J Periodontal Implant Sci.
2017 Aug;47(4):219-230. 10.5051/jpis.2017.47.4.219.
Comparison of periodontitis-associated oral biofilm formation under dynamic and static conditions
- Affiliations
-
- 1Department of Periodontology and Research Institute of Oral Sciences, Gangneung-Wonju National University College of Dentistry, Gangneung, Korea. dentist@gwnu.ac.kr
- 2Department of Microbiology and Immunology and Research Institute of Oral Sciences, Gangneung-Wonju National University College of Dentistry, Gangneung, Korea.
- KMID: 2388109
- DOI: http://doi.org/10.5051/jpis.2017.47.4.219
Abstract
- PURPOSE
The purpose of this study was to compare the characteristics of single- and dual-species in vitro oral biofilms made by static and dynamic methods.
METHODS
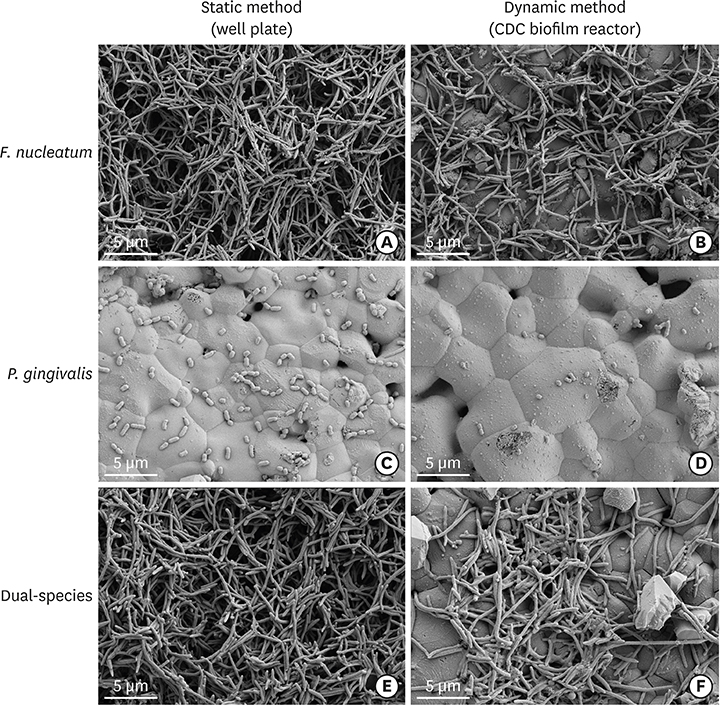
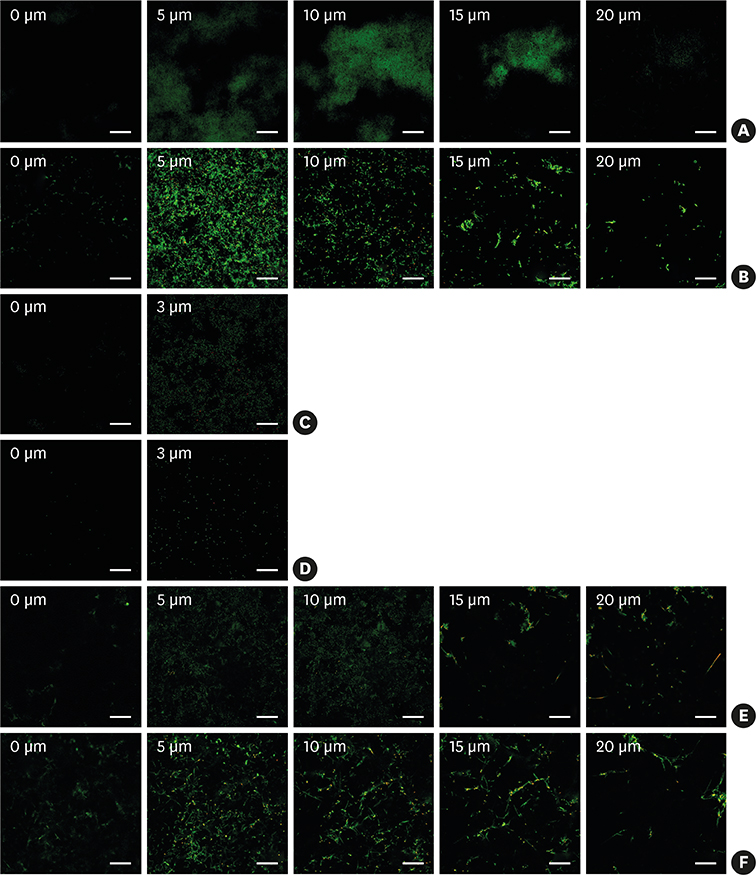
Hydroxyapatite (HA) disks, 12.7 mm in diameter and 3 mm thick, were coated with processed saliva for 4 hours. The disks were divided into a static method group and a dynamic method group. The disks treated with a static method were cultured in 12-well plates, and the disks in the dynamic method group were cultured in a Center for Disease Control and Prevention (CDC) biofilm reactor for 72 hours. In the single- and dual-species biofilms, Fusobacterium nucleatum and Porphyromonas gingivalis were used, and the amount of adhering bacteria, proportions of species, and bacterial reduction of chlorhexidine were examined. Bacterial adhesion was examined with scanning electron microscopy (SEM) and confocal laser scanning microscopy (CLSM).
RESULTS
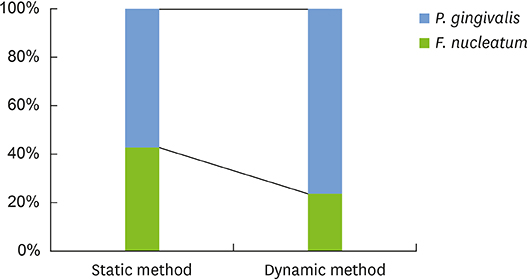
Compared with the biofilms made using the static method, the biofilms made using the dynamic method had significantly lower amounts of adhering and looser bacterial accumulation in SEM and CLSM images. The proportion of P. gingivalis was higher in the dynamic method group than in the static method group; however, the difference was not statistically significant. Furthermore, the biofilm thickness and bacterial reduction by chlorhexidine showed no significant differences between the 2 methods.
CONCLUSIONS
When used to reproduce periodontal biofilms composed of F. nucleatum and P. gingivalis, the dynamic method (CDC biofilm reactor) formed looser biofilms containing fewer bacteria than the well plate. However, this difference did not influence the thickness of the biofilms or the activity of chlorhexidine. Therefore, both methods are useful for mimicking periodontitis-associated oral biofilms.
MeSH Terms
-
Bacteria
Bacterial Adhesion
Biofilms*
Centers for Disease Control and Prevention (U.S.)
Chlorhexidine
Durapatite
Electron Microscope Tomography
Fusobacterium nucleatum
In Vitro Techniques
Methods
Microscopy, Confocal
Microscopy, Electron, Scanning
Periodontitis
Porphyromonas gingivalis
Saliva
Chlorhexidine
Durapatite
Figure
Reference
-
1. Costerton JW, Stewart PS, Greenberg EP. Bacterial biofilms: a common cause of persistent infections. Science. 1999; 284:1318–1322.
Article2. Hall-Stoodley L, Costerton JW, Stoodley P. Bacterial biofilms: from the natural environment to infectious diseases. Nat Rev Microbiol. 2004; 2:95–108.
Article3. Filoche S, Wong L, Sissons CH. Oral biofilms: emerging concepts in microbial ecology. J Dent Res. 2010; 89:8–18.
Article4. Kolenbrander PE, Andersen RN, Blehert DS, Egland PG, Foster JS, Palmer RJ Jr. Communication among oral bacteria. Microbiol Mol Biol Rev. 2002; 66:486–505.
Article5. Ximénez-Fyvie LA, Haffajee AD, Socransky SS. Comparison of the microbiota of supra- and subgingival plaque in health and periodontitis. J Clin Periodontol. 2000; 27:648–657.
Article6. Paster BJ, Boches SK, Galvin JL, Ericson RE, Lau CN, Levanos VA, et al. Bacterial diversity in human subgingival plaque. J Bacteriol. 2001; 183:3770–3783.
Article7. Blanc V, Isabal S, Sánchez MC, Llama-Palacios A, Herrera D, Sanz M, et al. Characterization and application of a flow system for in vitro multispecies oral biofilm formation. J Periodontal Res. 2014; 49:323–332.
Article8. Park JH, Lee JK, Um HS, Chang BS, Lee SY. A periodontitis-associated multispecies model of an oral biofilm. J Periodontal Implant Sci. 2014; 44:79–84.
Article9. Walker C, Sedlacek MJ. An in vitro biofilm model of subgingival plaque. Oral Microbiol Immunol. 2007; 22:152–161.10. Guggenheim B, Giertsen E, Schüpbach P, Shapiro S. Validation of an in vitro biofilm model of supragingival plaque. J Dent Res. 2001; 80:363–370.
Article11. Thomas WE, Trintchina E, Forero M, Vogel V, Sokurenko EV. Bacterial adhesion to target cells enhanced by shear force. Cell. 2002; 109:913–923.
Article12. Ding AM, Palmer RJ Jr, Cisar JO, Kolenbrander PE. Shear-enhanced oral microbial adhesion. Appl Environ Microbiol. 2010; 76:1294–1297.
Article13. Goeres DM, Loetterle LR, Hamilton MA, Murga R, Kirby DW, Donlan RM. Statistical assessment of a laboratory method for growing biofilms. Microbiology. 2005; 151:757–762.
Article14. Rudney JD, Chen R, Lenton P, Li J, Li Y, Jones RS, et al. A reproducible oral microcosm biofilm model for testing dental materials. J Appl Microbiol. 2012; 113:1540–1553.
Article15. Saunders KA, Greenman J. The formation of mixed culture biofilms of oral species along a gradient of shear stress. J Appl Microbiol. 2000; 89:564–572.
Article16. Haffajee AD, Socransky SS. Microbial etiological agents of destructive periodontal diseases. Periodontol 2000. 1994; 5:78–111.
Article17. Okuda T, Kokubu E, Kawana T, Saito A, Okuda K, Ishihara K. Synergy in biofilm formation between Fusobacterium nucleatum and Prevotella species. Anaerobe. 2012; 18:110–116.
Article18. Fonseca AP, Sousa JC. Effect of shear stress on growth, adhesion and biofilm formation of Pseudomonas aeruginosa with antibiotic-induced morphological changes. Int J Antimicrob Agents. 2007; 30:236–241.
Article19. Cotter JJ, O'Gara JP, Stewart PS, Pitts B, Casey E. Characterization of a modified rotating disk reactor for the cultivation of Staphylococcus epidermidis biofilm. J Appl Microbiol. 2010; 109:2105–2117.
Article20. Saito Y, Fujii R, Nakagawa KI, Kuramitsu HK, Okuda K, Ishihara K. Stimulation of Fusobacterium nucleatum biofilm formation by Porphyromonas gingivalis . Oral Microbiol Immunol. 2008; 23:1–6.
Article21. Abiko Y, Sato T, Mayanagi G, Takahashi N. Profiling of subgingival plaque biofilm microflora from periodontally healthy subjects and from subjects with periodontitis using quantitative real-time PCR. J Periodontal Res. 2010; 45:389–395.
Article22. Loozen G, Ozcelik O, Boon N, De Mol A, Schoen C, Quirynen M, et al. Inter-bacterial correlations in subgingival biofilms: a large-scale survey. J Clin Periodontol. 2014; 41:1–10.
Article23. Shaddox LM, Alfant B, Tobler J, Walker C. Perpetuation of subgingival biofilms in an in vitro model. Mol Oral Microbiol. 2010; 25:81–87.24. Fraud S, Maillard JY, Kaminski MA, Hanlon GW. Activity of amine oxide against biofilms of Streptococcus mutans: a potential biocide for oral care formulations. J Antimicrob Chemother. 2005; 56:672–677.25. Hope CK, Petrie A, Wilson M. In vitro assessment of the plaque-removing ability of hydrodynamic shear forces produced beyond the bristles by 2 electric toothbrushes. J Periodontol. 2003; 74:1017–1022.
Article26. Thurnheer T, Gmür R, Guggenheim B. Multiplex FISH analysis of a six-species bacterial biofilm. J Microbiol Methods. 2004; 56:37–47.27. Pratten J, Wills K, Barnett P, Wilson M. In vitro studies of the effect of antiseptic-containing mouthwashes on the formation and viability of Streptococcus sanguis biofilms. J Appl Microbiol. 1998; 84:1149–1155.
Article28. Dû LD, Kolenbrander PE. Identification of saliva-regulated genes of Streptococcus gordonii DL1 by differential display using random arbitrarily primed PCR. Infect Immun. 2000; 68:4834–4837.
Article29. Ahn SJ, Kho HS, Lee SW, Nahm DS. Roles of salivary proteins in the adherence of oral streptococci to various orthodontic brackets. J Dent Res. 2002; 81:411–415.30. Morris EJ, McBride BC. Adherence of Streptococcus sanguis to saliva-coated hydroxyapatite: evidence for two binding sites. Infect Immun. 1984; 43:656–663.31. Lamont RJ, El-Sabaeny A, Park Y, Cook GS, Costerton JW, Demuth DR. Role of the Streptococcus gordonii SspB protein in the development of Porphyromonas gingivalis biofilms on streptococcal substrates. Microbiology. 2002; 148:1627–1636.
Article32. Maeda K, Nagata H, Yamamoto Y, Tanaka M, Tanaka J, Minamino N, et al. Glyceraldehyde-3-phosphate dehydrogenase of Streptococcus oralis functions as a coadhesin for Porphyromonas gingivalis major fimbriae. Infect Immun. 2004; 72:1341–1348.
Article33. Kaplan CW, Lux R, Haake SK, Shi W. The Fusobacterium nucleatum outer membrane protein RadD is an arginine-inhibitable adhesin required for inter-species adherence and the structured architecture of multispecies biofilm. Mol Microbiol. 2009; 71:35–47.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Quorum Sensing Regulation of Biofilm Formation by Periodontal Pathogens
- Inhibition of biofilm formation of periodontal pathogens by D-Arabinose
- Comparison of biofilm on titanium and zirconia surfaces: in vivo study
- Inhibitory Effect of Pentose on Biofilm Formation by Oral Bacteria
- Antimicrobial effect of toothbrush with light emitting diode on dental biofilm attached to zirconia surface: an in vitro study