J Vet Sci.
2018 May;19(3):358-367. 10.4142/jvs.2018.19.3.358.
Phenotypic and genotypic analyses of an attenuated porcine reproductive and respiratory syndrome virus strain after serial passages in cultured porcine alveolar macrophages
- Affiliations
-
- 1Choongang Vaccine Laboratory, Daejeon 34055, Korea.
- 2Animal Virology Laboratory, School of Life Sciences, BK21 Plus KNU Creative BioResearch Group, Kyungpook National University, Daegu 41566, Korea. changhee@knu.ac.kr
- 3School of Applied Biosciences, College of Agriculture and Life Sciences, Kyungpook National University, Daegu 41566, Korea.
- 4College of Veterinary Medicine, Chungbuk National University, Cheongju 28644, Korea. sykang@cbu.ac.kr
- KMID: 2412126
- DOI: http://doi.org/10.4142/jvs.2018.19.3.358
Abstract
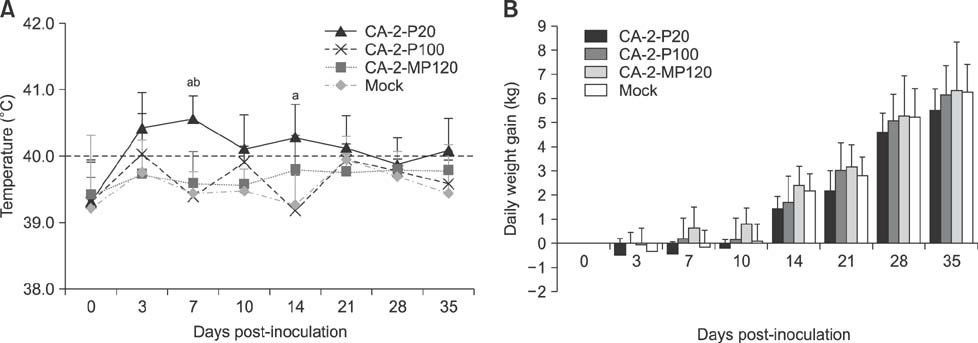
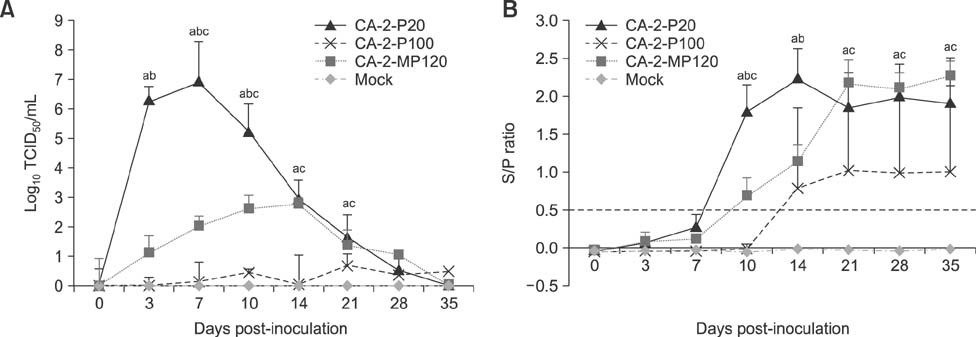
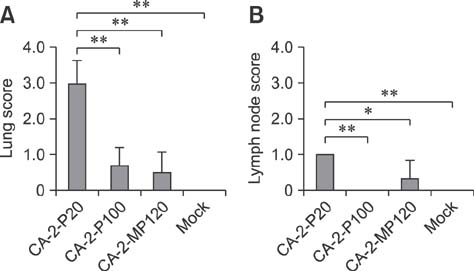
- The porcine reproductive and respiratory syndrome virus (PRRSV) is a globally ubiquitous swine viral pathogen that causes major economic losses worldwide. We previously reported an over-attenuated phenotype of cell-adapted PRRSV strain CA-2-P100 in vivo. In the present study, CA-2-P100 was serially propagated in cultured porcine alveolar macrophage (PAM) cells for up to 20 passages to obtain the derivative strain CA-2-MP120. Animal inoculation studies revealed that both CA-2-P100 and CA-2-MP120 had decreased virulence, eliciting weight gains, body temperatures, and histopathologic lesions similar to those in the negative control group. However, compared to CA-2-P100 infection, CA-2-MP120 yielded consistently higher viremia kinetics and enhanced antibody responses in pigs. All pigs inoculated with CA-2-MP120 developed viremia and seroconverted to PRRSV. During 20 passages in PAM cells, CA-2-MP120 acquired 15 amino acid changes that were mostly distributed in nsp2 and minor structural protein-coding regions. Among these changes, 6 mutations represented reversions to the sequences of the reference CA-2 and parental CA-2-P20 strains. These genetic drifts may be hypothetical molecular markers associated with PRRSV macrophage tropism and virulence. Our results indicate that the PAM-passaged CA-2-MP120 strain is a potential candidate for developing a live, attenuated PRRSV vaccine.
Keyword
MeSH Terms
Figure
Reference
-
1. Adams MJ, Lefkowitz EJ, King AMQ, Harrach B, Harrison RL, Knowles NJ, Kropinski AM, Krupovic M, Kuhn JH, Mushegian AR, Nibert M, Sabanadzovic S, Sanfaçon H, Siddell SG, Simmonds P, Varsani A, Zerbini FM, Gorbalenya AE, Davison AJ. Changes to taxonomy and the International Code of Virus Classification and Nomenclature ratified by the International Committee on Taxonomy of Viruses (2017). Arch Virol. 2017; 162:2505–2538.
Article2. Allende R, Kutish GF, Laegreid W, Lu Z, Lewis TL, Rock DL, Friesen J, Galeota JA, Doster AR, Osorio FA. Mutations in the genome of porcine reproductive and respiratory syndrome virus responsible for the attenuation phenotype. Arch Virol. 2000; 145:1149–1161.
Article3. Beura LK, Sarkar SN, Kwon B, Subramaniam S, Jones C, Pattnaik AK, Osorio FA. Porcine reproductive and respiratory syndrome virus nonstructural protein 1β modulates host innate immune response by antagonizing IRF3 activation. J Virol. 2010; 84:1574–1584.
Article4. Brockmeier SL, Loving CL, Vorwald AC, Kehrli ME Jr, Baker RB, Nicholson TL, Lager KM, Miller LC, Faaberg KS. Genomic sequence and virulence comparison of four type 2 porcine reproductive and respiratory syndrome virus strains. Virus Res. 2012; 169:212–221.
Article5. Chang CC, Yoon KJ, Zimmerman JJ, Harmon KM, Dixon PM, Dvorak CM, Murtaugh MP. Evolution of porcine reproductive and respiratory syndrome virus during sequential passages in pigs. J Virol. 2002; 76:4750–4763.
Article6. Choi HW, Nam E, Lee YJ, Noh YH, Lee SC, Yoon IJ, Kim HS, Kang SY, Choi YK, Lee C. Genomic analysis and pathogenic characteristics of type 2 porcine reproductive and respiratory syndrome virus nsp2 deletion strains isolated in Korea. Vet Microbiol. 2014; 170:232–245.
Article7. Das PB, Dinh PX, Ansari IH, de Lima M, Osorio FA, Pattnaik AK. The minor envelope glycoproteins GP2a and GP4 of porcine reproductive and respiratory syndrome virus interact with the receptor CD163. J Virol. 2010; 84:1731–1740.
Article8. Faaberg KS, Han J, Wang Y. Molecular dissection of porcine reproductive and respiratory virus putative nonstructural protein 2. Adv Exp Med Biol. 2006; 581:73–77.
Article9. Fang Y, Kim DY, Ropp S, Steen P, Christopher-Hennings J, Nelson EA, Rowland RR. Heterogeneity in Nsp2 of European-like porcine reproductive and respiratory syndrome viruses isolated in the United States. Virus Res. 2004; 100:229–235.
Article10. Halbur PG, Paul PS, Frey ML, Landgraf J, Eernisse K, Meng XJ, Lum MA, Andrews JJ, Rathje JA. Comparison of the pathogenicity of two US porcine reproductive and respiratory syndrome virus isolates with that of the Lelystad virus. Vet Pathol. 1995; 32:648–660.
Article11. Han J, Liu G, Wang Y, Faaberg KS. Identification of nonessential regions of the nsp2 replicase protein of porcine reproductive and respiratory syndrome virus strain VR-2332 for replication in cell culture. J Virol. 2007; 81:9878–9890.
Article12. Han J, Wang Y, Faaberg KS. Complete genome analysis of RFLP 184 isolates of porcine reproductive and respiratory syndrome virus. Virus Res. 2006; 122:175–182.
Article13. Holtkamp DJ, Kliebenstein JB, Neumann EJ, Zimmerman JJ, Rotto HF, Yoder TK, Wang C, Yeske PE, Mowrer CL, Haley CA. Assessment of the economic impact of porcine reproductive and respiratory syndrome virus on United States pork producers. J Swine Health Prod. 2013; 21:72–84.14. Kappes MA, Faaberg KS. PRRSV structure, replication and recombination: origin of phenotype and genotype diversity. Virology. 2015; 479-480:475–486.
Article15. Keffaber KK. Reproductive failure of unknown etiology. Am Assoc Swine Pract Newsl. 1989; 1:1–9.16. Kim HK, Lee CS, Kang BK, Yeom MJ, Moon HJ, Park SJ, Nguyen VG, Song DS, Park BK. Experimental infection of a newly emerging Korean type I porcine reproductive and respiratory syndrome virus isolate in colostrum-deprived pigs. Virol J. 2011; 8:177.
Article17. Kim JY, Lee SY, Sur JH, Lyoo YS. Serological and genetic characterization of the European strain of the porcine reproductive and respiratory syndrome virus isolated in Korea. Korean J Vet Res. 2006; 46:363–370.18. Kim SH, Roh IS, Choi EJ, Lee C, Lee CH, Lee KH, Lee KK, Song YK, Lee OS, Park CK. A molecular analysis of European porcine reproductive and respiratory syndrome virus isolated in South Korea. Vet Microbiol. 2010; 143:394–400.
Article19. Kweon CH, Kwon BJ, Lee HJ, Cho JJ, Hwang EK, Shin JH, Yoon YD, Kang YB, An SH, Kim YH, Huh W, Jun MH, Wensvoort G. Isolation of porcine reproductive and respiratory syndrome virus (PRRSV) in Korea. Korean J Vet Res. 1994; 34:77–83.20. Kwon B, Ansari IH, Pattnaik AK, Osorio FA. Identification of virulence determinants of porcine reproductive and respiratory syndrome virus through construction of chimeric clones. Virology. 2008; 380:371–378.
Article21. Lee S, Lee C. Outbreak-related porcine epidemic diarrhea virus strains similar to US strains, South Korea, 2013. Emerg Infect Dis. 2014; 20:1223–1226.
Article22. Lee SC, Choi HW, Nam E, Noh YH, Lee S, Lee YJ, Park GS, Shin JH, Yoon IJ, Kang SY, Lee C. Pathogenicity and genetic characteristics associated with cell adaptation of a virulent porcine reproductive and respiratory syndrome virus nsp2 DEL strain CA-2. Vet Microbiol. 2016; 186:174–188.
Article23. Lee YJ, Park CK, Nam E, Kim SH, Lee OS, Lee DS, Lee C. Generation of a porcine alveolar macrophage cell line for the growth of porcine reproductive and respiratory syndrome virus. J Virol Methods. 2010; 163:410–415.
Article24. Martínez-Lobo FJ, Díez-Fuertes F, Segalés J, García-Artiga C, Simarro I, Castro JM, Prieto C. Comparative pathogenicity of type 1 and type 2 isolates of porcine reproductive and respiratory syndrome virus (PRRSV) in a young pig infection model. Vet Microbiol. 2011; 154:58–68.
Article25. Nam E, Park CK, Kim SH, Joo YS, Yeo SG, Lee C. Complete genomic characterization of a European type 1 porcine reproductive and respiratory syndrome virus isolate in Korea. Arch Virol. 2009; 154:629–638.
Article26. Nelsen CJ, Murtaugh MP, Faaberg KS. Porcine reproductive and respiratory syndrome virus comparison: divergent evolution on two continents. J Virol. 1999; 73:270–280.
Article27. Sagong M, Park CK, Kim SH, Lee KK, Lee OS, Lee DS, Cha SY, Lee C. Human telomerase reverse transcriptase-immortalized porcine monomyeloid cell lines for the production of porcine reproductive and respiratory syndrome virus. J Virol Methods. 2012; 179:26–32.
Article28. Shi M, Lam TT, Hon CC, Murtaugh MP, Davies PR, Hui RK, Li J, Wong LT, Yip CW, Jiang JW, Leung FC. Phylogeny-based evolutionary demographical and geographical dissection of North American type 2 porcine reproductive and respiratory syndrome viruses. J Virol. 2010; 84:8700–8711.
Article29. Snijder EJ, Kikkert M, Fang Y. Arterivirus molecular biology and pathogenesis. J Gen Virol. 2013; 94:2141–2163.
Article30. Sun Y, Han M, Kim C, Calvert JG, Yoo D. Interplay between interferon-mediated innate immunity and porcine reproductive and respiratory syndrome virus. Viruses. 2012; 4:424–446.
Article31. Wang X, Sun L, Li Y, Lin T, Gao F, Yao H, He K, Tong G, Wei Z, Yuan S. Development of a differentiable virus via a spontaneous deletion in the nsp2 region associated with cell adaptation of porcine reproductive and respiratory syndrome virus. Virus Res. 2013; 171:150–160.
Article32. Wissink EH, Kroese MV, van Wijk HA, Rijsewijk FA, Meulenberg JJ, Rottier PJ. Envelope protein requirements for the assembly of infectious virions of porcine reproductive and respiratory syndrome virus. J Virol. 2005; 79:12495–12506.
Article33. Yuan S, Mickelson D, Murtaugh MP, Faaberg KS. Complete genome comparison of porcine reproductive and respiratory syndrome virus parental and attenuated strains. Virus Res. 2001; 79:189–200.
Article34. Zimmerman JJ, Benfield DA, Dee SA, Murtaugh MP, Stadejek T, Stevenson GW, Torremorell M. Porcine reproductive and respiratory syndrome virus (porcine arterivirus). In : Zimmerman JJ, Karriker LA, Ramirez A, Schwartz KJ, Stevenson GW, editors. Diseases of Swine. 10th ed. Ames: Wiley-Blackwell;2012. p. 461–486.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pathologic studies in lymph nodes of pigs infected with porcine circovirus type 2, porcine reproductive and respiratory syndrome virus
- Evaluation of the efficacy of an attenuated live vaccine based on virulent porcine reproductive and respiratory syndrome virus 2 in young pigs
- Intracellular Localization of the Porcine Reproductive and Respiratory Syndrome Virus Nucleocapsid Protein
- Porcine ear necrosis syndrome by coinfection of porcine reproductive and respiratory syndrome virus and Staphylococcus hyicus
- Prevalence of porcine reproductive and respiratory syndrome virus, porcine circovirus type 2 and porcine parvovirus from aborted fetuses and pigs with respiratory problems in Korea