Endocrinol Metab.
2015 Sep;30(3):272-279. 10.3803/EnM.2015.30.3.272.
Efficacy of a Once-Monthly Pill Containing Ibandronate and Cholecalciferol on the Levels of 25-Hydroxyvitamin D and Bone Markers in Postmenopausal Women with Osteoporosis
- Affiliations
-
- 1Department of Endocrinology and Metabolism, Kyung Hee University School of Medicine, Seoul, Korea.
- 2Dreampharma Corp., Seoul, Korea.
- 3Department of Endocrinology and Metabolism, Keimyung University School of Medicine, Daegu, Korea.
- 4Department of Endocrinology and Metabolism, Korea University Anam Hospital, Korea University College of Medicine, Seoul, Korea.
- 5Division of Endocrinology and Metabolism, Department of Internal Medicine, Dongguk University Ilsan Hospital, Dongguk University College of Medicine, Goyang, Korea.
- 6Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea.
- 7Division of Endocrinology and Metabolism, Department of Internal Medicine, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 8Divison of Endocrinology and Metabolism, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 9Department of Internal Medicine, Endocrine Research Institute, Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
- 10Department of Internal Medicine, Soonchunhyang University College of Medicine, Seoul, Korea.
- 11Department of Endocrinology and Metabolism, Ajou University School of Medicine, Suwon, Korea.
- 12Department of Internal Medicine, Inje University College of Medicine, Busan, Korea.
- 13Department of Internal Medicine, Chonnam National University Medical School, Gwangju, Korea.
- 14Department of Internal Medicine, Chungnam National University Hospital, Chungnam National University School of Medicine, Daejeon, Korea.
- 15Division of Endocrinology and Metabolism, Department of Internal Medicine, Hallym University Dongtan Sacred Heart Hospital, Hallym University College of Medicine, Hwaseong, Korea.
- 16Department of Endocrinology and Metabolism, Hanyang University Guri Hospital, Hanyang University College of Medicine, Guri, Korea.
- 17Department of Endocrinology and Metabolism, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea. mikang@catholic.ac.kr
- KMID: 2407077
- DOI: http://doi.org/10.3803/EnM.2015.30.3.272
Abstract
- BACKGROUND
The present study evaluated the efficacy of a combination of ibandronate and cholecalciferol on the restoration of the levels of 25-hydroxyvitamin D (25[OH]D) and various bone markers in postmenopausal women with osteoporosis.
METHODS
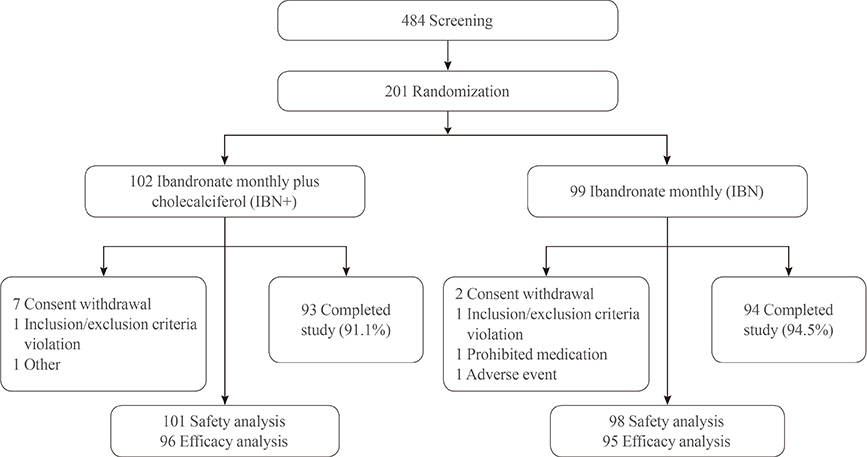
This was a randomized, double-blind, active-controlled, prospective 16-week clinical trial conducted in 20 different hospitals. A total of 201 postmenopausal women with osteoporosis were assigned randomly to one of two groups: the IBN group, which received a once-monthly pill containing 150 mg ibandronate (n=99), or the IBN+ group, which received a once-monthly pill containing 150 mg ibandronate and 24,000 IU cholecalciferol (n=102). Serum levels of 25(OH)D, parathyroid hormone (PTH), and various bone markers were assessed at baseline and at the end of a 16-week treatment period.
RESULTS
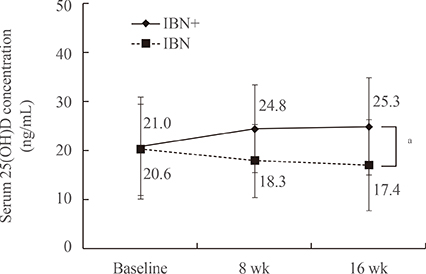
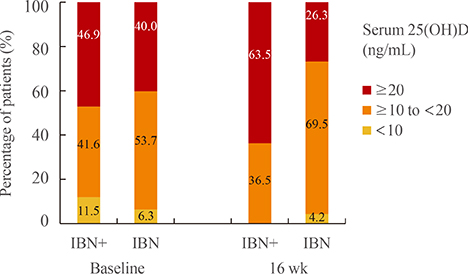
After 16 weeks of treatment, the mean serum levels of 25(OH)D significantly increased from 21.0 to 25.3 ng/mL in the IBN+ group but significantly decreased from 20.6 to 17.4 ng/mL in the IBN group. Additionally, both groups exhibited significant increases in mean serum levels of PTH but significant decreases in serum levels of bone-specific alkaline phosphatase and C-telopeptide of type 1 collagen (CTX) at 16 weeks; no significant differences were observed between the groups. However, in subjects with a vitamin D deficiency, IBN+ treatment resulted in a significant decrease in serum CTX levels compared with IBN treatment.
CONCLUSION
The present findings demonstrate that a once-monthly pill containing ibandronate and cholecalciferol may be useful for the amelioration of vitamin D deficiency in patients with postmenopausal osteoporosis. Moreover, this treatment combination effectively decreased serum levels of resorption markers, especially in subjects with a vitamin D deficiency, over the 16-week treatment period.
MeSH Terms
Figure
Cited by 1 articles
-
Pharmacologic treatment of osteoporosis
Yong-Ki Min
J Korean Med Assoc. 2016;59(11):847-856. doi: 10.5124/jkma.2016.59.11.847.
Reference
-
1. Cramer JA, Gold DT, Silverman SL, Lewiecki EM. A systematic review of persistence and compliance with bisphosphonates for osteoporosis. Osteoporos Int. 2007; 18:1023–1031.2. Srivastava M, Deal C. Osteoporosis in elderly: prevention and treatment. Clin Geriatr Med. 2002; 18:529–555.3. Thacher TD, Clarke BL. Vitamin D insufficiency. Mayo Clin Proc. 2011; 86:50–60.4. Lips P, Hosking D, Lippuner K, Norquist JM, Wehren L, Maalouf G, et al. The prevalence of vitamin D inadequacy amongst women with osteoporosis: an international epidemiological investigation. J Intern Med. 2006; 260:245–254.5. Rossini M, Bianchi G, Di Munno O, Giannini S, Minisola S, Sinigaglia L, et al. Determinants of adherence to osteoporosis treatment in clinical practice. Osteoporos Int. 2006; 17:914–921.6. Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011; 96:1911–1930.7. Ross AC, Taylor CL, Yaktine AL, Del Valle HB. Dietary reference intakes: calcium, vitamin D. Washington DC: National Academies Press;2011.8. Choi HS. Vitamin d status in Korea. Endocrinol Metab (Seoul). 2013; 28:12–16.9. Chung HY, Chin SO, Kang MI, Koh JM, Moon SH, Yoon BK, et al. Efficacy of risedronate with cholecalciferol on 25-hydroxyvitamin D level and bone turnover in Korean patients with osteoporosis. Clin Endocrinol (Oxf). 2011; 74:699–704.10. National Osteoporosis Foundation. Clinician's guide to prevention and treatment of osteoporosis. Washington DC: National Osteoporosis Foundation;2014.11. Korean Society for Bone and Mineral Research. Physician's guide for diagnosis and treatment of osteoporosis. Seoul: Korean Society for Bone and Mineral Research;2013.12. Chel V, Wijnhoven HA, Smit JH, Ooms M, Lips P. Efficacy of different doses and time intervals of oral vitamin D supplementation with or without calcium in elderly nursing home residents. Osteoporos Int. 2008; 19:663–671.13. Malabanan A, Veronikis IE, Holick MF. Redefining vitamin D insufficiency. Lancet. 1998; 351:805–806.14. Vieth R, Chan PC, MacFarlane GD. Efficacy and safety of vitamin D3 intake exceeding the lowest observed adverse effect level. Am J Clin Nutr. 2001; 73:288–294.15. Recker R, Lips P, Felsenberg D, Lippuner K, Benhamou L, Hawkins F, et al. Alendronate with and without cholecalciferol for osteoporosis: results of a 15-week randomized controlled trial. Curr Med Res Opin. 2006; 22:1745–1755.16. Garnero P, Shih WJ, Gineyts E, Karpf DB, Delmas PD. Comparison of new biochemical markers of bone turnover in late postmenopausal osteoporotic women in response to alendronate treatment. J Clin Endocrinol Metab. 1994; 79:1693–1700.17. Carmel AS, Shieh A, Bang H, Bockman RS. The 25(OH)D level needed to maintain a favorable bisphosphonate response is ≥33 ng/ml. Osteoporos Int. 2012; 23:2479–2487.18. Maxwell JD. Seasonal variation in vitamin D. Proc Nutr Soc. 1994; 53:533–543.19. Bolland MJ, Grey A, Avenell A, Gamble GD, Reid IR. Calcium supplements with or without vitamin D and risk of cardiovascular events: reanalysis of the Women's Health Initiative limited access dataset and meta-analysis. BMJ. 2011; 342:d2040.20. The American Society for Bone and Mineral Research. New recommendations for taking calcium and vitamin D: U.S. Preventive services task force releases latest on supplements and bone fracture in adults [Internet]. Washington DC: The American Society for Bone and Mineral Research;c2013. updated 2013 Feb 25. cited 2014 Oct 20. Available from: http://www.asbmr.org/about/pressreleases/detail.aspx?cid=e03036f7-5e78-40ae-a3ad-2749b64a8b50.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Efficacy and Safety of Monthly 150 mg Oral Ibandronate in Women with Postmenopausal Osteoporosis: A Systematic Review and Meta-analysis of Randomized Controlled Trials
- Effect of Ibandronate and hPTH on the Viscoelastic Response of Ovariectomized Rat Femur
- Changes of Biochemical Markers of Bone turnover in Pre-, Peri-and Postmenopausal Women
- The Effects of Oral Calcifediol in Postmenopausal Women with Osteopenia and Osteoporosis
- Efficacy, safety, and compliance of ibandronate treatment for 3 years in postmenopausal Japanese women with primary osteoporosis