Infect Chemother.
2013 Mar;45(1):11-21.
The Battle Between Influenza and the Innate Immune Response in the Human Respiratory Tract
- Affiliations
-
- 1Department of Pathology, Hong Kong University, Hong Kong, Hong Kong. Nicholls@Pathology.Hku.Hk
Abstract
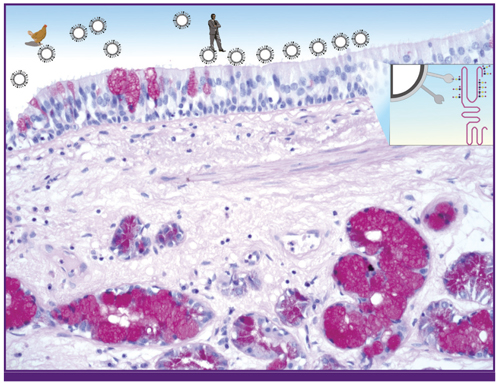
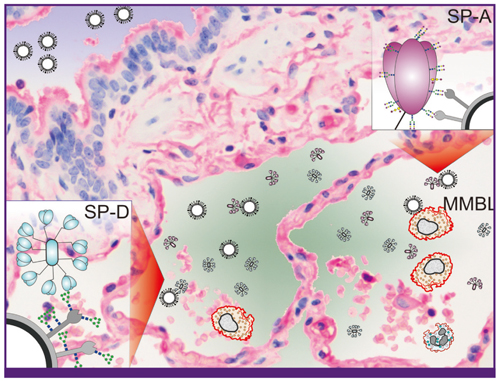
- Influenza is a viral infection of the respiratory tract. Infection is normally confined to the upper respiratory tract but certain viral strains have evolved the ability to infect the lower respiratory tract, including the alveoli, leading to inflammation and a disease pattern of diffuse alveolar damage. Factors leading to this sequence of events are novel influenza strains, or strains that have viral proteins, in particular the NS1 protein that allow it to escape the innate immune system. There are three main barriers that prevent infection of pneumocytes - mucin, host defence lectins and cells such as macrophages. Viruses have developed strategies such as neuraminidase and glycosylation patterns that allow this evasion. Though there has been much investment in antiviral drugs, it is proposed that more attention should be directed towards developing or utilizing compounds that enhance the ability of the innate immune system to combat viral infection.
Keyword
MeSH Terms
Figure
Reference
-
1. Abrahams A, Hallows N, French H. A further investigation into influenza pneumococcal and influenza streptococcal septicaemia: epidemic influenza pneumonia of highly fatal type and its relation to purulent bronchitis. Lancet. 1919. 1:1–9.2. Taubenberger JK. The origin and virulence of the 1918 "Spanish" influenza virus. Proc Am Philos Soc. 2006. 150:86–112.3. Reid AH, Fanning TG, Hultin JV, Taubenberger JK. Origin and evolution of the 1918 "Spanish" influenza virus hemagglutinin gene. Proc Natl Acad Sci U S A. 1999. 96:1651–1656.
Article4. Tscherne DM, García-Sastre A. An enzymatic assay for detection of viral entry. Curr Protoc Cell Biol. 2011. Chapter 26:Unit 26.12.
Article5. Walsh JJ, Dietlein LF, Low FN, Burch GE, Mogabgab WJ. Bronchotracheal response in human influenza. Type A, Asian strain, as studied by light and electron microscopic examination of bronchoscopic biopsies. Arch Intern Med. 1961. 108:376–388.6. Hers JF, Masurel N, Mulder J. Bacteriology and histopathology of the respiratory tract and lungs in fatal Asian influenza. Lancet. 1958. 2:1141–1143.
Article7. Guarner J, Paddock CD, Shieh WJ, Packard MM, Patel M, Montague JL, Uyeki TM, Bhat N, Balish A, Lindstrom S, Klimov A, Zaki SR. Histopathologic and immunohistochemical features of fatal influenza virus infection in children during the 2003-2004 season. Clin Infect Dis. 2006. 43:132–140.
Article8. Shieh WJ, Blau DM, Denison AM, Deleon-Carnes M, Adem P, Bhatnagar J, Sumner J, Liu L, Patel M, Batten B, Greer P, Jones T, Smith C, Bartlett J, Montague J, White E, Rollin D, Gao R, Seales C, Jost H, Metcalfe M, Goldsmith CS, Humphrey C, Schmitz A, Drew C, Paddock C, Uyeki TM, Zaki SR. 2009 pandemic influenza A (H1N1): pathology and pathogenesis of 100 fatal cases in the United States. Am J Pathol. 2010. 177:166–175.9. Uiprasertkul M, Puthavathana P, Sangsiriwut K, Pooruk P, Srisook K, Peiris M, Nicholls JM, Chokephaibulkit K, Vanprapar N, Auewarakul P. Influenza A H5N1 replication sites in humans. Emerg Infect Dis. 2005. 11:1036–1041.
Article10. Gu J, Xie Z, Gao Z, Liu J, Korteweg C, Ye J, Lau LT, Lu J, Gao Z, Zhang B, McNutt MA, Lu M, Anderson VM, Gong E, Yu AC, Lipkin WI. H5N1 infection of the respiratory tract and beyond: a molecular pathology study. Lancet. 2007. 370:1137–1145.
Article11. Liem NT, Nakajima N, Phat le P, Sato Y, Thach HN, Hung PV, San LT, Katano H, Kumasaka T, Oka T, Kawachi S, Matsushita T, Sata T, Kudo K, Suzuki K. H5N1-infected cells in lung with diffuse alveolar damage in exudative phase from a fatal case in Vietnam. Jpn J Infect Dis. 2008. 61:157–160.12. Writing Committee of the Second World Health Organization Consultation on Clinical Aspects of Human Infection with Avian Influenza A (H5N1) Virus. Abdel-Ghafar AN, Chotpitayasunondh T, Gao Z, Hayden FG, Nguyen DH, de Jong MD, Naghdaliyev A, Peiris JS, Shindo N, Soeroso S, Uyeki TM. Update on avian influenza A (H5N1) virus infection in humans. N Engl J Med. 2008. 358:261–273.
Article13. Suptawiwat O, Tantilipikorn P, Boonarkart C, Lumyongsatien J, Uiprasertkul M, Puthavathana P, Auewarakul P. Enhanced susceptibility of nasal polyp tissues to avian and human influenza viruses. PLoS One. 2010. 5:e12973.
Article14. Zhang Z, Zhang J, Huang K, Li KS, Yuen KY, Guan Y, Chen H, Ng WF. Systemic infection of avian influenza A virus H5N1 subtype in humans. Hum Pathol. 2009. 40:735–739.
Article15. Nicholls JM, Butany J, Poon LL, Chan KH, Beh SL, Poutanen S, Peiris JS, Wong M. Time course and cellular localization of SARS-CoV nucleoprotein and RNA in lungs from fatal cases of SARS. PLoS Med. 2006. 3:e27.
Article16. Imai Y, Kuba K, Neely GG, Yaghubian-Malhami R, Perkmann T, van Loo G, Ermolaeva M, Veldhuizen R, Leung YH, Wang H, Liu H, Sun Y, Pasparakis M, Kopf M, Mech C, Bavari S, Peiris JS, Slutsky AS, Akira S, Hultqvist M, Holmdahl R, Nicholls J, Jiang C, Binder CJ, Penninger JM. Identification of oxidative stress and Toll-like receptor 4 signaling as a key pathway of acute lung injury. Cell. 2008. 133:235–249.
Article17. Hirst GK. Adsorption of influenza hemagglutinins and virus by red blood Cells. J Exp Med. 1942. 76:195–209.
Article18. Varki A. Sialic acids as ligands in recognition phenomena. FASEB J. 1997. 11:248–255.
Article19. Varki NM, Varki A. Diversity in cell surface sialic acid presentations: implications for biology and disease. Lab Invest. 2007. 87:851–857.
Article20. Air GM. Influenza neuraminidase. Influenza Other Respi Viruses. 2012. 6:245–256.
Article21. Rogers GN, Paulson JC. Receptor determinants of human and animal influenza virus isolates: differences in receptor specificity of the H3 hemagglutinin based on species of origin. Virology. 1983. 127:361–373.
Article22. Baum LG, Paulson JC. Sialyloligosaccharides of the respiratory epithelium in the selection of human influenza virus receptor specificity. Acta Histochem Suppl. 1990. 40:35–38.23. Scholtissek C, Burger H, Kistner O, Shortridge KF. The nucleoprotein as a possible major factor in determining host specificity of influenza H3N2 viruses. Virology. 1985. 147:287–294.
Article24. Vines A, Wells K, Matrosovich M, Castrucci MR, Ito T, Kawaoka Y. The role of influenza A virus hemagglutinin residues 226 and 228 in receptor specificity and host range restriction. J Virol. 1998. 72:7626–7631.
Article25. Trebbien R, Larsen LE, Viuff BM. Distribution of sialic acid receptors and influenza A virus of avian and swine origin in experimentally infected pigs. Virol J. 2011. 8:434.
Article26. Nelli RK, Kuchipudi SV, White GA, Perez BB, Dunham SP, Chang KC. Comparative distribution of human and avian type sialic acid influenza receptors in the pig. BMC Vet Res. 2010. 6:4.
Article27. Van Poucke SG, Nicholls JM, Nauwynck HJ, Van Reeth K. Replication of avian, human and swine influenza viruses in porcine respiratory explants and association with sialic acid distribution. Virol J. 2010. 7:38.
Article28. Voynow JA, Rubin BK. Mucins, mucus, and sputum. Chest. 2009. 135:505–512.
Article29. Williams OW, Sharafkhaneh A, Kim V, Dickey BF, Evans CM. Airway mucus: From production to secretion. Am J Respir Cell Mol Biol. 2006. 34:527–536.30. Wheeler AH, Nungester WJ. Effect of mucin on influenza virus infection in hamsters. Science. 1942. 96:92–93.
Article31. Lo-Guidice JM, Merten MD, Lamblin G, Porchet N, Houvenaghel MC, Figarella C, Roussel P, Perini JM. Mucins secreted by a transformed cell line derived from human tracheal gland cells. Biochem J. 1997. 326:431–437.
Article32. Couceiro JN, Paulson JC, Baum LG. Influenza virus strains selectively recognize sialyloligosaccharides on human respiratory epithelium; the role of the host cell in selection of hemagglutinin receptor specificity. Virus Res. 1993. 29:155–165.
Article33. Nicholls JM, Bourne AJ, Chen H, Guan Y, Peiris JS. Sialic acid receptor detection in the human respiratory tract: evidence for widespread distribution of potential binding sites for human and avian influenza viruses. Respir Res. 2007. 8:73.
Article34. Ng WC, Tate MD, Brooks AG, Reading PC. Soluble host defense lectins in innate immunity to influenza virus. J Biomed Biotechnol. 2012. 2012:732191.
Article35. Basak S, Tomana M, Compans RW. Sialic acid is incorporated into influenza hemagglutinin glycoproteins in the absence of viral neuraminidase. Virus Res. 1985. 2:61–68.
Article36. Sun S, Wang Q, Zhao F, Chen W, Li Z. Glycosylation site alteration in the evolution of influenza A (H1N1) viruses. PLoS One. 2011. 6:e22844.
Article37. Reading PC, Tate MD, Pickett DL, Brooks AG. Glycosylation as a target for recognition of influenza viruses by the innate immune system. Adv Exp Med Biol. 2007. 598:279–292.
Article38. Pritchett TJ, Paulson JC. Basis for the potent inhibition of influenza virus infection by equine and guinea pig alpha 2-macroglobulin. J Biol Chem. 1989. 264:9850–9858.
Article39. Kingma PS, Zhang L, Ikegami M, Hartshorn K, McCormack FX, Whitsett JA. Correction of pulmonary abnormalities in Sftpd-/- mice requires the collagenous domain of surfactant protein D. J Biol Chem. 2006. 281:24496–24505.
Article40. Reading PC, Allison J, Crouch EC, Anders EM. Increased susceptibility of diabetic mice to influenza virus infection: compromise of collectin-mediated host defense of the lung by glucose? J Virol. 1998. 72:6884–6887.
Article41. Ward KA, Spokes PJ, McAnulty JM. Case-control study of risk factors for hospitalization caused by pandemic (H1N1) 2009. Emerg Infect Dis. 2011. 17:1409–1416.
Article42. Peiris JS, Yu WC, Leung CW, Cheung CY, Ng WF, Nicholls JM, Ng TK, Chan KH, Lai ST, Lim WL, Yuen KY, Guan Y. Re-emergence of fatal human influenza A subtype H5N1 disease. Lancet. 2004. 363:617–619.
Article43. Nicholls JM, Chan MC, Chan WY, Wong HK, Cheung CY, Kwong DL, Wong MP, Chui WH, Poon LL, Tsao SW, Guan Y, Peiris JS. Tropism of avian influenza A (H5N1) in the upper and lower respiratory tract. Nat Med. 2007. 13:147–149.
Article44. Yu WC, Chan RW, Wang J, Travanty EA, Nicholls JM, Peiris JS, Mason RJ, Chan MC. Viral replication and innate host responses in primary human alveolar epithelial cells and alveolar macrophages infected with influenza H5N1 and H1N1 viruses. J Virol. 2011. 85:6844–6855.
Article45. van Riel D, Leijten LM, van der Eerden M, Hoogsteden HC, Boven LA, Lambrecht BN, Osterhaus AD, Kuiken T. Highly pathogenic avian influenza virus H5N1 infects alveolar macrophages without virus production or excessive TNF-alpha induction. PLoS Pathog. 2011. 7:e1002099.
Article46. Upham JP, Pickett D, Irimura T, Anders EM, Reading PC. Macrophage receptors for influenza A virus: role of the macrophage galactose-type lectin and mannose receptor in viral entry. J Virol. 2010. 84:3730–3737.
Article47. Hwang I, Scott JM, Kakarla T, Duriancik DM, Choi S, Cho C, Lee T, Park H, French AR, Beli E, Gardner E, Kim S. Activation mechanisms of natural killer cells during influenza virus infection. PLoS One. 2012. 7:e51858.
Article48. Crouch E, Hartshorn K, Ofek I. Collectins and pulmonary innate immunity. Immunol Rev. 2000. 173:52–65.
Article49. Hillaire ML, van Eijk M, Nieuwkoop NJ, Vogelzang-van Trierum SE, Fouchier RA, Osterhaus AD, Haagsman HP, Rimmelzwaan GF. The number and position of N-linked glycosylation sites in the hemagglutinin determine differential recognition of seasonal and 2009 pandemic H1N1 influenza virus by porcine surfactant protein D. Virus Res. 2012. 169:301–305.
Article50. van Riel D, Munster VJ, de Wit E, Rimmelzwaan GF, Fouchier RA, Osterhaus AD, Kuiken T. Human and avian influenza viruses target different cells in the lower respiratory tract of humans and other mammals. Am J Pathol. 2007. 171:1215–1223.
Article51. Weinheimer VK, Becher A, Tönnies M, Holland G, Knepper J, Bauer TT, Schneider P, Neudecker J, Rückert JC, Szymanski K, Temmesfeld-Wollbrueck B, Gruber AD, Bannert N, Suttorp N, Hippenstiel S, Wolff T, Hocke AC. Influenza A viruses target type II pneumocytes in the human lung. J Infect Dis. 2012. 206:1685–1694.
Article52. Hampton T. Virulence of 1918 influenza virus linked to inflammatory innate immune response. JAMA. 2007. 297:580.
Article53. Chan MC, Cheung CY, Chui WH, Tsao SW, Nicholls JM, Chan YO, Chan RW, Long HT, Poon LL, Guan Y, Peiris JS. Proinflammatory cytokine responses induced by influenza A (H5N1) viruses in primary human alveolar and bronchial epithelial cells. Respir Res. 2005. 6:135.
Article54. van de Sandt CE, Kreijtz JH, Rimmelzwaan GF. Evasion of influenza a viruses from innate and adaptive immune responses. Viruses. 2012. 4:1438–1476.
Article55. Shinya K, Okamura T, Sueta S, Kasai N, Tanaka M, Ginting TE, Makino A, Eisfeld AJ, Kawaoka Y. Toll-like receptor pre-stimulation protects mice against lethal infection with highly pathogenic influenza viruses. Virol J. 2011. 8:97.
Article56. Shinya K, Ito M, Makino A, Tanaka M, Miyake K, Eisfeld AJ, Kawaoka Y. The TLR4-TRIF pathway protects against H5N1 influenza virus infection. J Virol. 2011. 86:19–24.
Article57. Guillot L, Le Goffic R, Bloch S, Escriou N, Akira S, Chignard M, Si-Tahar M. Involvement of toll-like receptor 3 in the immune response of lung epithelial cells to double-stranded RNA and influenza A virus. J Biol Chem. 2005. 280:5571–5580.
Article58. Jeisy-Scott V, Kim JH, Davis WG, Cao W, Katz JM, Sambhara S. TLR7 recognition is dispensable for influenza virus A infection but important for the induction of hemagglutinin-specific antibodies in response to the 2009 pandemic split vaccine in mice. J Virol. 2012. 86:10988–10998.
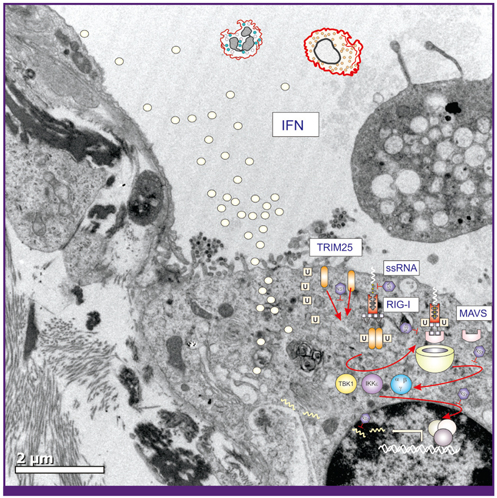
Article59. Bowzard JB, Davis WG, Jeisy-Scott V, Ranjan P, Gangappa S, Fujita T, Sambhara S. PAMPer and tRIGer: ligand-induced activation of RIG-I. Trends Biochem Sci. 2011. 36:314–319.
Article60. Wolff T, Zielecki F, Abt M, Voss D, Semmler I, Matthaei M. Sabotage of antiviral signaling and effectors by influenza viruses. Biol Chem. 2008. 389:1299–1305.
Article61. Munir M. TRIM proteins: another class of viral victims. Sci Signal. 2010. 3:jc2.
Article62. Ramos I, Fernandez-Sesma A. Cell receptors for influenza a viruses and the innate immune response. Front Microbiol. 2012. 3:117.
Article63. Hornung V, Ellegast J, Kim S, Brzózka K, Jung A, Kato H, Poeck H, Akira S, Conzelmann KK, Schlee M, Endres S, Hartmann G. 5'-Triphosphate RNA is the ligand for RIG-I. Science. 2006. 314:994–997.
Article64. Pichlmair A, Schulz O, Tan CP, Näslund TI, Liljeström P, Weber F, Reis e Sousa C. RIG-I-mediated antiviral responses to single-stranded RNA bearing 5'-phosphates. Science. 2006. 314:997–1001.
Article65. Gack MU, Shin YC, Joo CH, Urano T, Liang C, Sun L, Takeuchi O, Akira S, Chen Z, Inoue S, Jung JU. TRIM25 RING-finger E3 ubiquitin ligase is essential for RIG-I-mediated antiviral activity. Nature. 2007. 446:916–920.
Article66. Ozato K, Shin DM, Chang TH, Morse HC 3rd. TRIM family proteins and their emerging roles in innate immunity. Nat Rev Immunol. 2008. 8:849–860.
Article67. Steinberg BE, Goldenberg NM, Lee WL. Do viral infections mimic bacterial sepsis? The role of microvascular permeability: A review of mechanisms and methods. Antiviral Res. 2012. 93:2–15.
Article68. Chan MC, Chan RW, Yu WC, Ho CC, Chui WH, Lo CK, Yuen KM, Guan YI, Nicholls JM, Peiris JS. Influenza H5N1 virus infection of polarized human alveolar epithelial cells and lung microvascular endothelial cells. Respir Res. 2009. 10:102.
Article69. Iwasaki A, Medzhitov R. A new shield for a cytokine storm. Cell. 2011. 146:861–862.
Article70. Marsolais D, Hahm B, Walsh KB, Edelmann KH, McGavern D, Hatta Y, Kawaoka Y, Rosen H, Oldstone MB. A critical role for the sphingosine analog AAL-R in dampening the cytokine response during influenza virus infection. Proc Natl Acad Sci U S A. 2009. 106:1560–1565.
Article71. Teijaro JR, Walsh KB, Cahalan S, Fremgen DM, Roberts E, Scott F, Martinborough E, Peach R, Oldstone MB, Rosen H. Endothelial cells are central orchestrators of cytokine amplification during influenza virus infection. Cell. 2011. 146:980–991.
Article72. Walsh KB, Teijaro JR, Rosen H, Oldstone MB. Quelling the storm: utilization of sphingosine-1-phosphate receptor signaling to ameliorate influenza virus-induced cytokine storm. Immunol Res. 2011. 51:15–25.
Article73. Kochs G, García-Sastre A, Martínez-Sobrido L. Multiple anti-interferon actions of the influenza A virus NS1 protein. J Virol. 2007. 81:7011–7021.
Article74. García-Sastre A, Egorov A, Matassov D, Brandt S, Levy DE, Durbin JE, Palese P, Muster T. Influenza A virus lacking the NS1 gene replicates in interferon-deficient systems. Virology. 1998. 252:324–330.
Article75. Li M, Wang B. Homology modeling and examination of the effect of the D92E mutation on the H5N1 nonstructural protein NS1 effector domain. J Mol Model. 2007. 13:1237–1244.
Article76. García MA, Meurs EF, Esteban M. The dsRNA protein kinase PKR: virus and cell control. Biochimie. 2007. 89:799–811.
Article77. Chen GW, Chang SC, Mok CK, Lo YL, Kung YN, Huang JH, Shih YH, Wang JY, Chiang C, Chen CJ, Shih SR. Genomic signatures of human versus avian influenza A viruses. Emerg Infect Dis. 2006. 12:1353–1360.
Article78. Jackson D, Hossain MJ, Hickman D, Perez DR, Lamb RA. A new influenza virus virulence determinant: the NS1 protein four C-terminal residues modulate pathogenicity. Proc Natl Acad Sci U S A. 2008. 105:4381–4386.
Article79. Soubies SM, Volmer C, Croville G, Loupias J, Peralta B, Costes P, Lacroux C, Guérin JL, Volmer R. Species-specific contribution of the four C-terminal amino acids of influenza A virus NS1 protein to virulence. J Virol. 2010. 84:6733–6747.
Article80. Soubies SM, Hoffmann TW, Croville G, Larcher T, Ledevin M, Soubieux D, Quéré P, Guérin JL, Marc D, Volmer R. Deletion of the C-terminal ESEV domain of NS1 does not affect the replication of a low-pathogenic avian influenza virus H7N1 in ducks and chickens. J Gen Virol. 2013. 94:50–58.
Article81. Fedson DS. Confronting the next influenza pandemic with anti-inflammatory and immunomodulatory agents: why they are needed and how they might work. Influenza Other Respi Viruses. 2009. 3:129–142.
Article82. Radigan KA, Urich D, Misharin AV, Chiarella SE, Soberanes S, Gonzalez A, Perlman H, Wunderink RG, Budinger GR, Mutlu GM. The effect of rosuvastatin in a murine model of influenza A infection. PLoS One. 2012. 7:e35788.
Article83. Tu W, Zheng J, Liu Y, Sia SF, Liu M, Qin G, Ng IH, Xiang Z, Lam KT, Peiris JS, Lau YL. The aminobisphosphonate pamidronate controls influenza pathogenesis by expanding a gammadelta T cell population in humanized mice. J Exp Med. 2011. 208:1511–1522.
Article84. Szretter KJ, Gangappa S, Belser JA, Zeng H, Chen H, Matsuoka Y, Sambhara S, Swayne DE, Tumpey TM, Katz JM. Early control of H5N1 influenza virus replication by the type I interferon response in mice. J Virol. 2009. 83:5825–5834.
Article85. Loutfy MR, Blatt LM, Siminovitch KA, Ward S, Wolff B, Lho H, Pham DH, Deif H, LaMere EA, Chang M, Kain KC, Farcas GA, Ferguson P, Latchford M, Levy G, Dennis JW, Lai EK, Fish EN. Interferon alfacon-1 plus corticosteroids in severe acute respiratory syndrome: a preliminary study. JAMA. 2003. 290:3222–3228.
Article86. Van Hoeven N, Belser JA, Szretter KJ, Zeng H, Staeheli P, Swayne DE, Katz JM, Tumpey TM. Pathogenesis of 1918 pandemic and H5N1 influenza virus infections in a guinea pig model: antiviral potential of exogenous alpha interferon to reduce virus shedding. J Virol. 2009. 83:2851–2861.
Article87. Kugel D, Kochs G, Obojes K, Roth J, Kobinger GP, Kobasa D, Haller O, Staeheli P, von Messling V. Intranasal administration of alpha interferon reduces seasonal influenza A virus morbidity in ferrets. J Virol. 2009. 83:3843–3851.
Article88. Finter NB, Chapman S, Dowd P, Johnston JM, Manna V, Sarantis N, Sheron N, Scott G, Phua S, Tatum PB. The use of interferon-alpha in virus infections. Drugs. 1991. 42:749–765.89. Herzog C, Berger R, Fernex M, Friesecke K, Havas L, Just M, Dubach UC. Intranasal interferon (rIFN-alpha A, Ro 22-8181) for contact prophylaxis against common cold: a randomized, double-blind and placebo-controlled field study. Antiviral Res. 1986. 6:171–176.
Article90. Jia D, Rahbar R, Chan RW, Lee SM, Chan MC, Wang BX, Baker DP, Sun B, Peiris JS, Nicholls JM, Fish EN. Influenza virus non-structural protein 1 (NS1) disrupts interferon signaling. PLoS One. 2010. 5:e13927.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Host Immune Responses Against Type A Influenza Viruses
- Reactive Oxygen Species, Interferon and Antiviral Innate Immune Response in Nasal Mucosa
- Differential Roles of Lung Dendritic Cell Subsets Against Respiratory Virus Infection
- Recovery of respiratory syncytial virus, adenovirus, influenza virus , and parainfluenza virus from nasopharyngeal aspirates from children with acute respiratory tract infections
- The Gate Way of Communication between Microorganism and Human Body: Toll-like Receptors