Allergy Asthma Respir Dis.
2017 Mar;5(2):63-72. 10.4168/aard.2017.5.2.63.
Development and properties of hypoallergenic infant formula
- Affiliations
-
- 1Research and Development Center, Maeil Dairies Co., Ltd., Pyeongtaek, Korea. woelkyu@naver.com
- 2Development of Pediatrics, Yonsei University College of Medicine, Seoul, Korea.
- KMID: 2392376
- DOI: http://doi.org/10.4168/aard.2017.5.2.63
Abstract
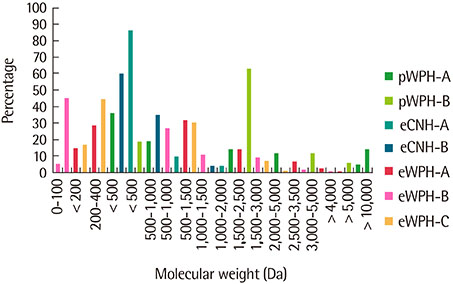
- Milk proteins are composed of casein, further classified into αS1-casein, αS2-casein, β-casein, and κ-casein, and whey protein, which is separated into α-lacatalbumin, β-lactoglobulin, serum albumin, and some minor proteins, such as lactoferrin and immunoglobulin. To reduce the allergenicity of protein, heat treatment and enzymatic protein hydrolysis by endopeptidase are necessarily required. Additionally, membrane technology should be applied to produce a protein hydrolyzate, which has consistent molecular weight of peptide and low in free amino acid without allergenic peptide or protein. Extensive casein hydrolyzate and whey protein hydrolyzate are used for protein source of mainly extensively hydrolyzed protein formula (eHF) intended for the treatment of cow's milk allergy. Also, partially hydrolyzed formula (pHF) is developed, which is using a single protein source e.g., whey protein hydrolyzate. The allergenicity of infant formula can be determined according to molecular weight profile and antigenicity reduction compared to intact protein. More than 90% peptides are present in eHF have a molecular weight of <3,000 Da. Peptide molecular weight profiles of pHF range mainly between 3,000 and 10,000 Da, but have a small percentage of >10,000 Da. Generally, antigenicity reduction in eHF and pHF is 10-6 and 10-3, respectively. Even if protein hydrolyzate is manufactured under strict quality control, there is still a risk of cross contamination of allergenic milk components through environmental conditions and the shared manufacturing process. Thus, quality assessment of protein hydrolyzate formula must be performed routinely.
Keyword
MeSH Terms
Figure
Reference
-
1. World Health Organization. Report of an expert consultation on the optimal duration of exclusive breastfeeding. Geneva (Switzerland): World Health Organization;2012.2. Institute of Medicine (U.S.), Committee on the Evaluation of the Addition of Ingredients New to Infant Formula, Food and Nutrition Board. Infant formula: evaluating the safety of new ingredients. Washington, DC: National Academies Press;2004. p. 22–42.3. Aas K. The Biochemistry of food allergens: what is essential for future research?. In : Schmit E, editor. Food allergy. New York: Vevey/Raven Press, Ltd.;1998. p. 1–14. (Nestle Nutrition Workshop Series; v. 17).4. Zeiger RS, Heller S, Mellon M, O'Connor R, Hamburger RN. Effectiveness of dietary manipulation in the prevention of food allergy in infants. J Allergy Clin Immunol. 1986; 78(1 Pt 2):224–238.
Article5. Schoch G. Milk protein and clinical nutrition. In : Barth CA, Schlimme E, editors. Milk proteins: nutritional, clinical, functional and technological aspects. Darmstadt: Stenikoff;1988. p. 299–301.6. Heppell LM. Determination of milk protein denaturation by an enzyme-linked immunosorbent assay. In : Morris BA, Clifford MN, editors. Immunoassay in food analysis. New York: Elsevier Applied Science Publishers;1985. p. 115–123.7. Ratner B, Dworetzky M, Oguri S, Aschheim L. Studies on the allergenicity of cow's milk. I. The allergenic properties of alpha-casein, beta-lactoglobulin and alpha-lactalbumin. Pediatrics. 1958; 22:449–452.8. Ratner B, Dworetzky M, Oguri S, Aschheim L. Studies on the allergenicity of cow's milk. II. Effect of heat treatment on the allergenicity of milk and protein fractions from milk as tested in guinea pigs by parenteral sensitization and challenge. Pediatrics. 1958; 22(4 Part 1):648–652.9. Kilshaw PJ, Heppell LM, Ford JE. Effects of heat treatment of cow's milk and whey on the nutritional quality and antigenic properties. Arch Dis Child. 1982; 57:842–847.
Article10. Ratner B, Dworetzky M, Oguri S, Aschheim L. Studies on the allergenicity of cow's milk. III. Effect of heat treatment on the allergenicity of milk and protein fractions from milk as tested in guinea pigs by sensitization and challenge by the oral route. Pediatrics. 1958; 22(4 Part 1):653–658.11. Jakobsson I, Lindberg T, Benediktsson B. In vitro digestion of cow's milk proteins by duodenal juice from infants with various gastrointestinal disorders. J Pediatr Gastroenterol Nutr. 1982; 1:183–191.
Article12. Asselin J, Aniot J, Gauthier SF, Maurad W, Hebert J. Immunogenicity and allergenicity of whey protein hydrolysates. J Food Sci. 1988; 53:1208–1211.
Article13. Asselin J, Hebert J, Amiot J. Effect of in vitro proteolysis on the allergenicity of major whey proteins. J Food Sci. 1989; 54:1037–1039.
Article14. Pahud JJ, Monti JC, Jost R. Allergenicity of whey protein: its modification by tryptic in vitro hydrolysis of the protein. J Pediatr Gastroenterol Nutr. 1985; 4:408–413.15. Okamoto M, Rikimaru H, Enomoto A, Kaminogawa S, Yamauchi K. High pressure digestion of food proteins: selective elimination of β-lactoglobulin in bovine milk whey concentrate. Agric Biol Chem. 1991; 55:1253–1257.
Article16. Schmidt DG. Association of caseins and casein micelle structure. In : Fox PF, editor. Development in dairy chemistry. London: Applied Science;1982. p. 61–86.17. Wal JM. Cow's milk proteins/allergens. Ann Allergy Asthma Immunol. 2002; 89:6 Suppl 1. 3–10.
Article18. Farrell HM Jr, Jimenez-Flores R, Bleck GT, Brown EM, Butler JE, Creamer LK, et al. Nomenclature of the proteins of cows' milk--sixth revision. J Dairy Sci. 2004; 87:1641–1674.
Article19. Steinke M, Fiocchi A, Kirchlechner V, Ballmer-Weber B, Brockow K, Hischenhuber C, et al. Perceived food allergy in children in 10 European nations. a randomised telephone survey. Int Arch Allergy Immunol. 2007; 143:290–295.
Article20. Park M, Kim D, Ahn K, Kim J, Han Y. Prevalence of immediate-type food allergy in early childhood in seoul. Allergy Asthma Immunol Res. 2014; 6:131–136.
Article21. Høst A, Husby S, Osterballe O. A prospective study of cow's milk allergy in exclusively breast-fed infants. Incidence, pathogenetic role of early inadvertent exposure to cow's milk formula, and characterization of bovine milk protein in human milk. Acta Paediatr Scand. 1988; 77:663–670.22. Gjesing B, Osterballe O, Schwartz B, Wahn U, Løwenstein H. Allergen-specific IgE antibodies against antigenic components in cow milk and milk substitutes. Allergy. 1986; 41:51–56.
Article23. Restani P, Ballabio C, Di Lorenzo C, Tripodi S, Fiocchi A. Molecular aspects of milk allergens and their role in clinical events. Anal Bioanal Chem. 2009; 395:47–56.
Article24. Bernard H, Créminon C, Yvon M, Wal JM. Specificity of the human IgE response to the different purified caseins in allergy to cow's milk proteins. Int Arch Allergy Immunol. 1998; 115:235–244.
Article25. Gaudin JC, Rabesona H, Choiset Y, Yeretssian G, Chobert JM, Sakanyan V, et al. Assessment of the immunoglobulin E-mediated immune response to milk-specific proteins in allergic patients using microarrays. Clin Exp Allergy. 2008; 38:686–693.
Article26. Bernard H, Meisel H, Creminon C, Wal JM. Post-translational phosphorylation affects the IgE binding capacity of caseins. FEBS Lett. 2000; 467:239–244.
Article27. Shek LP, Bardina L, Castro R, Sampson HA, Beyer K. Humoral and cellular responses to cow milk proteins in patients with milk-induced IgE-mediated and non-IgE-mediated disorders. Allergy. 2005; 60:912–919.
Article28. Ruiter B, Trégoat V, M'rabet L, Garssen J, Bruijnzeel-Koomen CA, Knol EF, et al. Characterization of T cell epitopes in alphas1-casein in cow's milk allergic, atopic and non-atopic children. Clin Exp Allergy. 2006; 36:303–310.
Article29. Schulmeister U, Hochwallner H, Swoboda I, Focke-Tejkl M, Geller B, Nystrand M, et al. Cloning, expression, and mapping of allergenic determinants of alphaS1-casein, a major cow's milk allergen. J Immunol. 2009; 182:7019–7029.
Article30. Permyakov EA, Berliner LJ. alpha-Lactalbumin: structure and function. FEBS Lett. 2000; 473:269–274.31. Sawyer WH. Complex between beta-lactoglobulin and kappa-casein. A review. J Dairy Sci. 1969; 52:1347–1355.32. Goldman AS, Sellars WA, Halpern SR, Anderson DW Jr, Furlow TE, Johnson CH Jr. Milk allergy. II. Skin testing of allergic and normal children with purified milk proteins. Pediatrics. 1963; 32:572–579.33. Freier S, Kletter B, Gery I, Lebenthal E, Geifman M. Intolerance to milk protein. J Pediatr. 1969; 75:623–631.
Article34. Lee JM, Yoon JS, Jeon SA, Lee SY. Sensitization patterns of cow's milk and major components in young children with atopic dermatitis. Asia Pac Allergy. 2013; 3:179–185.
Article35. Lebenthal E. Cow's milk protein allergy. Pediatr Clin North Am. 1975; 22:827–833.
Article36. Clement G, Boquet D, Frobert Y, Bernard H, Negroni L, Chatel JM, et al. Epitopic characterization of native bovine beta-lactoglobulin. J Immunol Methods. 2002; 266:67–78.37. Tsabouri S, Douros K, Priftis KN. Cow's milk allergenicity. Endocr Metab Immune Disord Drug Targets. 2014; 14:16–26.
Article38. Vila L, Beyer K, Järvinen KM, Chatchatee P, Bardina L, Sampson HA. Role of conformational and linear epitopes in the achievement of tolerance in cow's milk allergy. Clin Exp Allergy. 2001; 31:1599–1606.
Article39. Lin J, Sampson HA. The role of immunoglobulin E-binding epitopes in the characterization of food allergy. Curr Opin Allergy Clin Immunol. 2009; 9:357–363.
Article40. Nowak-Wegrzyn A, Fiocchi A. Rare, medium, or well done? The effect of heating and food matrix on food protein allergenicity. Curr Opin Allergy Clin Immunol. 2009; 9:234–237.
Article41. Hartmann R, Wal JM, Bernard H, Pentzien AK. Cytotoxic and allergenic potential of bioactive proteins and peptides. Curr Pharm Des. 2007; 13:897–920.
Article42. Alder-Nissen J. Enzymatic hydrolysis of food proteins. New York: Applied Science;1986. p. 57–131.43. Fersht A. Enzyme structure and mechanisms. San Francisco: Freeman;1999. p. 38–43.44. De Masi RV, Dragonetti A, Florino G. Multiple food intolerance: dietary management with a new formula characterized by low allergenic activity and low osmolarity. Riv Ital Pediatr. 1985; 11:273–279.45. Jost R, Monti JC. Partial enzymatic hydrolysis of whey protein by trypsin. J Dairy Sci. 1977; 60:1387–1393.
Article46. Porter DH, Swaisgood HE, Catignani GL. Characterization of a immobilized digestive enzyme system for determination of protein digestibility. J Agric Food Chem. 1984; 32:334–339.
Article47. Jost R, Monti JC, Pahud JJ. Whey protein allergenicity and its reduction by technological means. Food Technol. 1987; 41:118–121.48. Turgeon SL, Gauthier SF. Whey peptide fractions obtained with a two step ultrafiltration process: Production and characterization. J Food Sci. 1990; 55:106–110.
Article49. Monti JC, Jost R, Pahud JJ, Hughes G. Antigenic sites in bovine beta-lactoglobulin [abstract]. Experimentia. 1986; 42:670.50. Ha WK, Juhn SL, Kim JW, Lee SW, Lee JY, Sohn DW. Reduction of the antigenicity of whey protein by enzymatic hydrolysis. Korean J Food Sci Technol. 1994; 26:74–80.51. Görtler I, Urbanek R, Forster J. Characterization of antigens and allergens in hypo-allergenic infant formulae. Eur J Pediatr. 1995; 154:289–294.
Article52. Nakamura T, Sado H, Syukunobe Y. Antigenicity of whey protein hydrolysates fractionated with ultrafiltration membrane. J Japan Soc Food Sci Technol. 1992; 39:113–116.
Article53. Simensma AD, Weijer WJ, Bak HJ. The importance of peptide lengths in hypoallergenic infant formulae. Trends Food Sci Technol. 1990; 4:16–21.
Article54. Van Beresteijn EC, Peeters RA, Kaper J, Meijer R, Robben A, Schmidt D. Molecular mass distribution, immunological properties and nutritive value of whey protein hydrolysates. J Food Prot. 1994; 57:619–625.
Article55. Van Hoeyveld EM, Escalona-Monge M, de Swert LF, Stevens EA. Allergenic and antigenic activity of peptide fragments in a whey hydrolysate formula. Clin Exp Allergy. 1998; 28:1131–1137.
Article56. American Academy of Pediatrics. Committee on Nutrition. Hypoallergenic infant formulas. Pediatrics. 2000; 106(2 Pt 1):346–349.57. Mead Johnson & Company. International direction of company histories [Internet]. Evansville (IN): Mead Johnson & Company;c2016. cited 2016 Aug 30. Available from: http://www.encyclopedia.com/doc/1G2-3480000067.html.58. Plebani A, Albertini A, Scotta S, Ugazio AG. IgE antibodies to hydrolysates of cow milk proteins in children with cow milk allergy. Ann Allergy. 1990; 64:279–280.59. Cordano A. Infant formula – clinical evaluation, its uses, and current feeding practices in the United States and developing countries. In : Vitale JJ, Broitman SA, editors. Advances in human clinical nutrition. London: Martinus Nijhoff;1982. p. 54–62.60. Morinaga Milk Industry Co., Ltd.Research history [Internet]. Tokyo: Morinaga Milk Industry Co., Ltd.;c2016. cited 2016 Aug 30. Available from: http://morinagamilk.co.jp/research/about/history.html.61. Nutricia - Expert in Early-life nutrition. Our long-standing history in science [Internet]. Schiphol (Netherlands): Danone Nutricia Early Life Nutrition;c2016. cited 2016 Sep 12. Available from: http://www.nutriciaeln.com.hk/en/about.62. Kim BY. Pioneering Korea's dairy industry: Maeil's first 33 years. Seoul: Maeil Dairy Industry Co. Ltd.;2005.63. Nutricia – Advanced medical nutrition. a history we are proud of [Internet]. Schiphol (Netherlands): Danone Nutricia Early Life Nutrition;c2015. cited 2016 Sep 12. Available from: http://www.nutricia.com/about/our-history.64. McLaughlan P, Anderson KJ, Coombs RR. An oral screening procedure to determine the sensitizing capacity of infant feeding formulae. Clin Allergy. 1981; 11:311–318.
Article65. Knights R, Manes JD. Composition, molecular weight and antigenicity of casein hydrolyzates used in a formula for food allergic and malabsorptive infants. In : Candra RK, editor. Food allergy. St. John's New Foundland: Nutrition Research Education Foundation;1985. p. 273–285.66. Cordano A, Gastanaduy AS, Graham GG. Absorption and retention from an iso-osmol casein hydrolsates infant formula. Nutr Res. 1988; 8:1353–1362.
Article67. Galeano NF, Lepage G, Leroy C, Belli D, Levy E, Roy CC. Comparison of two special infant formulas designed for the treatment of protracted diarrhea. J Pediatr Gastroenterol Nutr. 1988; 7:76–83.
Article68. Walker-Smith JA, Digeon B, Phillips AD. Evaluation of a casein and a whey hydrolysate for treatment of cow's-milk-sensitive enteropathy. Eur J Pediatr. 1989; 149:68–71.
Article69. Zeiger RS, Heller S, Mellon MH, Forsythe AB, O'Connor RD, Hamburger RN, et al. Effect of combined maternal and infant food-allergen avoidance on development of atopy in early infancy: a randomized study. J Allergy Clin Immunol. 1989; 84:72–89.
Article70. Loeb H, Mozin MJ. Prevention of chronic diarrhea: nutritional implications. J Pediatr Gastroenterol Nutr. 1983; 2:Suppl 1. S328–S334.71. Bertele-Harms RM, Harms HK. Experience with hypoallergenic formulas in the treatment of food allergy in infancy. In : Harms HK, Wahn U, editors. Food allergy in infancy and childhood. Berlin: Springer Verlag;1989. p. 177–187.72. Isolauri E, Sütas Y, Mäkinen-Kiljunen S, Oja SS, Isosomppi R, Turjanmaa K. Efficacy and safety of hydrolyzed cow milk and amino acid-derived formulas in infants with cow milk allergy. J Pediatr. 1995; 127:550–557.
Article73. Cho J, Suh J, Lee JH, Han Y, Ahn K, Lee SI. Comparison of the effects of breast milk feeding with maternal restriction and hypoallergenic milk feeding on the acquisition of tolerance to cow milk. Pediatr Allergy Respir Dis. 2011; 21:207–214.
Article74. Fritsche R. The role of immune tolerance in allergy prevention. In : Hernell O, Schmitz J, editors. Feeding during late Infancy and early childhood: Impact on Health. Basel: Karger;2005. 56:p. 1–14. (Nestle Nutrition Workshop Series Pediatric Program; v. 56).75. Bindels JG, Boerma JA. Hydrolysed cow's milk formulae. Pediatr Allergy Immunol. 1994; 5:189–190.
Article76. Miraglia Del, D'Auria E, Peroni D, Palazzo S, Radaelli G, Comberiati P, et al. Flavor, relative palatability and components of cow's milk hydrolysed formulas and amino acid-based formula. Ital J Pediatr. 2015; 41:42.
Article77. Høst A, Halken S. Hypoallergenic formulas: when, to whom and how long: after more than 15 years we know the right indication! Allergy. 2004; 59:Suppl 78. 45–52.
Article78. von Berg A, Koletzko S, Grübl A, Filipiak-Pittroff B, Wichmann HE, Bauer CP, et al. The effect of hydrolyzed cow's milk formula for allergy prevention in the first year of life: the German Infant Nutritional Intervention Study, a randomized double-blind trial. J Allergy Clin Immunol. 2003; 111:533–540.
Article79. Han YS, Park HY, Ahn KM, Lee JS, Choi HM, Lee SI. Short-term effect of partially hydrolyzed formula on the prevention of development of atopic dermatitis in infants at high risk. J Korean Med Sci. 2003; 18:547–551.
Article80. Hays T, Wood RA. A systematic review of the role of hydrolyzed infant formulas in allergy prevention. Arch Pediatr Adolesc Med. 2005; 159:810–816.
Article81. Knights RJ. Processing and evaluation of protein hydrolysates. In : Lifshitz F, editor. Nutrition for special needs. New York: Marcel Dekker;1985. p. 105–115.82. Cordle CT. Control of food allergies using protein hydrolysates. Food Technol. 1994; 48:72. 76.83. Rosendal A, Barkholt V. Detection of potentially allergenic material in 12 hydrolyzed milk formulas. J Dairy Sci. 2000; 83:2200–2210.
Article84. Lorenz F, Seid M, Tangermann R, Wahn V. Detection of casein antigen in regular and hypoallergenic formula proteins by ELISA: characterization of formula protein fractions according to their molecular weight. In : Schmidt E, editor. Food allergy. New York: Raven Press;1988. p. 215–223. (Nestle Nutrition Workshop Series; v. 17).85. Chatchatee P, Järvinen KM, Bardina L, Beyer K, Sampson HA. Identification of IgE- and IgG-binding epitopes on alpha(s1)-casein: differences in patients with persistent and transient cow's milk allergy. J Allergy Clin Immunol. 2001; 107:379–383.
Article86. Busse PJ, Järvinen KM, Vila L, Beyer K, Sampson HA. Identification of sequential IgE-binding epitopes on bovine alpha(s2)-casein in cow's milk allergic patients. Int Arch Allergy Immunol. 2002; 129:93–96.
Article87. Cerecedo I, Zamora J, Shreffler WG, Lin J, Bardina L, Dieguez MC, et al. Mapping of the IgE and IgG4 sequential epitopes of milk allergens with a peptide microarray-based immunoassay. J Allergy Clin Immunol. 2008; 122:589–594.
Article88. Chatchatee P, Järvinen KM, Bardina L, Vila L, Beyer K, Sampson HA. Identification of IgE and IgG binding epitopes on beta- and kappa-casein in cow's milk allergic patients. Clin Exp Allergy. 2001; 31:1256–1262.
Article89. Järvinen KM, Chatchatee P, Bardina L, Beyer K, Sampson HA. IgE and IgG binding epitopes on alpha-lactalbumin and beta-lactoglobulin in cow's milk allergy. Int Arch Allergy Immunol. 2001; 126:111–118.
Article90. Hochwallner H, Schulmeister U, Swoboda I, Focke-Tejkl M, Civaj V, Balic N, et al. Visualization of clustered IgE epitopes on alpha-lactalbumin. J Allergy Clin Immunol. 2010; 125:1279–1285.e9.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Types of Special Infant Formulas Marketed in Korea and Their Indications
- Growth, Nutrient Intake and AlIergic Manifestations of the Infants with FamiIy HistoIT of Allergy Fed on HypoaIlergenic Formula vs. GeneraI FormuIa
- Special Infant Formula
- Clinician Experience with Using Hypoallergenic Formulas to Treat Infants with Suspected Cow’s Milk Protein Allergy: A Secondary Analysis of a Prospective Survey Cohort
- Zinc and Copper Contents in Infant Formula