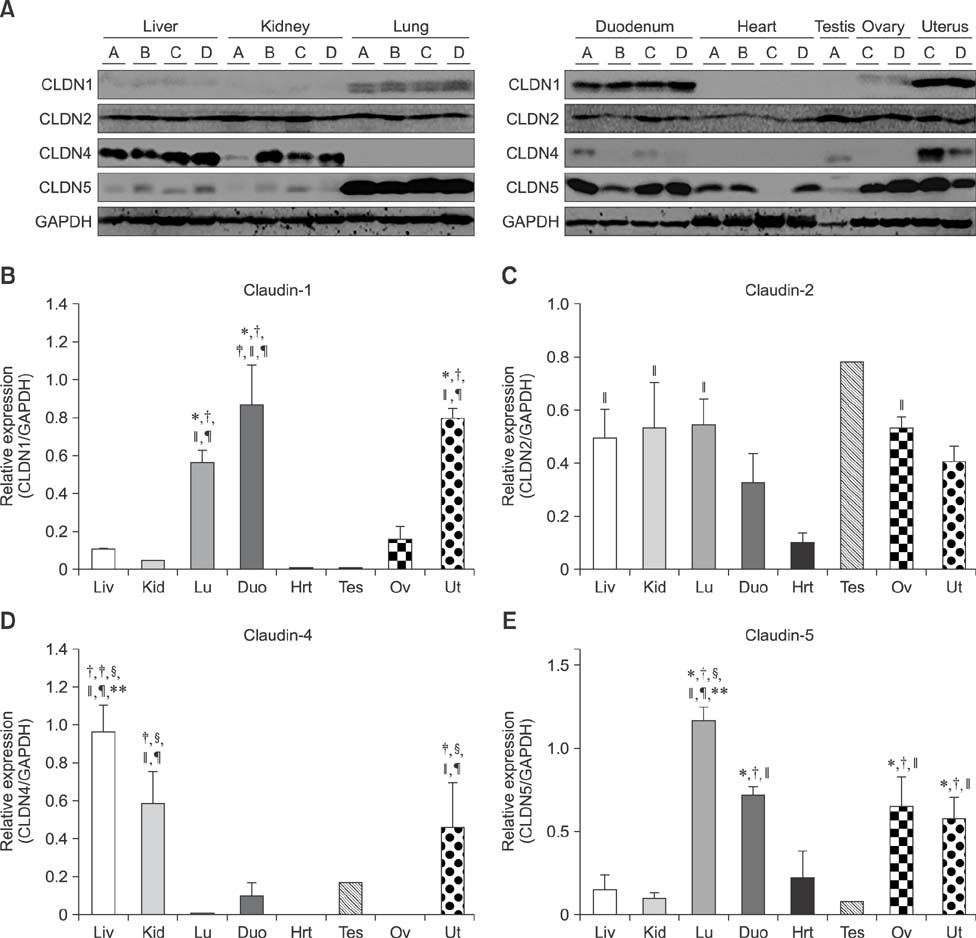
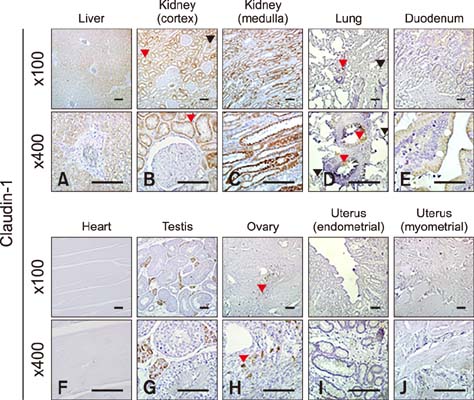
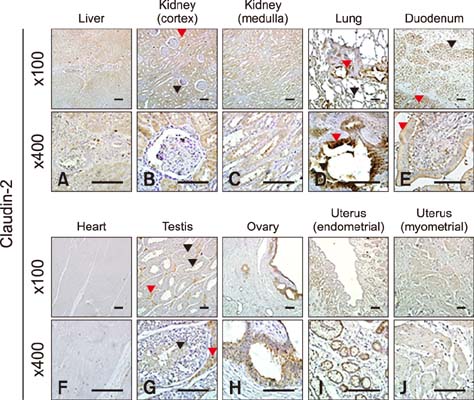
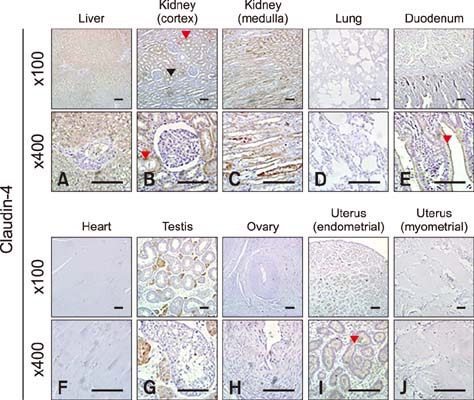
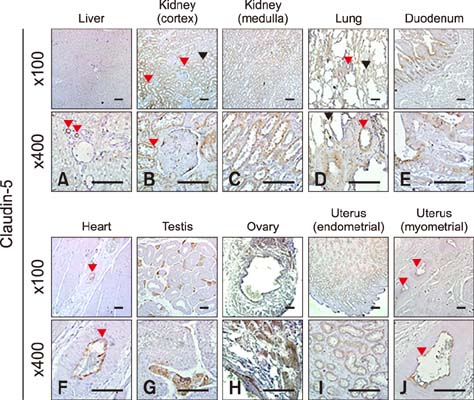
- Claudins, which are known as transmembrane proteins play an essential role in tight junctions (TJs) to form physical barriers and regulate paracellular transportation. To understand equine diseases, it is helpful to measure the tissue-specific expression of TJs in horses. Major equine diseases such as colic and West Nile cause damage to TJs. In this study, the expression level and distribution of claudin-1, -2, -4, and -5 in eight tissues were assessed by Western blotting and immunohistochemistry methods. Claudin-1 was primarily identified in the lung, duodenum, and uterus, claudin-2 was evenly observed in equine tissues, claudin-4 was abundantly detected in the liver, kidney and uterus, and claudin-5 was strongly expressed in the lung, duodenum, ovary, and uterus, as determined by Western blotting method. The localization of equine claudins was observed by immunohistochemistry methods. These findings provide knowledge regarding the expression patterns and localization of equine claudins, as well as valuable information to understand tight junction-related diseases according to tissue specificity and function of claudins in horses.
MeSH Terms
Figure
Reference
-
1. Amasheh S, Meiri N, Gitter AH, Schöneberg T, Mankertz J, Schulzke JD, Fromm M. Claudin-2 expression induces cation-selective channels in tight junctions of epithelial cells. J Cell Sci. 2002; 115:4969–4976.
Article2. Boyer JL. Bile formation and secretion. Compr Physiol. 2013; 3:1035–1078.
Article3. D'Souza T, Sherman-Baust CA, Poosala S, Mullin JM, Morin PJ. Age-related changes of claudin expression in mouse liver, kidney, and pancreas. J Gerontol A Biol Sci Med Sci. 2009; 64:1146–1153.4. Delfín DA, Xu Y, Schill KE, Mays TA, Canan BD, Zang KE, Barnum JA, Janssen PM, Rafael-Fortney JA. Sustaining cardiac claudin-5 levels prevents functional hallmarks of cardiomyopathy in a muscular dystrophy mouse model. Mol Ther. 2012; 20:1378–1383.
Article5. Furuse M, Fujita K, Hiiragi T, Fujimoto K, Tsukita S. Claudin-1 and -2: novel integral membrane proteins localizing at tight junctions with no sequence similarity to occludin. J Cell Biol. 1998; 141:1539–1550.
Article6. Furuse M, Furuse K, Sasaki H, Tsukita S. Conversion of zonulae occludentes from tight to leaky strand type by introducing claudin-2 into Madin-Darby canine kidney I cells. J Cell Biol. 2001; 153:263–272.
Article7. Furuse M, Hata M, Furuse K, Yoshida Y, Haratake A, Sugitani Y, Noda T, Kubo A, Tsukita S. Claudin-based tight junctions are crucial for the mammalian epidermal barrier: a lesson from claudin-1-deficient mice. J Cell Biol. 2002; 156:1099–1111.
Article8. Furuse M, Hirase T, Itoh M, Nagafuchi A, Yonemura S, Tsukita S, Tsukita S. Occludin: a novel integral membrane protein localizing at tight junctions. J Cell Biol. 1993; 123:1777–1788.
Article9. Furuse M, Itoh M, Hirase T, Nagafuchi A, Yonemura S, Tsukita S, Tsukita S. Direct association of occludin with ZO-1 and its possible involvement in the localization of occludin at tight junctions. J Cell Biol. 1994; 127:1617–1626.
Article10. Furuse M, Sasaki H, Tsukita S. Manner of interaction of heterogeneous claudin species within and between tight junction strands. J Cell Biol. 1999; 147:891–903.
Article11. Gobe GC, Johnson DW. Distal tubular epithelial cells of the kidney: potential support for proximal tubular cell survival after renal injury. Int J Biochem Cell Biol. 2007; 39:1551–1561.
Article12. Grosche A, Morton AJ, Graham AS, Sanchez LC, Blikslager AT, Polyak MM, Freeman DE. Ultrastructural changes in the equine colonic mucosa after ischaemia and reperfusion. Equine Vet J Suppl. 2011; 8–15.
Article13. Grossman ML. The glands of Brunner. Physiol Rev. 1958; 38:675–690.
Article14. Günzel D, Yu AS. Claudins and the modulation of tight junction permeability. Physiol Rev. 2013; 93:525–569.
Article15. Hou J, Renigunta A, Yang J, Waldegger S. Claudin-4 forms paracellular chloride channel in the kidney and requires claudin-8 for tight junction localization. Proc Natl Acad Sci U S A. 2010; 107:18010–18015.
Article16. Hwang I, Yang H, Kang HS, Ahn CH, Lee GS, Hong EJ, An BS, Jeung EB. Spatial expression of claudin family members in various organs of mice. Mol Med Rep. 2014; 9:1806–1812.
Article17. Kojima T, Yamamoto T, Murata M, Chiba H, Kokai Y, Sawada N. Regulation of the blood-biliary barrier: interaction between gap and tight junctions in hepatocytes. Med Electron Microsc. 2003; 36:157–164.
Article18. Krug SM, Schulzke JD, Fromm M. Tight junction, selective permeability, and related diseases. Semin Cell Dev Biol. 2014; 36:166–176.
Article19. Medigeshi GR, Hirsch AJ, Brien JD, Uhrlaub JL, Mason PW, Wiley C, Nikolich-Zugich J, Nelson JA. West Nile virus capsid degradation of claudin proteins disrupts epithelial barrier function. J Virol. 2009; 83:6125–6134.
Article20. Mitic LL, Van Itallie CM, Anderson JM. Molecular physiology and pathophysiology of tight junctions I. Tight junction structure and function: lessons from mutant animals and proteins. Am J Physiol Gastrointest Liver Physiol. 2000; 279:G250–G254.
Article21. Morita K, Sasaki H, Furuse M, Tsukita S. Endothelial claudin: claudin-5/TMVCF constitutes tight junction strands in endothelial cells. J Cell Biol. 1999; 147:185–194.22. Muto S, Hata M, Taniguchi J, Tsuruoka S, Moriwaki K, Saitou M, Furuse K, Sasaki H, Fujimura A, Imai M, Kusano E, Tsukita S, Furuse M. Claudin-2-deficient mice are defective in the leaky and cation-selective paracellular permeability properties of renal proximal tubules. Proc Natl Acad Sci U S A. 2010; 107:8011–8016.
Article23. Nitta T, Hata M, Gotoh S, Seo Y, Sasaki H, Hashimoto N, Furuse M, Tsukita S. Size-selective loosening of the blood-brain barrier in claudin-5-deficient mice. J Cell Biol. 2003; 161:653–660.
Article24. Rangel LB, Agarwal R, D'Souza T, Pizer ES, Alò PL, Lancaster WD, Gregoire L, Schwartz DR, Cho KR, Morin PJ. Tight junction proteins claudin-3 and claudin-4 are frequently overexpressed in ovarian cancer but not in ovarian cystadenomas. Clin Cancer Res. 2003; 9:2567–2575.25. Rokkam D, LaFemina MJ, Lee JW, Matthay MA, Frank JA. Claudin-4 levels are associated with intact alveolar fluid clearance in human lungs. Am J Pathol. 2011; 179:1081–1087.
Article26. Tsukita S, Furuse M, Itoh M. Multifunctional strands in tight junctions. Nat Rev Mol Cell Biol. 2001; 2:285–293.
Article27. Van Itallie C, Rahner C, Anderson JM. Regulated expression of claudin-4 decreases paracellular conductance through a selective decrease in sodium permeability. J Clin Invest. 2001; 107:1319–1327.
Article28. Van Itallie CM, Anderson JM. Claudins and epithelial paracellular transport. Annu Rev Physiol. 2006; 68:403–429.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of Claudin-1 and -7 in Clear Cell Renal Cell Carcinoma and Its Clinical Significance
- Increased expressions of claudin-1 and claudin-7 in cervical squamous intraepithelial neoplasias and invasive squamous cell carcinomas
- Expressions of Tight Junction Protein Claudin-1 and Claudin-7 in Squamous Cell Carcinoma of the Uterine Cervix
- Correlation of Decreased Expressions of Claudin 4 and E-cadherin Proteins and the Clinicopathologic Factors of Stomach Cancer
- Correlation between Claudins Expression and Prognostic Factors in Prostate Cancer