Korean J Neurotrauma.
2017 Oct;13(2):68-75. 10.13004/kjnt.2017.13.2.68.
Expression of Aquaporin 1 and 4 in the Choroid Plexus and Brain Parenchyma of Kaolin-Induced Hydrocephalic Rats
- Affiliations
-
- 1Department of Neurosurgery, Kyungpook National Univeristy Hospital, Daegu, Korea. shwang@knu.ac.kr
- KMID: 2394537
- DOI: http://doi.org/10.13004/kjnt.2017.13.2.68
Abstract
OBJECTIVE
Aquaporin (AQP) is a recently discovered protein that regulates water homeostasis. The present study examines changes in AQP 1 and 4 in kaolin induced experimental hydrocephalic rats to elucidate the pathophysiology of water homeostasis in the disease.
METHODS
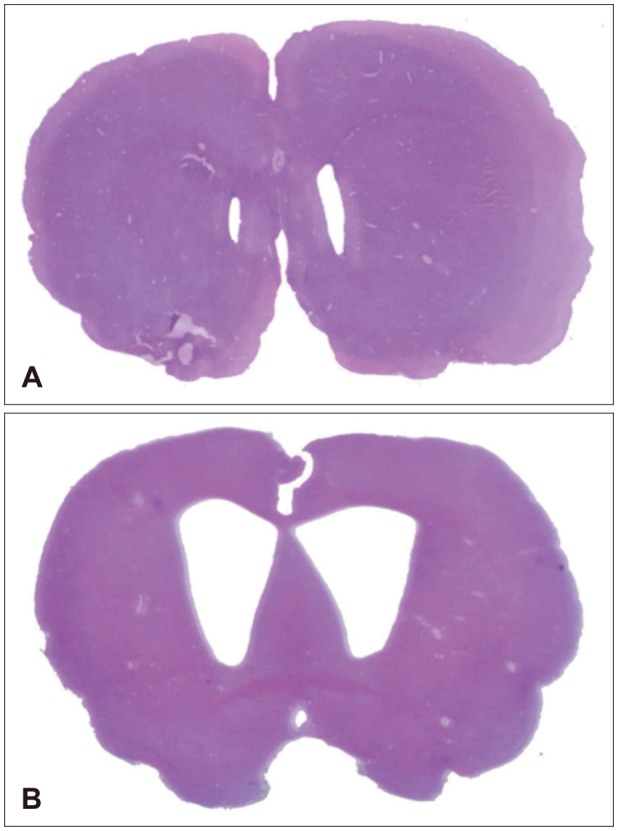
Hydrocephalus was induced by percutaneous intracisternal injection of kaolin. The brain parenchyma and choroid plexus were obtained at 3, 7, 14 and 30 days after injection. Protein expressions of AQP 1 and 4 were measured by western blot, immunohistochemistry (IHC) and immunofluorescence (IF) stains.
RESULTS
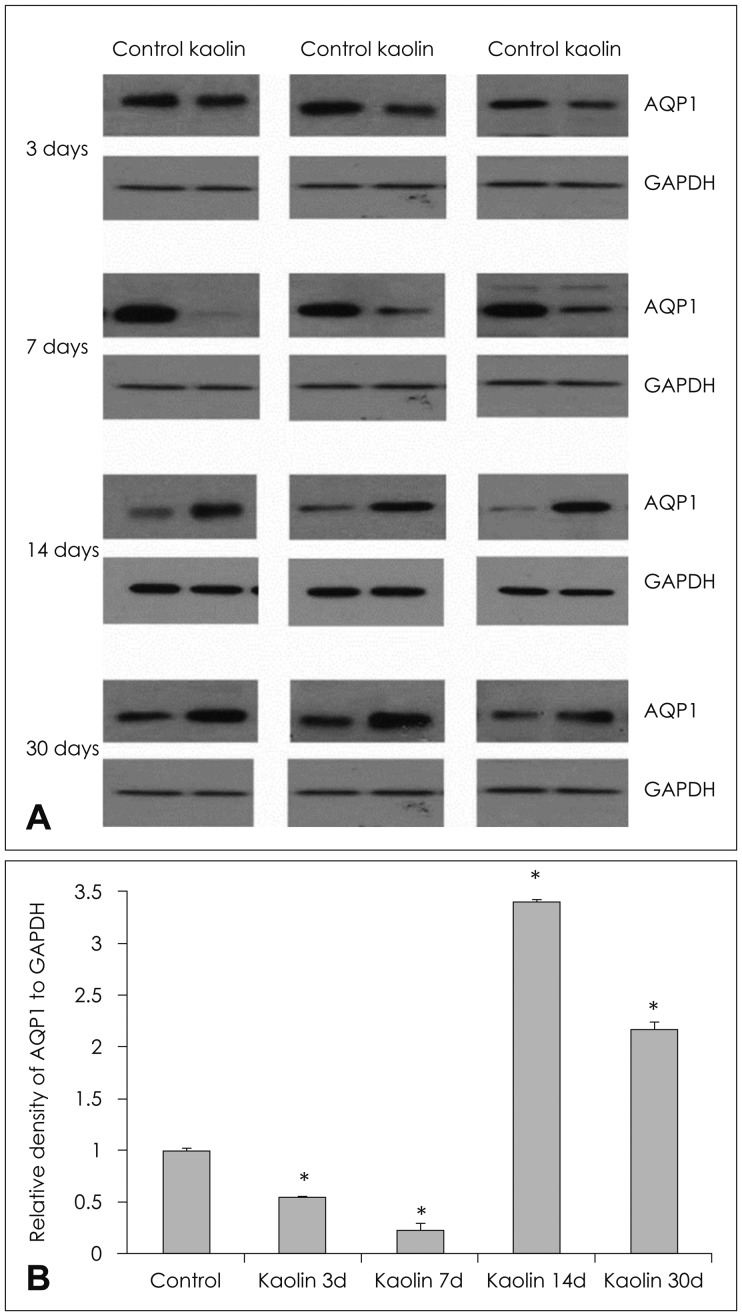
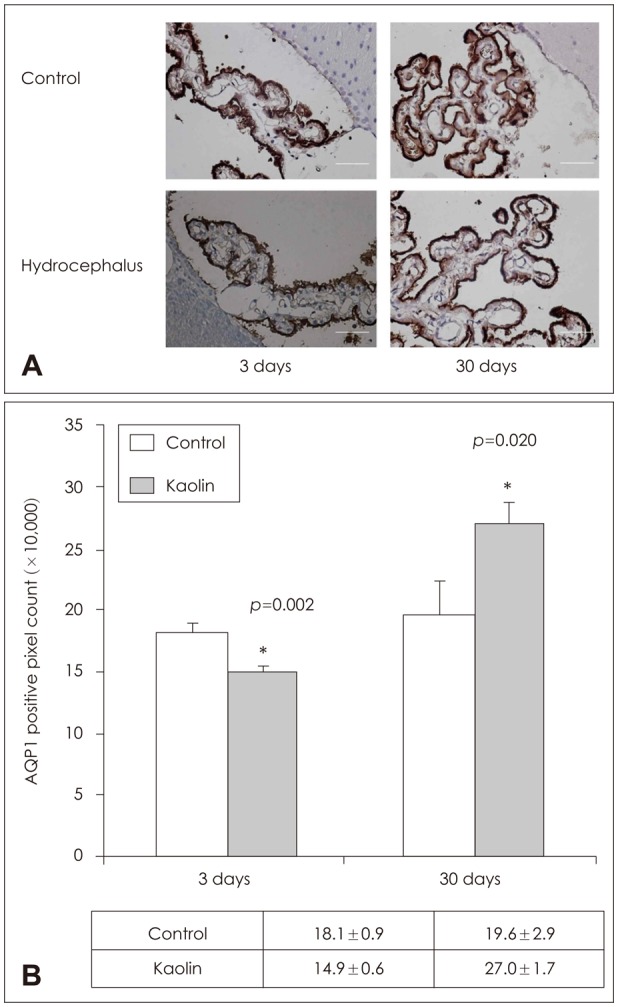
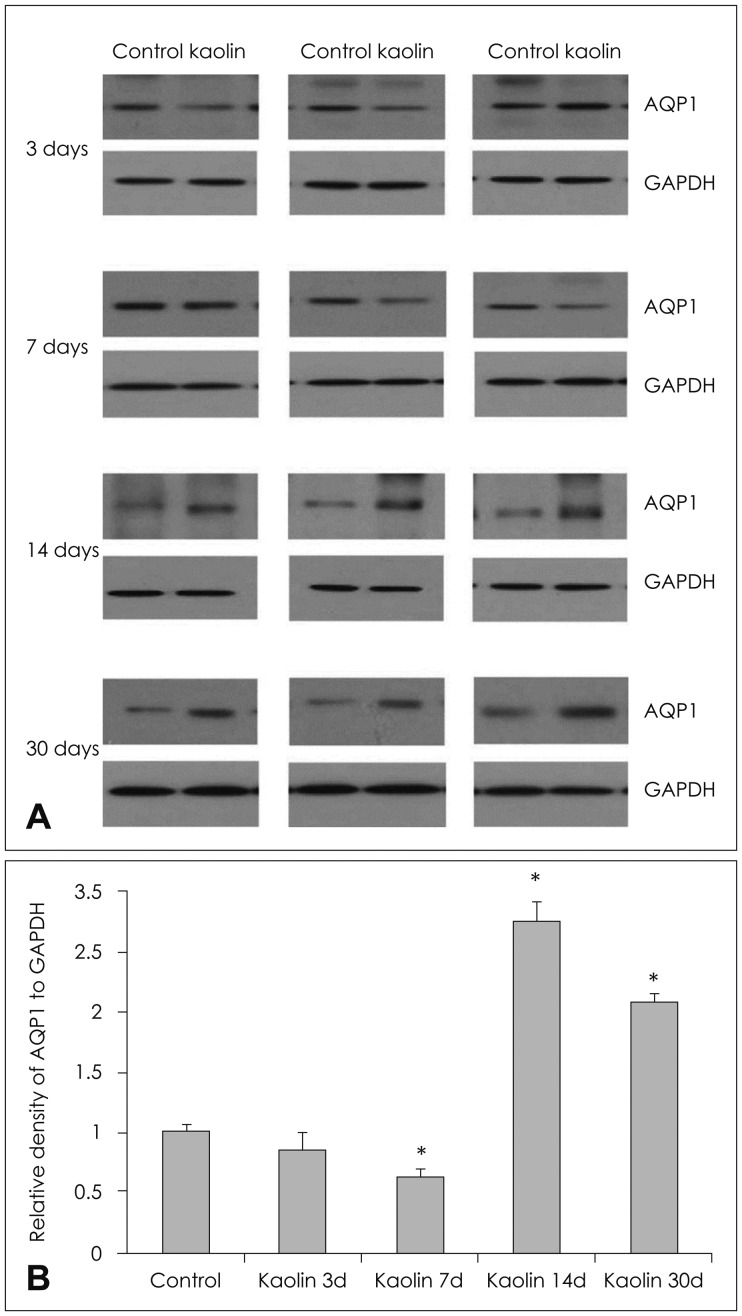
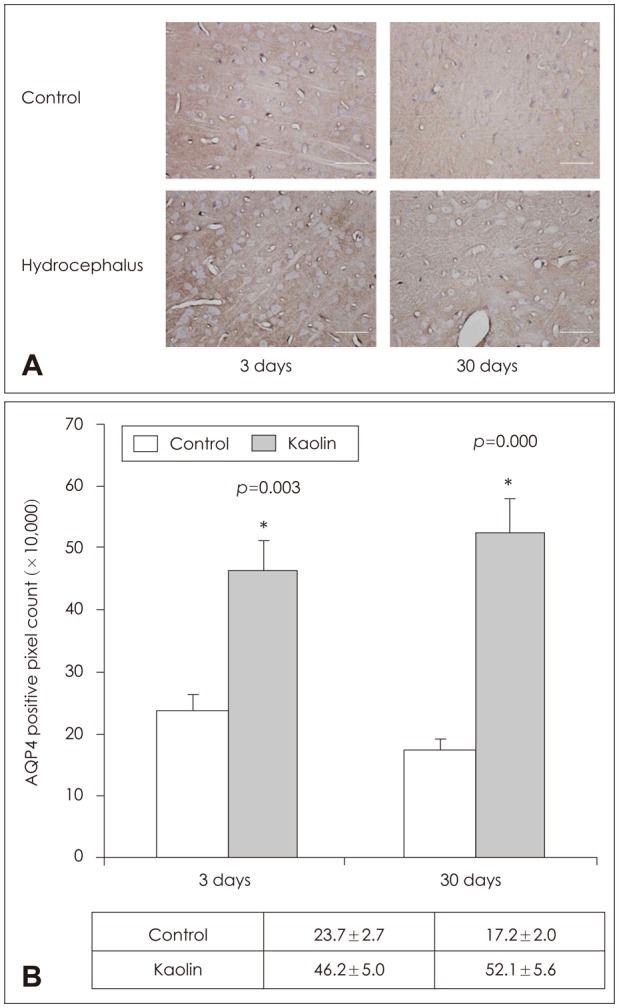
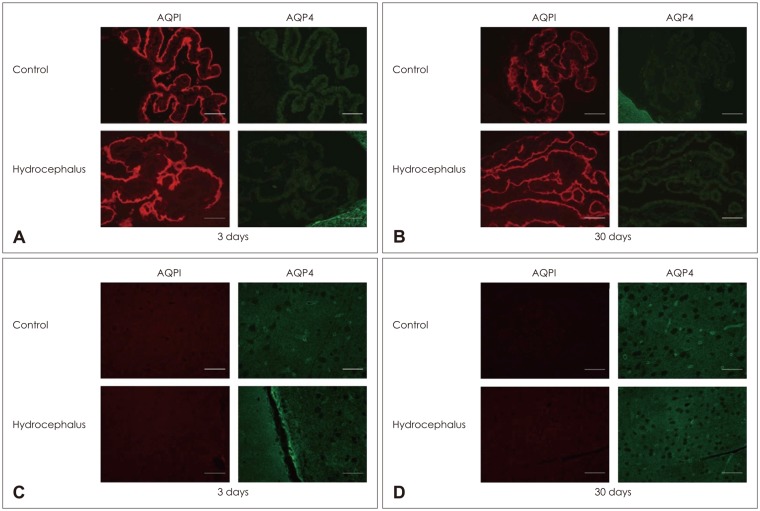
In the choroid plexus of the kaolin-induced hydrocephalus group, AQP 1 expression identified by western blot exhibited sharp decrease in the early stage (55% by the 3rd day and 22% by the 7th day), but indicated a 2.2-fold increase in the later stage (30th day) in comparison with control groups. In the parenchyma, a quantitative measurement of AQP 4 expression revealed variable results on the 3rd and 7th days, but indicated expression 2.1 times higher than the control in the later stage (30th day). In addition, the IHC and IF findings supported the patterns of expression of AQP 1 in the choroid plexus and AQP 4 in the parenchyma.
CONCLUSION
Expression of AQP 1 decreased sharply in the choroid plexus of acute hydrocephalus rats and increased at later stages. Expression of AQP 4 in the brain parenchyma was variable in the early stage in the hydrocephalus group, but was higher than in the control in the later stage. These findings suggest a compensating role of AQPs in water physiology in hydrocephalus.
Keyword
MeSH Terms
Figure
Reference
-
1. Al-Tamimi YZ, Sinha P, Chumas PD, Crimmins D, Drake J, Kestle J, et al. Ventriculoperitoneal shunt 30-day failure rate: a retrospective international cohort study. Neurosurgery. 2014; 74:29–34. PMID: 24089046.
Article2. Ameli PA, Madan M, Chigurupati S, Yu A, Chan SL, Pattisapu JV. Effect of acetazolamide on aquaporin-1 and fluid flow in cultured choroid plexus. Acta Neurochir Suppl. 2012; 113:59–64. PMID: 22116425.
Article3. Bloch O, Auguste KI, Manley GT, Verkman AS. Accelerated progression of kaolin-induced hydrocephalus in aquaporin-4-deficient mice. J Cereb Blood Flow Metab. 2006; 26:1527–1537. PMID: 16552421.
Article4. Drake JM, Kulkarni AV, Kestle J. Endoscopic third ventriculostomy versus ventriculoperitoneal shunt in pediatric patients: a decision analysis. Childs Nerv Syst. 2009; 25:467–472. PMID: 19139908.
Article5. Filippidis AS, Kalani MY, Rekate HL. Hydrocephalus and aquaporins: the role of aquaporin-4. Acta Neurochir Suppl. 2012; 113:55–58. PMID: 22116424.
Article6. Ivkovic M, Reiss-Zimmermann M, Katzen H, Preuss M, Kovanlikaya I, Heier L, et al. MRI assessment of the effects of acetazolamide and external lumbar drainage in idiopathic normal pressure hydrocephalus. Fluids Barriers CNS. 2015; 12:9. PMID: 25928394.
Article7. Johanson CE, Duncan JA 3rd, Klinge PM, Brinker T, Stopa EG, Silverberg GD. Multiplicity of cerebrospinal fluid functions: New challenges in health and disease. Cerebrospinal Fluid Res. 2008; 5:10. PMID: 18479516.
Article8. Juhler M. Animal models of hydrocephalus. In : Rigamonti D, editor. Adult hydrocephalus. New York, NY: Cambridge University Press;2014. p. 28–35.9. Kadrian D, van Gelder J, Florida D, Jones R, Vonau M, Teo C, et al. Long-term reliability of endoscopic third ventriculostomy. Neurosurgery. 2005; 56:1271–1278. PMID: 15918943.
Article10. Kalani MY, Filippidis AS, Rekate HL. Hydrocephalus and aquaporins: the role of aquaporin-1. Acta Neurochir Suppl. 2012; 113:51–54. PMID: 22116423.
Article11. Kowarik MC, Soltys J, Bennett JL. The treatment of neuromyelitis optica. J Neuroophthalmol. 2014; 34:70–82. PMID: 24531318.
Article12. Li J, McAllister JP 2nd, Shen Y, Wagshul ME, Miller JM, Egnor MR, et al. Communicating hydrocephalus in adult rats with kaolin obstruction of the basal cisterns or the cortical subarachnoid space. Exp Neurol. 2008; 211:351–361. PMID: 18433747.
Article13. Longatti P, Basaldella L, Orvieto E, Dei Tos A, Martinuzzi A. Aquaporin(s) expression in choroid plexus tumours. Pediatr Neurosurg. 2006; 42:228–233. PMID: 16714863.
Article14. Mao X, Enno TL, Del Bigio MR. Aquaporin 4 changes in rat brain with severe hydrocephalus. Eur J Neurosci. 2006; 23:2929–2936. PMID: 16819982.15. McAllister JP, Eskandari R, Limbrick DD Jr. Experimental hydrocephalus. In : Winn HR, editor. Youmans & Winn neurological surgery. Philadelphia, PA: Elsevier;2017. p. 1614.16. Nagelhus EA, Ottersen OP. Physiological roles of aquaporin-4 in brain. Physiol Rev. 2013; 93:1543–1562. PMID: 24137016.
Article17. Oshio K, Watanabe H, Song Y, Verkman AS, Manley GT. Reduced cerebrospinal fluid production and intracranial pressure in mice lacking choroid plexus water channel Aquaporin-1. FASEB J. 2005; 19:76–78. PMID: 15533949.
Article18. Papadopoulos MC, Bennett JL, Verkman AS. Treatment of neuromyelitis optica: state-of-the-art and emerging therapies. Nat Rev Neurol. 2014; 10:493–506. PMID: 25112508.
Article19. Papadopoulos MC, Manley GT, Krishna S, Verkman AS. Aquaporin-4 facilitates reabsorption of excess fluid in vasogenic brain edema. FASEB J. 2004; 18:1291–1293. PMID: 15208268.
Article20. Papadopoulos MC, Verkman AS. Aquaporin water channels in the nervous system. Nat Rev Neurosci. 2013; 14:265–277. PMID: 23481483.
Article21. Park MK, Kim M, Park KS, Park SH, Hwang JH, Hwang SK. A retrospective analysis of ventriculoperitoneal shunt revision cases of a single institute. J Korean Neurosurg Soc. 2015; 57:359–363. PMID: 26113963.
Article22. Paul L, Madan M, Rammling M, Chigurupati S, Chan SL, Pattisapu JV. Expression of aquaporin 1 and 4 in a congenital hydrocephalus rat model. Neurosurgery. 2011; 68:462–473. PMID: 21135737.
Article23. Pollay M. Overview of the CSF dual outflow system. Acta Neurochir Suppl. 2012; 113:47–50. PMID: 22116422.
Article24. Preston GM, Agre P. Isolation of the cDNA for erythrocyte integral membrane protein of 28 kilodaltons: member of an ancient channel family. Proc Natl Acad Sci U S A. 1991; 88:11110–11114. PMID: 1722319.
Article25. Reddy GK, Bollam P, Caldito G. Ventriculoperitoneal shunt surgery and the risk of shunt infection in patients with hydrocephalus: long-term single institution experience. World Neurosurg. 2012; 78:155–163. PMID: 22120565.
Article26. Shen XQ, Miyajima M, Ogino I, Arai H. Expression of the water-channel protein aquaporin 4 in the H-Tx rat: possible compensatory role in spontaneously arrested hydrocephalus. J Neurosurg. 2006; 105:459–464. PMID: 17184078.
Article27. Smith ZA, Moftakhar P, Malkasian D, Xiong Z, Vinters HV, Lazareff JA. Choroid plexus hyperplasia: surgical treatment and immunohistochemical results. Case report. J Neurosurg. 2007; 107:255–262. PMID: 17918538.28. Stone JJ, Walker CT, Jacobson M, Phillips V, Silberstein HJ. Revision rate of pediatric ventriculoperitoneal shunts after 15 years. J Neurosurg Pediatr. 2013; 11:15–19. PMID: 23101557.
Article29. Tait MJ, Saadoun S, Bell BA, Papadopoulos MC. Water movements in the brain: role of aquaporins. Trends Neurosci. 2008; 31:37–43. PMID: 18054802.
Article30. Verkman AS. Aquaporins in clinical medicine. Annu Rev Med. 2012; 63:303–316. PMID: 22248325.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Different expressions of AQP1, AQP4, eNOS, and VEGF proteins in ischemic versus non-ischemic cerebropathy in rats: potential roles of AQP1 and eNOS in hydrocephalic and vasogenic edema formation
- Effect of Hydrocephalus and Skull Defect on the Pressure Volume Index
- Effect on the Regional Cerebral Blood Flow and Biomechanical Factors in Ventricular Enlargement of Experimental Hydrocephalus
- Somatosensory Evoked Potentials in Ventricular Enlargement of Experimental Hydrocephalus
- A Case of Choroid Plexus Carcinoma with Rapid Dissemination after Gross Total Resection