Clin Exp Vaccine Res.
2018 Jan;7(1):82-86. 10.7774/cevr.2018.7.1.82.
Evaluation of two different enzyme-linked immunosorbent assay for severe fever with thrombocytopenia syndrome virus diagnosis
- Affiliations
-
- 1Department of Microbiology and Medical Research Institute, Chungbuk National University College of Medicine, Cheongju, Korea. choiki55@chungbuk.ac.kr
- 2Zoonotic Infectious Diseases Research Center, Chungbuk National University, Cheongju, Korea.
- 3Department of Internal Medicine, Chungbuk National University Hospital, Cheongju, Korea.
- 4Department of Intermal Medicine, Chungbuk National University College of Medicine, Cheongju, Korea.
- 5Business Development Division, Green Cross WellBeing, Seongnam, Korea.
- KMID: 2402542
- DOI: http://doi.org/10.7774/cevr.2018.7.1.82
Abstract
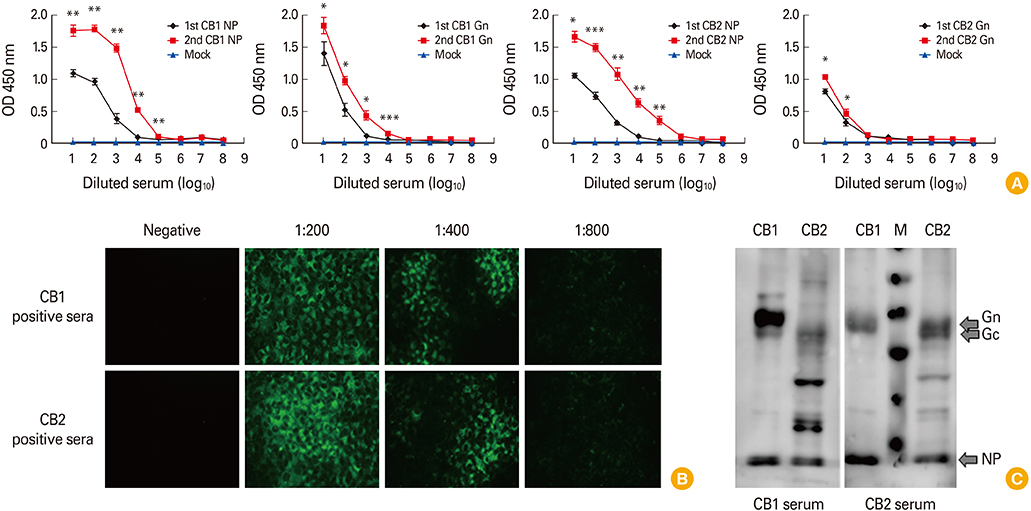
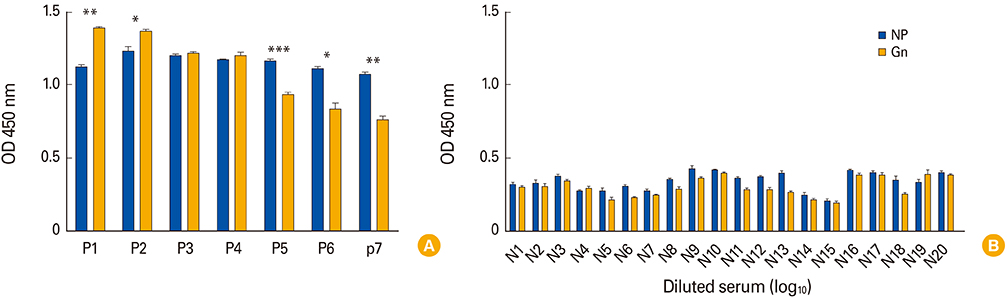
- To develop the large scale serological assay for severe fever with thrombocytopenia syndrome virus (SFTSV) infection, we evaluated two different enzyme-linked immunosorbent assay (ELISA) methods using nucleocapsid protein (NP) and Gn proteins of CB1 (genotype B) SFTSV strains. The NP-based ELISA tests showed more sensitive with broad cross-reactivity between two different genotype A and B strains compared with those of Gn-based ELISA tests. However, Gn-based ELISA showed more genotype specificity and specificity. These result suggested that NP-based ELISA test could be applicable for general sero-prevalence studies of SFTSV infections, while Gn-based ELISA could be applicable for a certain specific genotype sero-prevalence study.
Keyword
MeSH Terms
Figure
Reference
-
1. Jin C, Liang M, Ning J, et al. Pathogenesis of emerging severe fever with thrombocytopenia syndrome virus in C57/BL6 mouse model. Proc Natl Acad Sci U S A. 2012; 109:10053–10058.
Article2. Liu S, Chai C, Wang C, et al. Systematic review of severe fever with thrombocytopenia syndrome: virology, epidemiology, and clinical characteristics. Rev Med Virol. 2014; 24:90–102.
Article3. Yu XJ, Liang MF, Zhang SY, et al. Fever with thrombocytopenia associated with a novel bunyavirus in China. N Engl J Med. 2011; 364:1523–1532.4. Takahashi T, Maeda K, Suzuki T, et al. The first identification and retrospective study of severe fever with thrombocytopenia syndrome in Japan. J Infect Dis. 2014; 209:816–827.5. Kim KH, Yi J, Kim G, et al. Severe fever with thrombocytopenia syndrome, South Korea, 2012. Emerg Infect Dis. 2013; 19:1892–1894.
Article6. Zhang YZ, Xu J. The emergence and cross species transmission of newly discovered tick-borne Bunyavirus in China. Curr Opin Virol. 2016; 16:126–131.
Article7. Niu G, Li J, Liang M, et al. Severe fever with thrombocytopenia syndrome virus among domesticated animals, China. Emerg Infect Dis. 2013; 19:756–763.
Article8. Zhao L, Zhai S, Wen H, et al. Severe fever with thrombocytopenia syndrome virus, Shandong Province, China. Emerg Infect Dis. 2012; 18:963–965.
Article9. Fu Y, Li S, Zhang Z, et al. Phylogeographic analysis of severe fever with thrombocytopenia syndrome virus from Zhoushan Islands, China: implication for transmission across the ocean. Sci Rep. 2016; 6:19563.
Article10. Rudd RJ, Trimarchi CV. Development and evaluation of an in vitro virus isolation procedure as a replacement for the mouse inoculation test in rabies diagnosis. J Clin Microbiol. 1989; 27:2522–2528.
Article11. Xu B, Liu L, Huang X, et al. Metagenomic analysis of fever, thrombocytopenia and leukopenia syndrome (FTLS) in Henan Province, China: discovery of a new bunyavirus. PLoS Pathog. 2011; 7:e1002369.
Article12. Fukuma A, Fukushi S, Yoshikawa T, et al. Severe fever with thrombocytopenia syndrome virus antigen detection using monoclonal antibodies to the nucleocapsid protein. PLoS Negl Trop Dis. 2016; 10:e0004595.
Article13. Yu F, Du Y, Huang X, et al. Application of recombinant severe fever with thrombocytopenia syndrome virus nucleocapsid protein for the detection of SFTSV-specific human IgG and IgM antibodies by indirect ELISA. Virol J. 2015; 12:117.
Article14. Lee H, Kim EJ, Song JY, et al. Development and evaluation of a competitive enzyme-linked immunosorbent assay using a monoclonal antibody for diagnosis of severe fever with thrombocytopenia syndrome virus in bovine sera. J Vet Sci. 2016; 17:307–314.
Article15. Chen S, Sun L, Liu Y, et al. Development of human antibodies against the Gn protein of severe fever with thrombocytopenia syndrome virus. Bing Du Xue Bao. 2015; 31:24–29.16. Yun SM, Park SJ, Park SW, et al. Molecular genomic characterization of tick- and human-derived severe fever with thrombocytopenia syndrome virus isolates from South Korea. PLoS Negl Trop Dis. 2017; 11:e0005893.
Article17. Harper S, Speicher DW. Expression and purification of GST fusion proteins. Curr Protoc Protein Sci 2008. Chapter 6:Unit 6.6.18. Palmer I, Wingfield PT. Preparation and extraction of insoluble (inclusion-body) proteins from Escherichia coli. Curr Protoc Protein Sci 2004. Chapter 6:Unit 6.3.19. Facility HF. Protocol for immunofluorescence staining of adhesion cells [Internet]. Hong Kong: Li Ka Shing Faculty of Medicine;2017. cited 2017 Dec 2. Available from: https://www.med.hku.hk/corefac/downloads/immunostaining%20protocol%20cell%20culture.pdf.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A serological study of severe fever with thrombocytopenia syndrome using a virus neutralization test and competitive enzyme-linked immunosorbent assay
- Development and evaluation of a competitive enzyme-linked immunosorbent assay using a monoclonal antibody for diagnosis of severe fever with thrombocytopenia syndrome virus in bovine sera
- Two Cases of Dengue Fever Due to Dengue Virus-1 Developed in a Family
- Dengue Fever Mimicking Acute Appendicitis: A Case Report
- Evaluation of enzymum system@(ES-300) for enzyme linked immunosorbent assay: comparison with RIA and CLIA for T3, T4, fT4 and TSH