Yonsei Med J.
2016 Jul;57(4):928-935. 10.3349/ymj.2016.57.4.928.
Efficacy and Safety of Roflumilast in Korean Patients with COPD
- Affiliations
-
- 1Department of Pulmonary and Critical Care Medicine and Clinical Research Center for Chronic Obstructive Airway Diseases, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. sdlee@amc.seoul.kr
- 2Department of Internal Medicine, Kangwon National University College of Medicine, Chuncheon, Korea.
- KMID: 2374125
- DOI: http://doi.org/10.3349/ymj.2016.57.4.928
Abstract
- PURPOSE
Roflumilast is the only oral phosphodiesterase 4 inhibitor approved to treat chronic obstructive pulmonary disease (COPD) patients [post-bronchodilator forced expiratory volume in 1 second (FEV1) <50% predicted] with chronic bronchitis and a history of frequent exacerbations. This study evaluated the efficacy and safety of roflumilast in Korean patients with COPD and compared the efficacy based on the severity of airflow limitation.
MATERIALS AND METHODS
A post-hoc subgroup analysis was performed in Korean COPD patients participating in JADE, a 12-week, double-blinded, placebo-controlled, parallel-group, phase III trial in Asia. The primary efficacy endpoint was the mean [least-squares mean adjusted for covariates (LSMean)] change in post-bronchodilator FEV1 from baseline to each post-randomization visit. Safety endpoints included adverse events (AEs) and changes in laboratory values, vital signs, and electrocardiograms.
RESULTS
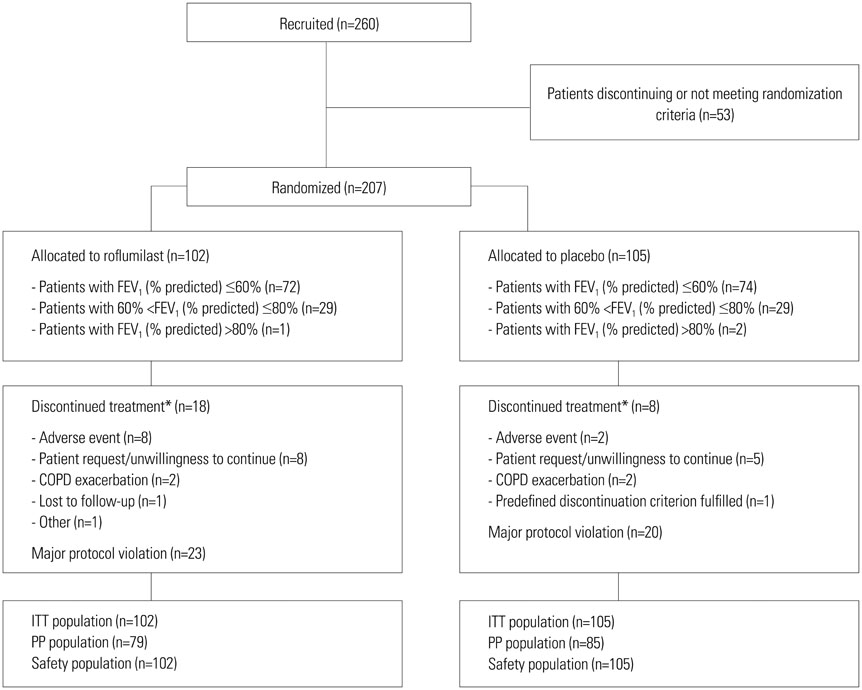
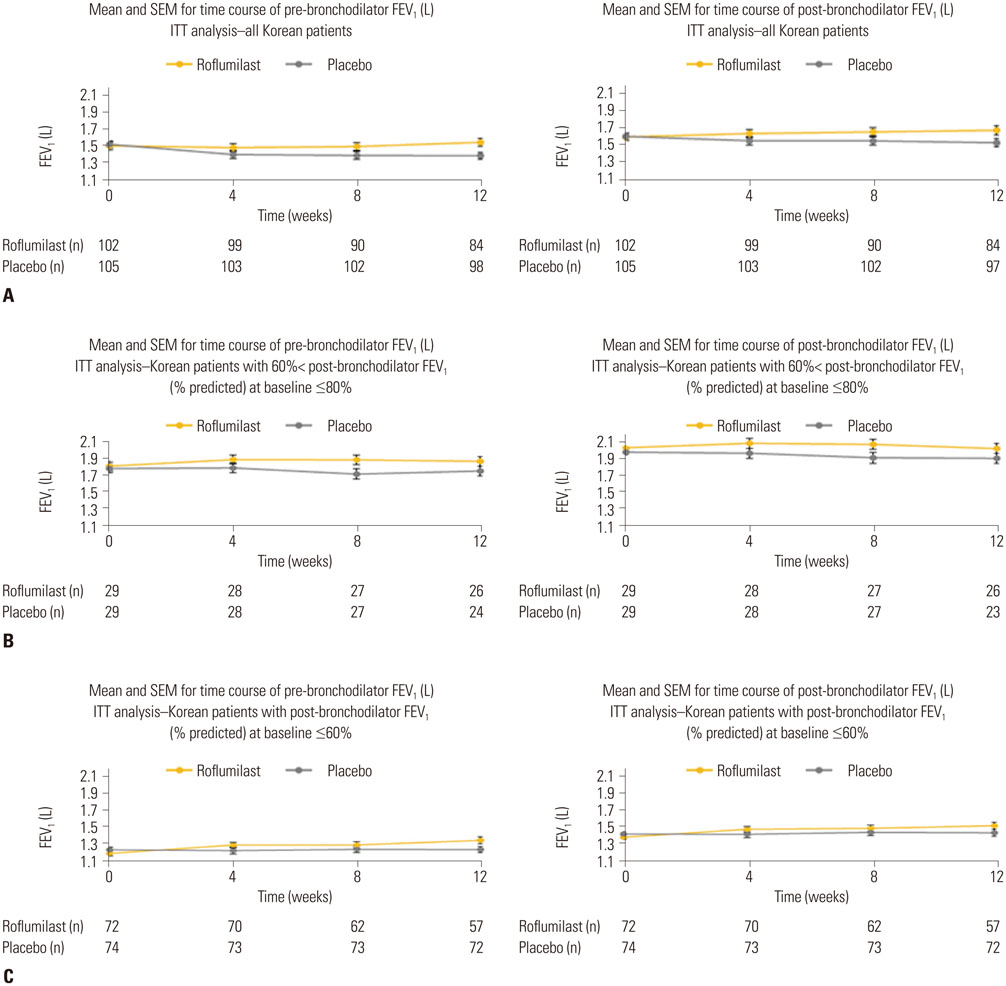
A total of 260 Korean COPD patients were recruited, of which 207 were randomized to roflumilast (n=102) or placebo (n=105) treatment. After 12 weeks, LSMean post-bronchodilator FEV1 increased by 43 mL for patients receiving roflumilast and decreased by 60 mL for those taking placebo. Adverse events were more common in the roflumilast group than in the placebo group; however, the types and frequency of AEs were comparable to those reported in previous studies.
CONCLUSION
Roflumilast significantly improved lung function with a tolerable safety profile in Korean COPD patients irrespective of the severity of airflow limitation.
MeSH Terms
-
Aged
Aminopyridines/*therapeutic use
*Asian Continental Ancestry Group
Benzamides/*therapeutic use
Cyclopropanes/therapeutic use
Double-Blind Method
Female
Humans
Male
Middle Aged
Phosphodiesterase 4 Inhibitors/*therapeutic use
Pulmonary Disease, Chronic Obstructive/*drug therapy/*ethnology/physiopathology
Republic of Korea
Respiratory Function Tests
Treatment Outcome
Aminopyridines
Benzamides
Cyclopropanes
Phosphodiesterase 4 Inhibitors
Figure
Reference
-
1. Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006; 3:e442.
Article2. Lopez AD, Shibuya K, Rao C, Mathers CD, Hansell AL, Held LS, et al. Chronic obstructive pulmonary disease: current burden and future projections. Eur Respir J. 2006; 27:397–412.
Article3. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. updated 2014. accessed on 2015 May. Available at: http://www.goldcopd.org/uploads/users/les/GOLD_Report2014_Feb07.pdf.4. Calverley PM, Anderson JA, Celli B, Ferguson GT, Jenkins C, Jones PW, et al. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. N Engl J Med. 2007; 356:775–789.
Article5. Drummond MB, Dasenbrook EC, Pitz MW, Murphy DJ, Fan E. Inhaled corticosteroids in patients with stable chronic obstructive pulmonary disease: a systematic review and meta-analysis. JAMA. 2008; 300:2407–2416.
Article6. Rabe KF. Update on roflumilast, a phosphodiesterase 4 inhibitor for the treatment of chronic obstructive pulmonary disease. Br J Pharmacol. 2011; 163:53–67.
Article7. Rabe KF, Bateman ED, O'Donnell D, Witte S, Bredenbröker D, Bethke TD. Roflumilast--an oral anti-inflammatory treatment for chronic obstructive pulmonary disease: a randomised controlled trial. Lancet. 2005; 366:563–571.
Article8. Calverley PM, Sanchez-Toril F, McIvor A, Teichmann P, Breden-broeker D, Fabbri LM. Effect of 1-year treatment with roflumilast in severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2007; 176:154–161.
Article9. Calverley PM, Rabe KF, Goehring UM, Kristiansen S, Fabbri LM, Martinez FJ. M2-124 and M2-125 study groups. Roflumilast in symptomatic chronic obstructive pulmonary disease: two randomised clinical trials. Lancet. 2009; 374:685–694.
Article10. Fabbri LM, Calverley PM, Izquierdo-Alonso JL, Bundschuh DS, Brose M, Martinez FJ, et al. Roflumilast in moderate-to-severe chronic obstructive pulmonary disease treated with longacting bronchodilators: two randomised clinical trials. Lancet. 2009; 374:695–703.
Article11. Rennard SI, Calverley PM, Goehring UM, Bredenbröker D, Martinez FJ. Reduction of exacerbations by the PDE4 inhibitor roflumilast--the importance of defining different subsets of patients with COPD. Respir Res. 2011; 12:18.12. Oba Y, Lone NA. Efficacy and safety of roflumilast in patients with chronic obstructive pulmonary disease: a systematic review and meta-analysis. Ther Adv Respir Dis. 2013; 7:13–24.
Article13. Yasuda SU, Zhang L, Huang SM. The role of ethnicity in variability in response to drugs: focus on clinical pharmacology studies. Clin Pharmacol Ther. 2008; 84:417–423.
Article14. Lee SD, Hui DS, Mahayiddin AA, Roa CC Jr, Kwa KH, Goehring UM, et al. Roflumilast in Asian patients with COPD: a randomized placebo-controlled trial. Respirology. 2011; 16:1249–1257.
Article15. Pang JA, Zhang YG, Swaminathan R. The pharmacokinetics and efficacy of slow-release theophylline with asymmetric dosing in asthmatic Chinese. Chest. 1988; 93:785–789.
Article16. Choudhry S, Ung N, Avila PC, Ziv E, Nazario S, Casal J, et al. Pharmacogenetic differences in response to albuterol between Puerto Ricans and Mexicans with asthma. Am J Respir Crit Care Med. 2005; 171:563–570.
Article17. Cazzola M, Matera MG. Safety of long-acting beta2-agonists in the treatment of asthma. Ther Adv Respir Dis. 2007; 1:35–46.
Article18. Zheng J, Yang J, Zhou X, Zhao L, Hui F, Wang H, et al. Roflumilast for the treatment of COPD in an Asian population: a randomized, double-blind, parallel-group study. Chest. 2014; 145:44–52.
Article19. Hwang YI, Park YB, Oh YM, Lee JH, Kim TH, Yoo KH, et al. Comparison of Korean COPD guideline and GOLD initiative report in term of acute exacerbation: a validation study for Korean COPD guideline. J Korean Med Sci. 2014; 29:1108–1112.
Article20. Hanania NA, Calverley PM, Dransfield MT, Karpel JP, Brose M, Zhu H, et al. Pooled subpopulation analyses of the effects of roflumilast on exacerbations and lung function in COPD. Respir Med. 2014; 108:366–375.
Article21. Cazzola M, MacNee W, Martinez FJ, Rabe KF, Franciosi LG, Barnes PJ, et al. Outcomes for COPD pharmacological trials: from lung function to biomarkers. Eur Respir J. 2008; 312:416–469.
Article22. Redelmeier DA, Goldstein RS, Min ST, Hyland RH. Spirometry and dyspnea in patients with COPD. When small differences mean little. Chest. 1996; 109:1163–1168.23. Jones PW, Bosh TK. Quality of life changes in COPD patients treated with salmeterol. Am J Respir Crit Care Med. 1997; 155:1283–1289.
Article24. Martinez FJ, Calverley PM, Goehring UM, Brose M, Fabbri LM, Rabe KF. Effect of roflumilast on exacerbations in patients with severe chronic obstructive pulmonary disease uncontrolled by combination therapy (REACT): a multicentre randomised controlled trial. Lancet. 2015; 385:857–866.
Article25. Oh YM, Bhome AB, Boonsawat W, Gunasekera KD, Madegedara D, Idolor L, et al. Characteristics of stable chronic obstructive pulmonary disease patients in the pulmonology clinics of seven Asian cities. Int J Chron Obstruct Pulmon Dis. 2013; 8:31–39.26. Tatsumi K, Kasahara Y, Kurosu K, Tanabe N, Takiguchi Y, Kuriyama T. Respiratory Failure Research Group in Japan. Clinical phenotypes of COPD: results of a Japanese epidemiological survey. Respirology. 2004; 9:331–336.
Article27. Lee JS, Kim SO, Seo JB, Lee JH, Kim EK, Kim TH, et al. Longitudinal lung volume changes in patients with chronic obstructive pulmonary disease. Lung. 2013; 191:405–412.
Article28. Lee JH, Lee YK, Kim EK, Kim TH, Huh JW, Kim WJ, et al. Responses to inhaled long-acting beta-agonist and corticosteroid according to COPD subtype. Respir Med. 2010; 104:542–549.
Article29. Lee JS, Huh JW, Chae EJ, Seo JB, Ra SW, Lee JH, et al. Predictors of pulmonary function response to treatment with salmeterol/fluticasone in patients with chronic obstructive pulmonary disease. J Korean Med Sci. 2011; 26:379–385.
Article30. Lee JS, Huh JW, Chae EJ, Seo JB, Ra SW, Lee JH, et al. Different therapeutic responses in chronic obstructive pulmonary disease subgroups. Int J Tuberc Lung Dis. 2011; 15:1104–1110.
Article31. Lee JS, Huh JW, Chae EJ, Seo JB, Ra SW, Lee JH, et al. Response patterns to bronchodilator and quantitative computed tomography in chronic obstructive pulmonary disease. Clin Physiol Funct Imaging. 2012; 32:12–18.
Article32. Yoo JW, Hong Y, Seo JB, Chae EJ, Ra SW, Lee JH, et al. Comparison of clinico-physiologic and CT imaging risk factors for COPD exacerbation. J Korean Med Sci. 2011; 26:1606–1612.
Article33. Hurst JR, Vestbo J, Anzueto A, Locantore N, Mllerova H, Tal-Singer R, et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med. 2010; 363:1128–1138.
Article34. Murciano D, Auclair MH, Pariente R, Aubier M. A randomized, controlled trial of theophylline in patients with severe chronic obstructive pulmonary disease. N Engl J Med. 1989; 320:1521–1525.
Article35. Ram FS, Jones PW, Castro AA, De Brito JA, Atallah AN, Lacasse Y, et al. Oral theophylline for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2002; CD003902.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Incidence of Adverse Effects and Discontinuation Rate between Patients Receiving 250 Micrograms and 500 Micrograms of Roflumilast: A Comparative Study
- Role of phosphodiesterase-4 inhibitors in chronic obstructive pulmonary disease
- Efficacy of Roflumilast in Bronchiectasis Patients with Frequent Exacerbations: A Double-Blinded, Randomized, Placebo-Controlled Pilot Clinical Trial
- Effect of a Dose-Escalation Regimen for Improving Adherence to Roflumilast in Patients with Chronic Obstructive Pulmonary Disease
- Interpretation and application of recent large clinical studies in COPD