J Lipid Atheroscler.
2017 Jun;6(1):1-7. 10.12997/jla.2017.6.1.1.
Bile Acid Receptor Farnesoid X Receptor: A Novel Therapeutic Target for Metabolic Diseases
- Affiliations
-
- 1Severance Biomedical Science Institute, BK21 PLUS project for Medical Science, Yonsei University College of Medicine, Seoul, Korea. sfang@yuhs.ac
- KMID: 2385838
- DOI: http://doi.org/10.12997/jla.2017.6.1.1
Abstract
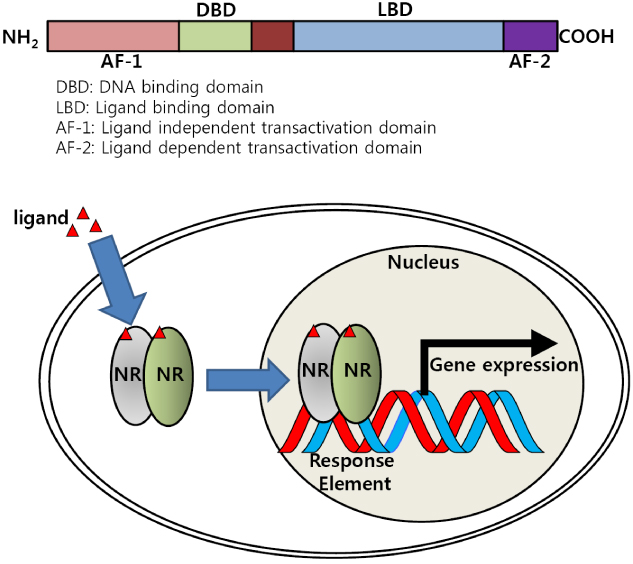
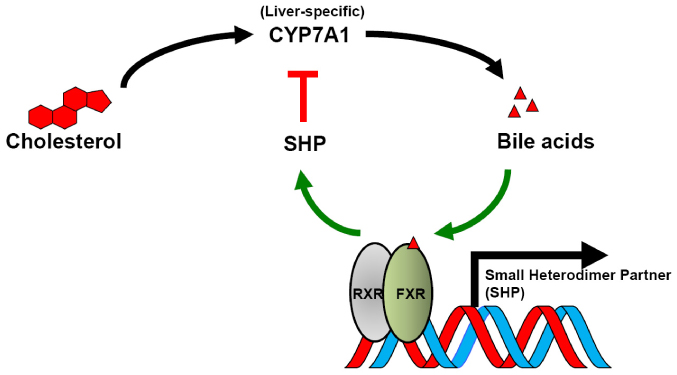
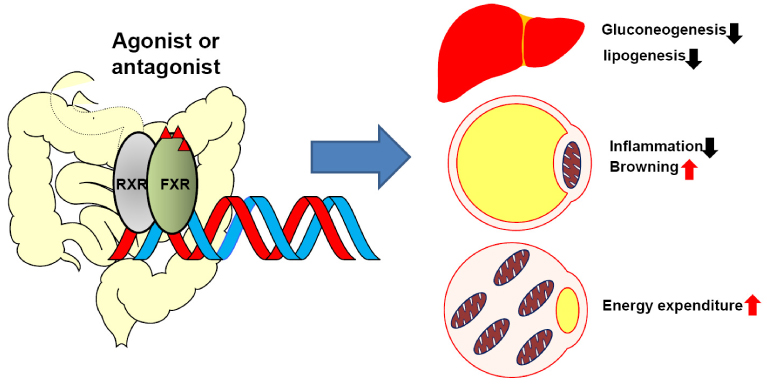
- Bile acid has been well known to serve as a hormone in regulating transcriptional activity of Farnesoid X receptor (FXR), an endogenous bile acid nuclear receptor. Moreover, bile acid regulates diverse biological processes, including cholesterol/bile acid metabolism, glucose/lipid metabolism and energy expenditure. Alteration of bile acid metabolism has been revealed in type II diabetic (T2D) patients. FXR-mediated bile acid signaling has been reported to play key roles in improving metabolic parameters in vertical sleeve gastrectomy surgery, implying that FXR is an essential modulator in the metabolic homeostasis. Using a genetic mouse model, intestinal specific FXR-null mice have been reported to be resistant to diet-induced obesity and insulin resistance. Moreover, intestinal specific FXR agonism using gut-specific FXR synthetic agonist has been shown to enhance thermogenesis in brown adipose tissue and browning in white adipose tissue to increase energy expenditure, leading to reduced body weight gain and improved insulin resistance. Altogether, FXR is a potent therapeutic target for the treatment of metabolic diseases.
Keyword
MeSH Terms
Figure
Reference
-
1. Russell DW. The enzymes, regulation, and genetics of bile acid synthesis. Annu Rev Biochem. 2003; 72:137–174.
Article2. Tu H, Okamoto AY, Shan B. FXR, a bile acid receptor and biological sensor. Trends Cardiovasc Med. 2000; 10:30–35.
Article3. Makishima M, Okamoto AY, Repa JJ, Tu H, Learned RM, Luk A, et al. Identification of a nuclear receptor for bile acids. Science. 1999; 284:1362–1365.
Article4. Forman BM, Goode E, Chen J, Oro AE, Bradley DJ, Perlmann T, et al. Identification of a nuclear receptor that is activated by farnesol metabolites. Cell. 1995; 81:687–693.
Article5. Chiang JY. Bile acid regulation of gene expression: roles of nuclear hormone receptors. Endocr Rev. 2002; 23:443–463.
Article6. Lee FY, Lee H, Hubbert ML, Edwards PA, Zhang Y. FXR, a multipurpose nuclear receptor. Trends Biochem Sci. 2006; 31:572–580.
Article7. Edwards PA, Kast HR, Anisfeld AM. BAREing it all: the adoption of LXR and FXR and their roles in lipid homeostasis. J Lipid Res. 2002; 43:2–12.
Article8. Liu J, Lu H, Lu YF, Lei X, Cui JY, Ellis E, et al. Potency of individual bile acids to regulate bile acid synthesis and transport genes in primary human hepatocyte cultures. Toxicol Sci. 2014; 141:538–546.
Article9. Modica S, Gadaleta RM, Moschetta A. Deciphering the nuclear bile acid receptor FXR paradigm. Nucl Recept Signal. 2010; 8:e005.
Article10. Fujita K, Iguchi Y, Une M, Watanabe S. Ursodeoxycholic acid suppresses lipogenesis in mouse liver: possible role of the decrease in beta-muricholic acid, a farnesoid X receptor antagonist. Lipids. 2017; 52:335–344.
Article11. Jones DE. Obeticholic acid for the treatment of primary biliary cirrhosis. Expert Rev Gastroenterol Hepatol. 2016; 2:1–9.
Article12. Thomas H. Therapy: obeticholic acid for PBC. Nat Rev Gastroenterol Hepatol. 2016; 13:558–559.13. Li T, Chiang JY. Nuclear receptors in bile acid metabolism. Drug Metab Rev. 2013; 45:145–155.
Article14. Lee H, Zhang Y, Lee FY, Nelson SF, Gonzalez FJ, Edwards PA. FXR regulates organic solute transporters alpha and beta in the adrenal gland, kidney, and intestine. J Lipid Res. 2006; 47:201–214.
Article15. Li-Hawkins J, Gåfvels M, Olin M, Lund EG, Andersson U, Schuster G, et al. Cholic acid mediates negative feedback regulation of bile acid synthesis in mice. J Clin Invest. 2002; 110:1191–1200.
Article16. Lu TT, Makishima M, Repa JJ, Schoonjans K, Kerr TA, Auwerx J, et al. Molecular basis for feedback regulation of bile acid synthesis by nuclear receptors. Mol Cell. 2000; 6:507–515.
Article17. Wang L, Lee YK, Bundman D, Han Y, Thevananther S, Kim CS, et al. Redundant pathways for negative feedback regulation of bile acid production. Dev Cell. 2002; 2:721–731.
Article18. Kliewer SA, Mangelsdorf DJ. Bile acids as hormones: the FXR-FGF15/19 pathway. Dig Dis. 2015; 33:327–331.
Article19. Stroeve JH, Brufau G, Stellaard F, Gonzalez FJ, Staels B, Kuipers F. Intestinal FXR-mediated FGF15 production contributes to diurnal control of hepatic bile acid synthesis in mice. Lab Invest. 2010; 90:1457–1467.
Article20. Inagaki T, Choi M, Moschetta A, Peng L, Cummins CL, McDonald JG, et al. Fibroblast growth factor 15 functions as an enterohepatic signal to regulate bile acid homeostasis. Cell Metab. 2005; 2:217–225.
Article21. Song KH, Li T, Owsley E, Strom S, Chiang JY. Bile acids activate fibroblast growth factor 19 signaling in human hepatocytes to inhibit cholesterol 7alpha-hydroxylase gene expression. Hepatology. 2009; 49:297–305.
Article22. Stayrook KR, Bramlett KS, Savkur RS, Ficorilli J, Cook T, Christe ME, et al. Regulation of carbohydrate metabolism by the farnesoid X receptor. Endocrinology. 2005; 146:984–991.
Article23. Ma K, Saha PK, Chan L, Moore DD. Farnesoid X receptor is essential for normal glucose homeostasis. J Clin Invest. 2006; 116:1102–1109.
Article24. Zhang Y, Lee FY, Barrera G, Lee H, Vales C, Gonzalez FJ, et al. Activation of the nuclear receptor FXR improves hyperglycemia and hyperlipidemia in diabetic mice. Proc Natl Acad Sci U S A. 2006; 103:1006–1011.
Article25. Zhang Y, Yin L, Anderson J, Ma H, Gonzalez FJ, Willson TM, et al. Identification of novel pathways that control farnesoid X receptor-mediated hypocholesterolemia. J Biol Chem. 2010; 285:3035–3043.
Article26. Sinal CJ, Tohkin M, Miyata M, Ward JM, Lambert G, Gonzalez FJ. Targeted disruption of the nuclear receptor FXR/BAR impairs bile acid and lipid homeostasis. Cell. 2000; 102:731–744.
Article27. Inagaki T, Moschetta A, Lee YK, Peng L, Zhao G, Downes M, et al. Regulation of antibacterial defense in the small intestine by the nuclear bile acid receptor. Proc Natl Acad Sci U S A. 2006; 103:3920–3925.
Article28. Zhou X, Cao L, Jiang C, Xie Y, Cheng X, Krausz KW, et al. PPARalpha-UGT axis activation represses intestinal FXR-FGF15 feedback signalling and exacerbates experimental colitis. Nat Commun. 2014; 5:4573.
Article29. Modica S, Murzilli S, Salvatore L, Schmidt DR, Moschetta A. Nuclear bile acid receptor FXR protects against intestinal tumorigenesis. Cancer Res. 2008; 68:9589–9594.
Article30. Ryan KK, Tremaroli V, Clemmensen C, Kovatcheva-Datchary P, Myronovych A, Karns R, et al. FXR is a molecular target for the effects of vertical sleeve gastrectomy. Nature. 2014; 509:183–188.
Article31. Fang S, Suh JM, Reilly SM, Yu E, Osborn O, Lackey D, et al. Intestinal FXR agonism promotes adipose tissue browning and reduces obesity and insulin resistance. Nat Med. 2015; 21:159–165.
Article32. Li F, Jiang C, Krausz KW, Li Y, Albert I, Hao H, et al. Microbiome remodelling leads to inhibition of intestinal farnesoid X receptor signalling and decreased obesity. Nat Commun. 2013; 4:2384.
Article33. Watanabe M, Houten SM, Mataki C, Christoffolete MA, Kim BW, Sato H, et al. Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature. 2006; 439:484–489.
Article34. Gerhard GS, Styer AM, Wood GC, Roesch SL, Petrick AT, Gabrielsen J, et al. A role for fibroblast growth factor 19 and bile acids in diabetes remission after Roux-en-Y gastric bypass. Diabetes Care. 2013; 36:1859–1864.
Article35. Kohli R, Bradley D, Setchell KD, Eagon JC, Abumrad N, Klein S. Weight loss induced by Roux-en-Y gastric bypass but not laparoscopic adjustable gastric banding increases circulating bile acids. J Clin Endocrinol Metab. 2013; 98:E708–E712.
Article36. Liu Y, Zhou Y, Wang Y, Geng D, Liu J. Roux-en-Y gastric bypass-induced improvement of glucose tolerance and insulin resistance in type 2 diabetic rats are mediated by glucagon-like peptide-1. Obes Surg. 2011; 21:1424–1431.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Crosstalk between FXR and TGR5 controls glucagon-like peptide 1 secretion to maintain glycemic homeostasis
- Implications for Farnesoid X Receptor Signaling on Bile Acid Metabolism as a Potential Therapeutic Strategy for Nonalcoholic Fatty Liver Disease
- Bile Acid Nuclear Receptor Farnesoid X Receptor: Therapeutic Target for Nonalcoholic Fatty Liver Disease
- Understanding Bile Acid Signaling in Diabetes: From Pathophysiology to Therapeutic Targets
- Bile Acids and the Metabolic Disorders