J Gastric Cancer.
2017 Sep;17(3):204-211. 10.5230/jgc.2017.17.e21.
External Validation of a Gastric Cancer Nomogram Derived from a Large-volume Center Using Dataset from a Medium-volume Center
- Affiliations
-
- 1Department of Surgery, Konkuk University Medical Center, Seoul, Korea. handongseok@gmail.com
- 2Department of Surgery, Asan Medical Center, Seoul, Korea.
- 3Department of Pathology, Konkuk University Medical Center, Seoul, Korea.
- 4Department of Surgery, Seoul National University Hospital, Seoul, Korea.
- KMID: 2389824
- DOI: http://doi.org/10.5230/jgc.2017.17.e21
Abstract
- PURPOSE
Recently, a nomogram predicting overall survival after gastric resection was developed and externally validated in Korea and Japan. However, this gastric cancer nomogram is derived from large-volume centers, and the applicability of the nomogram in smaller centers must be proven. The purpose of this study is to externally validate the gastric cancer nomogram using a dataset from a medium-volume center in Korea.
MATERIALS AND METHODS
We retrospectively analyzed 610 patients who underwent radical gastrectomy for gastric cancer from August 1, 2005 to December 31, 2011. Age, sex, number of metastatic lymph nodes (LNs), number of examined LNs, depth of invasion, and location of the tumor were investigated as variables for validation of the nomogram. Both discrimination and calibration of the nomogram were evaluated.
RESULTS
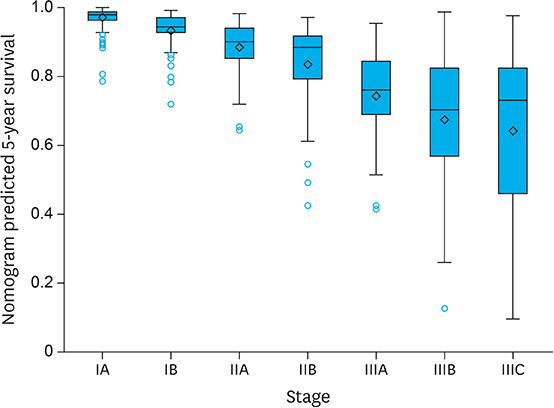
The discrimination was evaluated using Harrell's C-index. The Harrell's C-index was 0.83 and the discrimination of the gastric cancer nomogram was appropriate. Regarding calibration, the 95% confidence interval of predicted survival appeared to be on the ideal reference line except in the poorest survival group. However, we observed a tendency for actual survival to be constantly higher than predicted survival in this cohort.
CONCLUSIONS
Although the discrimination power was good, actual survival was slightly higher than that predicted by the nomogram. This phenomenon might be explained by elongated life span in the recent patient cohort due to advances in adjuvant chemotherapy and improved nutritional status. Future gastric cancer nomograms should consider elongated life span with the passage of time.
Keyword
MeSH Terms
Figure
Reference
-
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87–108.2. Jung KW, Won YJ, Oh CM, Kong HJ, Lee DH, Lee KH. Community of Population-Based Regional Cancer Registries. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2014. Cancer Res Treat. 2017; 49:292–305.3. Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A 3rd. AJCC Cancer Staging Manual. 7th ed. New York (NY): Springer;2010.4. Green D, Ponce de Leon S, Leon-Rodriguez E, Sosa-Sanchez R. Adenocarcinoma of the stomach: univariate and multivariate analysis of factors associated with survival. Am J Clin Oncol. 2002; 25:84–89.5. Talamonti MS, Kim SP, Yao KA, Wayne JD, Feinglass J, Bennett CL, et al. Surgical outcomes of patients with gastric carcinoma: the importance of primary tumor location and microvessel invasion. Surgery. 2003; 134:720–727.6. Kunisaki C, Akiyama H, Nomura M, Matsuda G, Otsuka Y, Ono HA, et al. Clinicopathologic characteristics and surgical outcomes of mucinous gastric carcinoma. Ann Surg Oncol. 2006; 13:836–842.7. Kattan MW, Karpeh MS, Mazumdar M, Brennan MF. Postoperative nomogram for disease-specific survival after an R0 resection for gastric carcinoma. J Clin Oncol. 2003; 21:3647–3650.8. Han DS, Suh YS, Kong SH, Lee HJ, Choi Y, Aikou S, et al. Nomogram predicting long-term survival after d2 gastrectomy for gastric cancer. J Clin Oncol. 2012; 30:3834–3840.9. Liu L, Forman S, Barton B. Fitting cox model using PROC PHREG and beyond in SAS. In : SAS Global Forum 2009; 2009 Mar 22??5; Washington, D.C. Cary (NC): SAS Institute Inc;2009. p. Paper 236-2009.10. Harrell FE Jr, Lee KL, Mark DB. Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med. 1996; 15:361–387.11. Zhang BY, Yuan J, Cui ZS, Li ZW, Li XH, Lu YY. Evaluation of the prognostic value of the metastatic lymph node ratio for gastric cancer. Am J Surg. 2014; 207:555–565.12. Kattan MW, Zelefsky MJ, Kupelian PA, Scardino PT, Fuks Z, Leibel SA. Pretreatment nomogram for predicting the outcome of three-dimensional conformal radiotherapy in prostate cancer. J Clin Oncol. 2000; 18:3352–3359.13. Peeters KC, Kattan MW, Hartgrink HH, Kranenbarg EK, Karpeh MS, Brennan MF, et al. Validation of a nomogram for predicting disease-specific survival after an R0 resection for gastric carcinoma. Cancer. 2005; 103:702–707.14. Novotny AR, Schuhmacher C, Busch R, Kattan MW, Brennan MF, Siewert JR. Predicting individual survival after gastric cancer resection: validation of a U.S.-derived nomogram at a single high-volume center in Europe. Ann Surg. 2006; 243:74–81.15. Chen D, Jiang B, Xing J, Liu M, Cui M, Liu Y, et al. Validation of the memorial Sloan-Kettering Cancer Center nomogram to predict disease-specific survival after R0 resection in a Chinese gastric cancer population. PLoS One. 2013; 8:e76041.16. Memon MA, Subramanya MS, Khan S, Hossain MB, Osland E, Memon B. Meta-analysis of D1 versus D2 gastrectomy for gastric adenocarcinoma. Ann Surg. 2011; 253:900–911.17. Songun I, Putter H, Kranenbarg EM, Sasako M, van de Velde CJ. Surgical treatment of gastric cancer: 15-year follow-up results of the randomised nationwide Dutch D1D2 trial. Lancet Oncol. 2010; 11:439–449.18. The Information Committee of the Korean Gastric Cancer Association. 2004 Nationwide gastric cancer report in Korea. J Korean Gastric Cancer Assoc. 2007; 7:47–54.19. D'Angelica M, Gonen M, Brennan MF, Turnbull AD, Bains M, Karpeh MS. Patterns of initial recurrence in completely resected gastric adenocarcinoma. Ann Surg. 2004; 240:808–816.20. Ahn JS, Ryu SW, Kim IH, Sohn SS. Clinicopathological analysis of recurrent gastric cancer after curative resection. J Korean Surg Soc. 2003; 65:210–216.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- External validation of IBTR! 2.0 nomogram for prediction of ipsilateral breast tumor recurrence
- Feasibility of Gastric Cancer Surgery at Low Volume Hospitals
- Verification of a Western Nomogram for Predicting Oncotype DXâ„¢ Recurrence Scores in Korean Patients with Breast Cancer
- Does Hospital Volume Really Affect the Surgical and Oncological Outcomes of Gastric Cancer in Korea?
- Nomogram for predicting overall survival in patients with large (>5 cm) hepatocellular carcinoma based on real-world practice