Korean J Physiol Pharmacol.
2016 Mar;20(2):213-220. 10.4196/kjpp.2016.20.2.213.
Mitochondrial calcium uniporter inhibition attenuates mouse bone marrow-derived mast cell degranulation induced by beta-1,3-glucan
- Affiliations
-
- 1National Research Laboratory for Mitochondrial Signaling, Department of Physiology, Department of Health Sciences and Technology, BK21 Project Team, College of Medicine, Cardiovascular and Metabolic Disease Center, Inje University, Busan 47392, Korea. phyhanj@inje.ac.kr
- 2Department of Integrated Biomedical Science, College of Medicine, Inje University, Busan 47392, Korea.
- KMID: 2388678
- DOI: http://doi.org/10.4196/kjpp.2016.20.2.213
Abstract
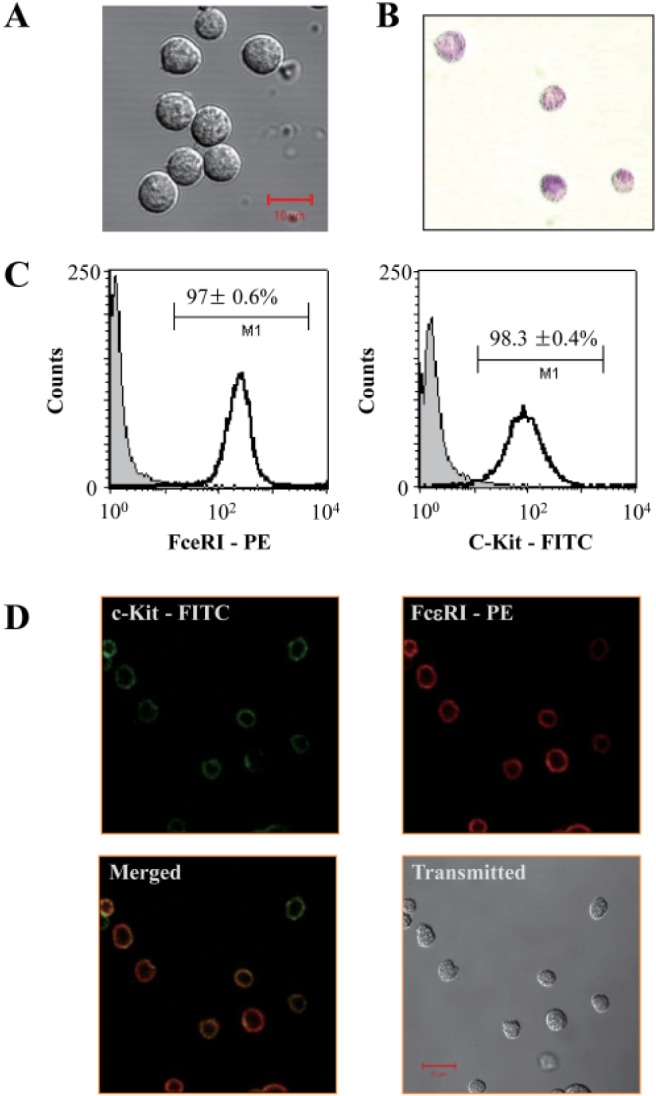
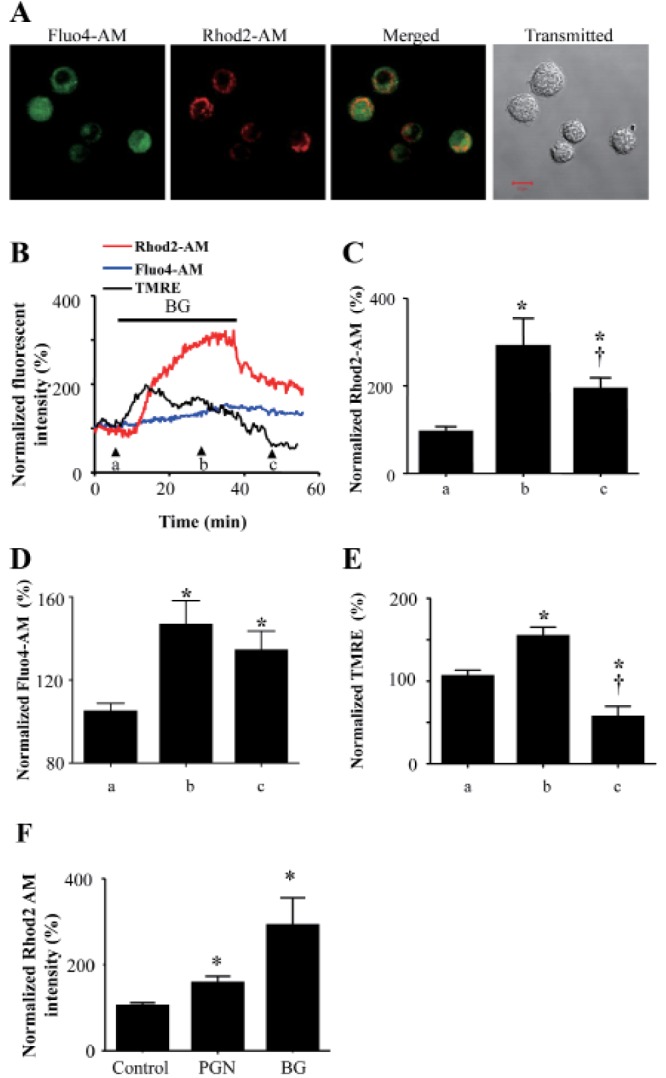
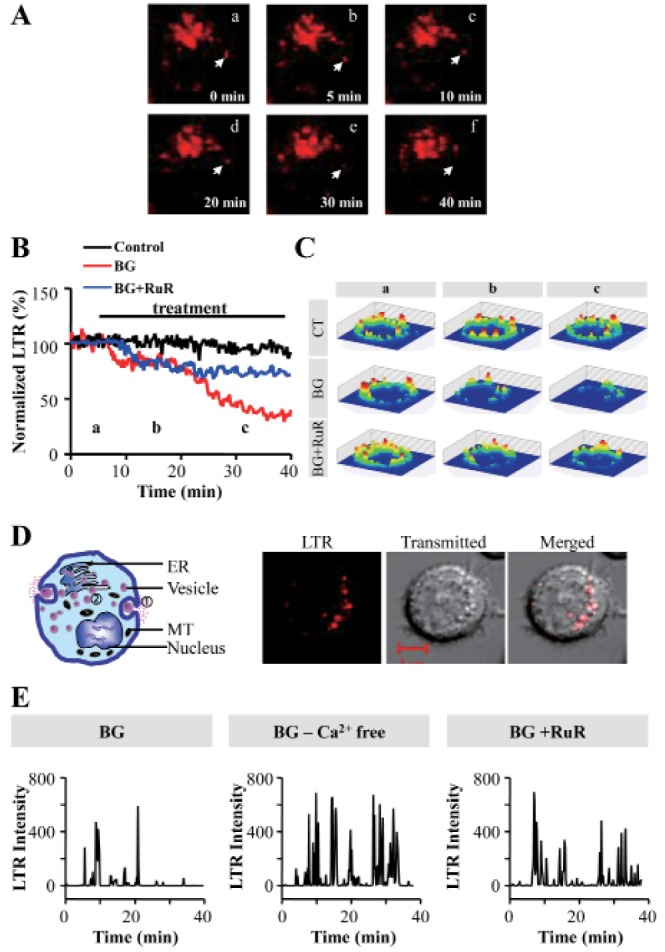
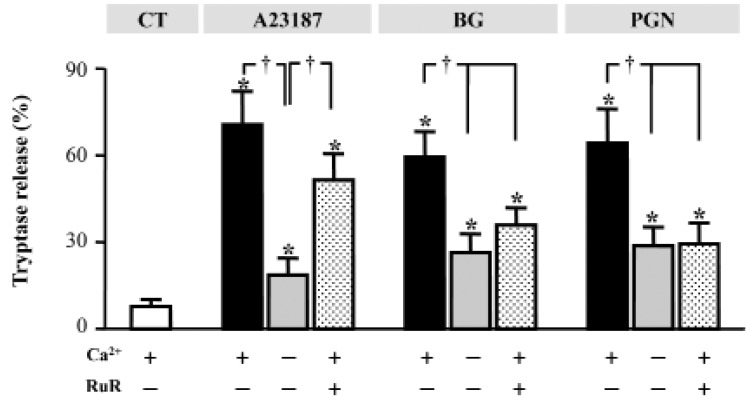
- Mast cells are primary mediators of allergic inflammation. Beta-1,3-glucan (BG) protects against infection and shock by activating immune cells. Activation of the BG receptor induces an increase in intracellular Ca2+, which may induce exocytosis. However, little is known about the precise mechanisms underlying BG activation of immune cells and the possible role of mitochondria in this process. The present study examined whether BG induced mast cell degranulation, and evaluated the role of calcium transients during mast cell activation. Our investigation focused on the role of the mitochondrial calcium uniporter (MCU) in BG-induced degranulation. Black mouse (C57) bone marrow-derived mast cells were stimulated with 0.5 microg/ml BG, 100 microg/ml peptidoglycan (PGN), or 10 microM A23187 (calcium ionophore), and dynamic changes in cytosolic and mitochondrial calcium and membrane potential were monitored. BG-induced mast cell degranulation occurred in a time-dependent manner, and was significantly reduced under calcium-free conditions. Ruthenium red, a mitochondrial Ca2+ uniporter blocker, significantly reduced mast cell degranulation induced by BG, PGN, and A23187. These results suggest that the mitochondrial Ca2+ uniporter has an important regulatory role in BG-induced mast cell degranulation.
MeSH Terms
Figure
Reference
-
1. Metcalfe DD, Baram D, Mekori YA. Mast cells. Physiol Rev. 1997; 77:1033–1079. PMID: 9354811.
Article2. Wedemeyer J, Galli SJ. Mast cells and basophils in acquired immunity. Br Med Bull. 2000; 56:936–955. PMID: 11359630.
Article3. Kulka M, Sheen CH, Tancowny BP, Grammer LC, Schleimer RP. Neuropeptides activate human mast cell degranulation and chemokine production. Immunology. 2008; 123:398–410. PMID: 17922833.
Article4. Roth K, Chen WM, Lin TJ. Positive and negative regulatory mechanisms in high-affinity IgE receptor-mediated mast cell activation. Arch Immunol Ther Exp (Warsz). 2008; 56:385–399. PMID: 19082920.
Article5. Williams D, Mueller A, Browder W. Glucan-based macrophage stimulators. Clin Immunother. 1996; 5:392–399.
Article6. Benacerraf B, Thorbecke GJ, Jacoby D. Effect of zymosan on endotoxin toxicity in mice. Proc Soc Exp Biol Med. 1959; 100:796–799. PMID: 13645722.
Article7. Williams JD, Topley N, Alobaidi HM, Harber MJ. Activation of human polymorphonuclear leucocytes by particulate zymosan is related to both its major carbohydrate components: glucan and mannan. Immunology. 1986; 58:117–124. PMID: 3710519.8. Adams DS, Pero SC, Petro JB, Nathans R, Mackin WM, Wakshull E. PGG-Glucan activates NF-kappaB-like and NF-IL-6-like transcription factor complexes in a murine monocytic cell line. J Leukoc Biol. 1997; 62:865–873. PMID: 9400829.9. Zhang GH, Helmke RJ, Mörk AC, Martinez JR. Regulation of cytosolic free Ca2+ in cultured rat alveolar macrophages (NR8383). J Leukoc Biol. 1997; 62:341–348. PMID: 9307072.10. Mörk AC, Sun X, Liu X, Rodriguez D, Martinez JR, Castro R, Zhang GH. Regulation of (1-3)-beta-glucan-stimulated Ca2+ influx by protein kinase C in NR8383 alveolar macrophages. J Cell Biochem. 2000; 78:131–140. PMID: 10797572.11. Sengeløv H, Kjeldsen L, Borregaard N. Control of exocytosis in early neutrophil activation. J Immunol. 1993; 150:1535–1543. PMID: 8381838.12. Mohr FC, Fewtrell C. The effect of mitochondrial inhibitors on calcium homeostasis in tumor mast cells. Am J Physiol. 1990; 258:C217–C226. PMID: 2137675.
Article13. Suzuki Y, Yoshimaru T, Inoue T, Ra C. Mitochondrial Ca2+ flux is a critical determinant of the Ca2+ dependence of mast cell degranulation. J Leukoc Biol. 2006; 79:508–518. PMID: 16365155.14. Suzuki Y, Yoshimaru T, Inoue T, Nunomura S, Ra C. The high-affinity immunoglobulin E receptor (FcepsilonRI) regulates mitochondrial calcium uptake and a dihydropyridine receptor-mediated calcium influx in mast cells: Role of the FcepsilonRIbeta chain immunoreceptor tyrosine-based activation motif. Biochem Pharmacol. 2008; 75:1492–1503. PMID: 18243160.15. Ma HT, Beaven MA. Regulation of Ca2+ signaling with particular focus on mast cells. Crit Rev Immunol. 2009; 29:155–186. PMID: 19496745.16. Inoue T, Suzuki Y, Ra C. Epigallocatechin-3-gallate inhibits mast cell degranulation, leukotriene C4 secretion, and calcium influx via mitochondrial calcium dysfunction. Free Radic Biol Med. 2010; 49:632–640. PMID: 20510351.
Article17. Furuno T, Shinkai N, Inoh Y, Nakanishi M. Impaired expression of the mitochondrial calcium uniporter suppresses mast cell degranulation. Mol Cell Biochem. 2015; 410:215–221. PMID: 26350567.
Article18. Gurish MF, Friend DS, Webster M, Ghildyal N, Nicodemus CF, Stevens RL. Mouse mast cells that possess segmented/multi-lobular nuclei. Blood. 1997; 90:382–390. PMID: 9207474.
Article19. Akramiene D, Kondrotas A, Didziapetriene J, Kevelaitis E. Effects of beta-glucans on the immune system. Medicina (Kaunas). 2007; 43:597–606. PMID: 17895634.20. Supajatura V, Ushio H, Nakao A, Akira S, Okumura K, Ra C, Ogawa H. Differential responses of mast cell Toll-like receptors 2 and 4 in allergy and innate immunity. J Clin Invest. 2002; 109:1351–1359. PMID: 12021251.21. Hosokawa J, Suzuki K, Nakagomi D, Tamachi T, Takatori H, Suto A, Nakajima H. Role of calcium ionophore A23187-induced activation of IkappaB kinase 2 in mast cells. Int Arch Allergy Immunol. 2013; 161(Suppl 2):37–43.
Article22. Mizuno K, Tolmachova T, Ushakov DS, Romao M, Abrink M, Ferenczi MA, Raposo G, Seabra MC. Rab27b regulates mast cell granule dynamics and secretion. Traffic. 2007; 8:883–892. PMID: 17587407.
Article23. Slamenová D, Lábaj J, Krizková L, Kogan G, Sandula J, Bresgen N, Eckl P. Protective effects of fungal (1-->3)-beta-D-glucan derivatives against oxidative DNA lesions in V79 hamster lung cells. Cancer Lett. 2003; 198:153–160. PMID: 12957353.24. Krizková L, Duracková Z, Sandula J, Slamenová D, Sasinková V, Sivonová M, Krajcovic J. Fungal beta-(1-3)-D-glucan derivatives exhibit high antioxidative and antimutagenic activity in vitro. Anticancer Res. 2003; 23:2751–2756. PMID: 12894570.25. Nasrollahi Z, Mohammadi SR, Mollarazi E, Yadegari MH, Hassan ZM, Talaei F, Dinarvand R, Akbari H, Atyabi F. Functionalized nanoscale β-1,3-glucan to improve Her2+ breast cancer therapy: In vitro and in vivo study. J Control Release. 2015; 202:49–56. PMID: 25597638.26. Zanoni I, Granucci F. Regulation and dysregulation of innate immunity by NFAT signaling downstream of pattern recognition receptors (PRRs). Eur J Immunol. 2012; 42:1924–1931. PMID: 22706795.
Article27. Sakurai A, Yamaguchi N, Sonoyama K. Cell Wall Polysaccharides of Candida albicans Induce Mast Cell Degranulation in the Gut. Biosci Microbiota Food Health. 2012; 31:67–70. PMID: 24936351.28. Kimura Y, Chihara K, Honjoh C, Takeuchi K, Yamauchi S, Yoshiki H, Fujieda S, Sada K. Dectin-1-mediated signaling leads to characteristic gene expressions and cytokine secretion via spleen tyrosine kinase (Syk) in rat mast cells. J Biol Chem. 2014; 289:31565–31575. PMID: 25246527.
Article29. Cleary JA, Kelly GE, Husband AJ. The effect of molecular weight and beta-1,6-linkages on priming of macrophage function in mice by (1,3)-beta-D-glucan. Immunol Cell Biol. 1999; 77:395–403. PMID: 10540205.30. Kolaczkowska E, Seljelid R, Plytycz B. Role of mast cells in zymosan-induced peritoneal inflammation in Balb/c and mast cell-deficient WBB6F1 mice. J Leukoc Biol. 2001; 69:33–42. PMID: 11200065.31. Marshall JS, Stead RH, McSharry C, Nielsen L, Bienenstock J. The role of mast cell degranulation products in mast cell hyperplasia .I. Mechanism of action of nerve growth factor. J Immunol. 1990; 144:1886–1892. PMID: 2106555.32. Dvorak AM, Galli SJ, Schulman ES, Lichtenstein LM, Dvorak HF. Basophil and mast cell degranulation: ultrastructural analysis of mechanisms of mediator release. Fed Proc. 1983; 42:2510–2515. PMID: 6840300.33. Nishida K, Yamasaki S, Ito Y, Kabu K, Hattori K, Tezuka T, Nishizumi H, Kitamura D, Goitsuka R, Geha RS, Yamamoto T, Yagi T, Hirano T. Fc{epsilon}RI-mediated mast cell degranulation requires calcium-independent microtubule-dependent translocation of granules to the plasma membrane. J Cell Biol. 2005; 170:115–126. PMID: 15998803.34. Brown GD, Taylor PR, Reid DM, Willment JA, Williams DL, Martinez-Pomares L, Wong SY, Gordon S. Dectin-1 is a major beta-glucan receptor on macrophages. J Exp Med. 2002; 196:407–412. PMID: 12163569.35. Weatherly LM, Shim J, Hashmi HN, Kennedy RH, Hess ST, Gosse JA. Antimicrobial agent triclosan is a proton ionophore uncoupler of mitochondria in living rat and human mast cells and in primary human keratinocytes. J Appl Toxicol. 2015; DOI: 10.1002/jat.3209. [Epub ahead of print].
Article36. Yoshimaru T, Suzuki Y, Inoue T, Nishida S, Ra C. Extracellular superoxide released from mitochondria mediates mast cell death by advanced glycation end products. Biochim Biophys Acta. 2008; 1783:2332–2343. PMID: 18822320.
Article37. Inoue T, Suzuki Y, Mizuno K, Nakata K, Yoshimaru T, Ra C. SHP-1 exhibits a pro-apoptotic function in antigen-stimulated mast cells: positive regulation of mitochondrial death pathways and negative regulation of survival signaling pathways. Mol Immunol. 2009; 47:222–232. PMID: 19875169.
Article38. Suzuki Y, Yoshimaru T, Inoue T, Ra C. Ca v 1.2 L-type Ca2+ channel protects mast cells against activation-induced cell death by preventing mitochondrial integrity disruption. Mol Immunol. 2009; 46:2370–2380. PMID: 19447492.39. Takekawa M, Furuno T, Hirashima N, Nakanishi M. Mitochondria take up Ca2+ in two steps dependently on store-operated Ca2+ entry in mast cells. Biol Pharm Bull. 2012; 35:1354–1360. PMID: 22863937.
Article40. Erlich TH, Yagil Z, Kay G, Peretz A, Migalovich-Sheikhet H, Tshori S, Nechushtan H, Levi-Schaffer F, Saada A, Razin E. Mitochondrial STAT3 plays a major role in IgE-antigen-mediated mast cell exocytosis. J Allergy Clin Immunol. 2014; 134:460–469. PMID: 24582310.
Article41. Zhang B, Weng Z, Sismanopoulos N, Asadi S, Therianou A, Alysandratos KD, Angelidou A, Shirihai O, Theoharides TC. Mitochondria distinguish granule-stored from de novo synthesized tumor necrosis factor secretion in human mast cells. Int Arch Allergy Immunol. 2012; 159:23–32. PMID: 22555146.
Article42. Sadroddiny E, Ai J, Carroll K, Pham TK, Wright P, Pathak A, Helm B. Protein profiling of the secretome of FcεRI activated RBL-2H3.1 cells. Iran J Immunol. 2012; 9:1–31. PMID: 22426164.43. Ali H, Maeyama K, Sagi-Eisenberg R, Beaven MA. Antigen and thapsigargin promote influx of Ca2+ in rat basophilic RBL-2H3 cells by ostensibly similar mechanisms that allow filling of inositol 1,4,5-trisphosphate-sensitive and mitochondrial Ca2+ stores. Biochem J. 1994; 304:431–440. PMID: 7998977.44. Dvorak AM, Galli SJ, Morgan E, Galli AS, Hammond ME, Dvorak HF. Anaphylactic degranulation of guinea pig basophilic leukocytes. I. Fusion of granule membranes and cytoplasmic vesicles formation and resolution of degranulation sacs. Lab Invest. 1981; 44:174–191. PMID: 6162057.45. Zhang B, Alysandratos KD, Angelidou A, Asadi S, Sismanopoulos N, Delivanis DA, Weng Z, Miniati A, Vasiadi M, Katsarou-Katsari A, Miao B, Leeman SE, Kalogeromitros D, Theoharides TC. Human mast cell degranulation and preformed TNF secretion require mitochondrial translocation to exocytosis sites: relevance to atopic dermatitis. J Allergy Clin Immunol. 2011; 127:1522–1531.e8. PMID: 21453958.
Article46. Zhang B, Asadi S, Weng Z, Sismanopoulos N, Theoharides TC. Stimulated human mast cells secrete mitochondrial components that have autocrine and paracrine inflammatory actions. PLoS One. 2012; 7:e49767. PMID: 23284625.
Article47. Wang X, Schwarz TL. The mechanism of Ca2+-dependent regulation of kinesin-mediated mitochondrial motility. Cell. 2009; 136:163–174. PMID: 19135897.48. Chang KT, Niescier RF, Min KT. Mitochondrial matrix Ca2+ as an intrinsic signal regulating mitochondrial motility in axons. Proc Natl Acad Sci U S A. 2011; 108:15456–15461. PMID: 21876166.49. Niescier RF, Chang KT, Min KT. Miro, MCU, and calcium: bridging our understanding of mitochondrial movement in axons. Front Cell Neurosci. 2013; 7:148. PMID: 24058334.
Article50. Vetvicka V, Vetvickova J. β(1-3)-D-glucan affects adipogenesis, wound healing and inflammation. Orient Pharm Exp Med. 2011; 11:169–175.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Maturation of bone marrow-derived dendritic cells by a novel beta-glucan purified from Paenibacillus polymyxa JB115
- Inhibitory Effect of Cortex mori on the Colchicine-induced Mast Cell Activation
- Atrial natriuretic peptide induces rat peritoneal mast cell activation by cGMP-independent and calcium uptake-dependent mechanism
- Inhibitory effect of Cortex mori on the substance P-induced mast cell degranulation, granular leukocyte infiltraion, and vascular permeability in vivo
- Imperatorin Suppresses Degranulation and Eicosanoid Generation in Activated Bone Marrow-Derived Mast Cells