Yonsei Med J.
2017 Jan;58(1):51-58. 10.3349/ymj.2017.58.1.51.
The Effectiveness of Ferritin as a Contrast Agent for Cell Tracking MRI in Mouse Cancer Models
- Affiliations
-
- 1Department of Medicine, The Graduate School of Yonsei University, Seoul, Korea.
- 2Research Institute & Hospital, National Cancer Center, Goyang, Korea. sensia37@ncc.re.kr dkim@ncc.re.kr
- 3Department of Life Science, Ewha Womans University, Seoul, Korea.
- 4Department of System Cancer Science, Graduate School of Cancer Science & Policy, National Cancer Center, Goyang, Korea.
- 5College of Veterinary Medicine, Konkuk University, Seoul, Korea.
- KMID: 2374188
- DOI: http://doi.org/10.3349/ymj.2017.58.1.51
Abstract
- PURPOSE
We aimed to investigate the effectiveness of ferritin as a contrast agent and a potential reporter gene for tracking tumor cells or macrophages in mouse cancer models.
MATERIALS AND METHODS
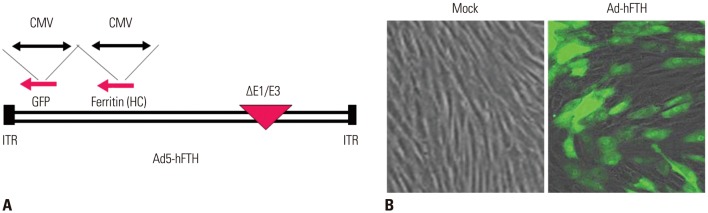
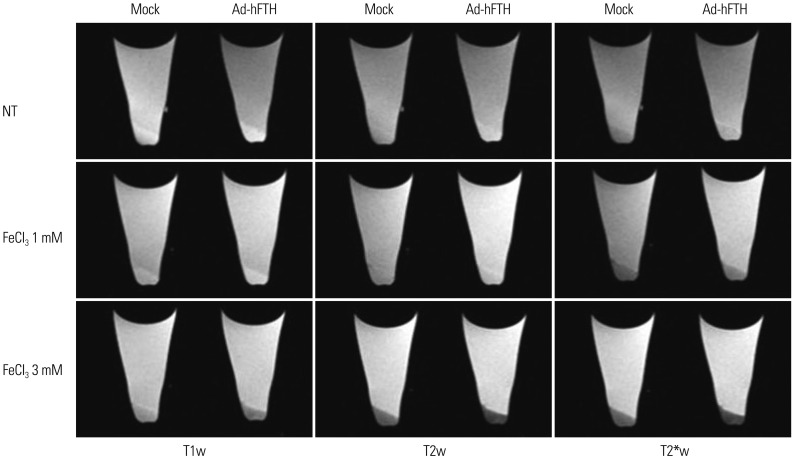
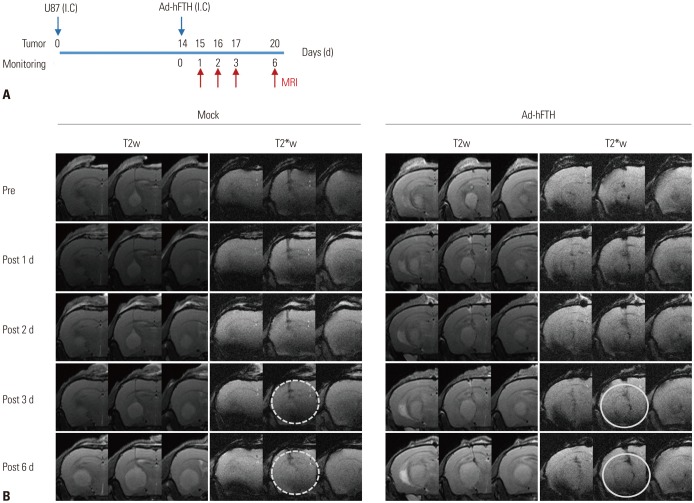
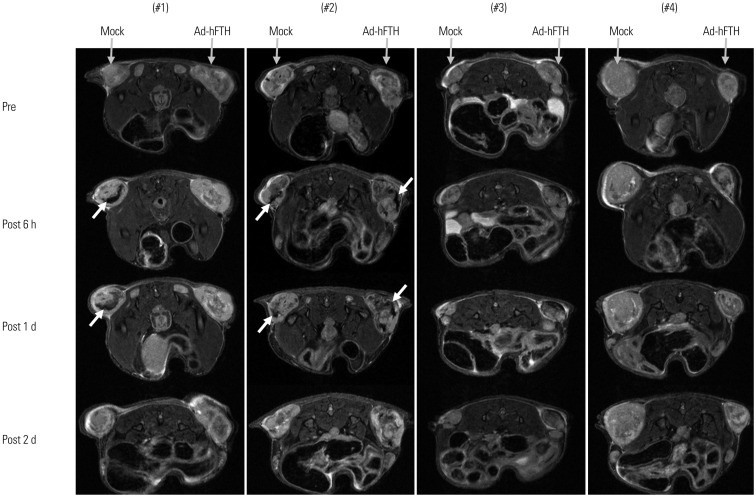
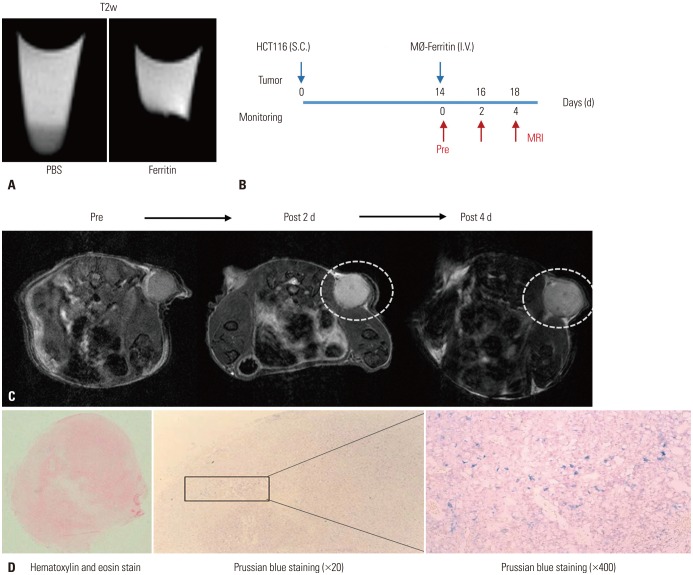
Adenoviral human ferritin heavy chain (Ad-hFTH) was administrated to orthotopic glioma models and subcutaneous colon cancer mouse models using U87MG and HCT116 cells, respectively. Brain MR images were acquired before and daily for up to 6 days after the intracranial injection of Ad-hFTH. In the HCT116 tumor model, MR examinations were performed before and at 6, 24, and 48 h after intratumoral injection of Ad-hFTH, as well as before and every two days after intravenous injection of ferritin-labeled macrophages. The contrast effect of ferritin in vitro was measured by MR imaging of cell pellets. MRI examinations using a 7T MR scanner comprised a T1-weighted (T1w) spin-echo sequence, T2-weighted (T2w) relaxation enhancement sequence, and T2*-weighted (T2*w) fast low angle shot sequence.
RESULTS
Cell pellet imaging of Ad-hFTH in vitro showed a strong negatively enhanced contrast in T2w and T2*w images, presenting with darker signal intensity in high concentrations of Fe. T2w images of glioma and subcutaneous HCT116 tumor models showed a dark signal intensity around or within the Ad-hFTH tumor, which was distinct with time and apparent in T2*w images. After injection of ferritin-labeled macrophages, negative contrast enhancement was identified within the tumor.
CONCLUSION
Ferritin could be a good candidate as an endogenous MR contrast agent and a potential reporter gene that is capable of maintaining cell labeling stability and cellular safety.
Keyword
MeSH Terms
-
Animals
Brain Neoplasms/*diagnostic imaging/pathology
Cell Line, Tumor
Cell Tracking/*methods
Colonic Neoplasms/*diagnostic imaging/pathology
*Contrast Media/administration & dosage
Disease Models, Animal
Female
*Ferritins/administration & dosage
Genes, Reporter
Glioma/*diagnostic imaging/pathology
Humans
Injections, Intravenous
Macrophages
Magnetic Resonance Imaging/*methods
Male
Mice
Neoplasm Transplantation
Skin Neoplasms/*diagnostic imaging/pathology
Time Factors
Contrast Media
Ferritins
Figure
Reference
-
1. Brader P, Serganova I, Blasberg RG. Noninvasive molecular imaging using reporter genes. J Nucl Med. 2013; 54:167–172. PMID: 23318292.
Article2. Youn H, Chung JK. Reporter gene imaging. AJR Am J Roentgenol. 2013; 201:W206–W214. PMID: 23883235.
Article3. Weissleder R, Moore A, Mahmood U, Bhorade R, Benveniste H, Chiocca EA, et al. In vivo magnetic resonance imaging of transgene expression. Nat Med. 2000; 6:351–355. PMID: 10700241.
Article4. Kang HW, Josephson L, Petrovsky A, Weissleder R, Bogdanov A Jr. Magnetic resonance imaging of inducible E-selectin expression in human endothelial cell culture. Bioconjug Chem. 2002; 13:122–127. PMID: 11792187.
Article5. Zhao M, Beauregard DA, Loizou L, Davletov B, Brindle KM. Noninvasive detection of apoptosis using magnetic resonance imaging and a targeted contrast agent. Nat Med. 2001; 7:1241–1244. PMID: 11689890.
Article6. Artemov D. Molecular magnetic resonance imaging with targeted contrast agents. J Cell Biochem. 2003; 90:518–524. PMID: 14523986.
Article7. Huh YM, Jun YW, Song HT, Kim S, Choi JS, Lee JH, et al. In vivo magnetic resonance detection of cancer by using multifunctional magnetic nanocrystals. J Am Chem Soc. 2005; 127:12387–12391. PMID: 16131220.
Article8. Jun YW, Huh YM, Choi JS, Lee JH, Song HT, Kim S, et al. Nanoscale size effect of magnetic nanocrystals and their utilization for cancer diagnosis via magnetic resonance imaging. J Am Chem Soc. 2005; 127:5732–5733. PMID: 15839639.
Article9. Czernin J, Phelps ME. Positron emission tomography scanning: current and future applications. Annu Rev Med. 2002; 53:89–112. PMID: 11818465.
Article10. Graves EE, Weissleder R, Ntziachristos V. Fluorescence molecular imaging of small animal tumor models. Curr Mol Med. 2004; 4:419–430. PMID: 15354872.
Article11. Bulte JW, Kraitchman DL. Iron oxide MR contrast agents for molecular and cellular imaging. NMR Biomed. 2004; 17:484–499. PMID: 15526347.
Article12. Ahn SJ, Koom WS, An CS, Lim JS, Lee SK, Suh JS, et al. Quantitative assessment of tumor responses after radiation therapy in a DLD-1 colon cancer mouse model using serial dynamic contrast-enhanced magnetic resonance imaging. Yonsei Med J. 2012; 53:1147–1153. PMID: 23074115.
Article13. Lee JH, Huh YM, Jun YW, Seo JW, Jang JT, Song HT, et al. Artificially engineered magnetic nanoparticles for ultra-sensitive molecular imaging. Nat Med. 2007; 13:95–99. PMID: 17187073.
Article14. Song HT, Choi JS, Huh YM, Kim S, Jun YW, Suh JS, et al. Surface modulation of magnetic nanocrystals in the development of highly efficient magnetic resonance probes for intracellular labeling. J Am Chem Soc. 2005; 127:9992–9993. PMID: 16011350.
Article15. Pawelczyk E, Arbab AS, Pandit S, Hu E, Frank JA. Expression of transferrin receptor and ferritin following ferumoxides-protamine sulfate labeling of cells: implications for cellular magnetic resonance imaging. NMR Biomed. 2006; 19:581–592. PMID: 16673357.
Article16. Gilad AA, Winnard PT Jr, van Zijl PC, Bulte JW. Developing MR reporter genes: promises and pitfalls. NMR Biomed. 2007; 20:275–290. PMID: 17451181.
Article17. Cohen B, Ziv K, Plaks V, Israely T, Kalchenko V, Harmelin A, et al. MRI detection of transcriptional regulation of gene expression in transgenic mice. Nat Med. 2007; 13:498–503. PMID: 17351627.
Article18. Genove G, DeMarco U, Xu H, Goins WF, Ahrens ET. A new transgene reporter for in vivo magnetic resonance imaging. Nat Med. 2005; 11:450–454. PMID: 15778721.
Article19. Uchida M, Terashima M, Cunningham CH, Suzuki Y, Willits DA, Willis AF, et al. A human ferritin iron oxide nano-composite magnetic resonance contrast agent. Magn Reson Med. 2008; 60:1073–1081. PMID: 18956458.
Article20. Carrondo MA. Ferritins, iron uptake and storage from the bacterioferritin viewpoint. EMBO J. 2003; 22:1959–1968. PMID: 12727864.21. Gossuin Y, Muller RN, Gillis P. Relaxation induced by ferritin: a better understanding for an improved MRI iron quantification. NMR Biomed. 2004; 17:427–432. PMID: 15526352.
Article22. Kim HS, Woo J, Lee JH, Joo HJ, Choi Y, Kim H, et al. In vivo tracking of dendritic cell using MRI reporter gene, ferritin. PLoS One. 2015; 10:e0125291. PMID: 25993535.
Article23. Ghosh SS, Gopinath P, Ramesh A. Adenoviral vectors: a promising tool for gene therapy. Appl Biochem Biotechnol. 2006; 133:9–29. PMID: 16622281.
Article24. D'souza N, Rossignoli F, Golinelli G, Grisendi G, Spano C, Candini O, et al. Mesenchymal stem/stromal cells as a delivery platform in cell and gene therapies. BMC Med. 2015; 13:186. PMID: 26265166.25. Corot C, Robert P, Idée JM, Port M. Recent advances in iron oxide nanocrystal technology for medical imaging. Adv Drug Deliv Rev. 2006; 58:1471–1504. PMID: 17116343.
Article26. Bach-Gansmo T. Ferrimagnetic susceptibility contrast agents. Acta Radiol Suppl. 1993; 387:1–30. PMID: 8390776.27. Wang YX. Superparamagnetic iron oxide based MRI contrast agents: current status of clinical application. Quant Imaging Med Surg. 2011; 1:35–40. PMID: 23256052.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Preclinical Models of Follicular Cell-Derived Thyroid Cancer: An Overview from Cancer Cell Lines to Mouse Models
- Mouse Models of Gastric Carcinogenesis
- In Vivo Cell Tracking with Bioluminescence Imaging
- In vitro MRI and Characterization of Rat Mesenchymal Stem Cells Transduced with Ferritin as MR Reporter Gene
- Use of Abbreviated Magnetic Resonance Imaging in Breast Cancer Screening