J Gastric Cancer.
2017 Mar;17(1):43-51. 10.5230/jgc.2017.17.e5.
Analysis of G3BP1 and VEZT Expression in Gastric Cancer and Their Possible Correlation with Tumor Clinicopathological Factors
- Affiliations
-
- 1Department of Biology, East Tehran Branch, Islamic Azad University, Tehran, Iran. elham_moslemi60@yahoo.com
- KMID: 2372581
- DOI: http://doi.org/10.5230/jgc.2017.17.e5
Abstract
- PURPOSE
This study aimed to analyze G3BP1 and VEZT expression profiles in patients with gastric cancer, and examine the possible relationship between the expressions of each gene and clinicopathological factors.
MATERIALS AND METHODS
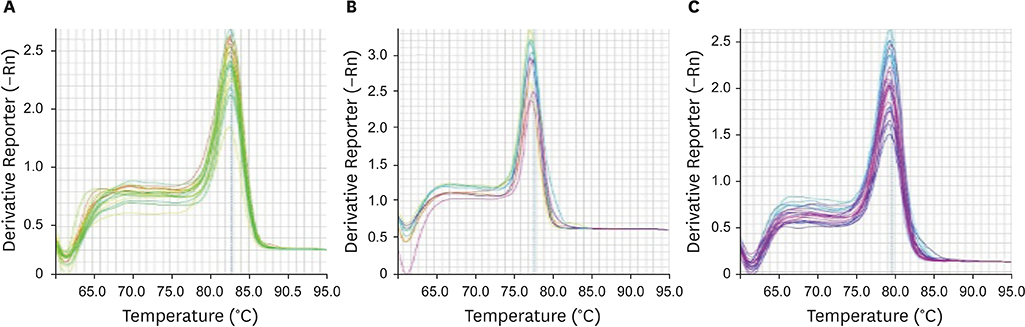
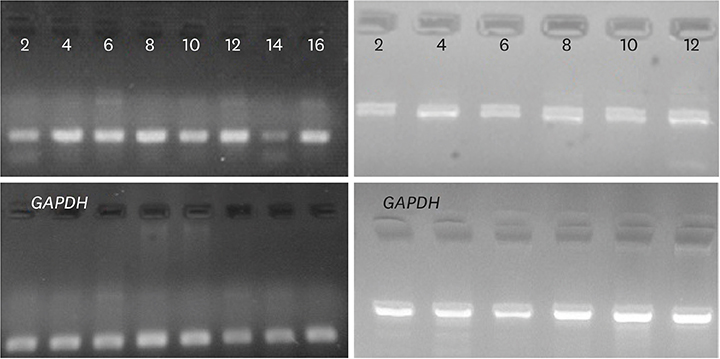
Expression of these genes in formalin-fixed paraffin embedded (FFPE) tissues, collected from 40 patients with gastric cancer and 40 healthy controls, was analyzed. Differences in gene expression among patient and normal samples were identified using the GraphPad Prism 5 software. For the analysis of real-time polymerase chain reaction products, GelQuantNET software was used.
RESULTS
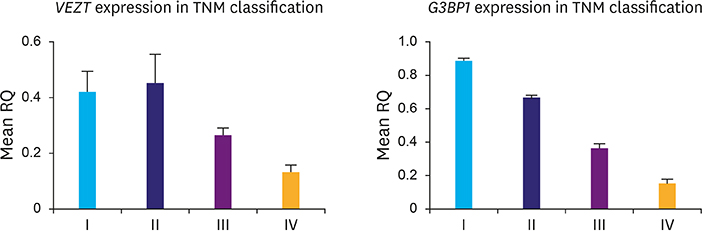
Our findings demonstrated that both VEZT and G3BP1 mRNA expression levels were downregulated in gastric cancer samples compared with those in the normal controls. No significant relationship was found between the expression of these genes and gender (P-value, 0.4835 vs. 0.6350), but there were significant changes associated with age (P-value, 0.0004 vs. 0.0001) and stage of disease (P-value, 0.0019 vs. 0.0001). In addition, there was a direct relationship between VEZT gene expression and metastasis (P-value, 0.0462), in contrast to G3BP1 that did not demonstrate any significant correlation (P-value, 0.1833).
CONCLUSIONS
The results suggest that expression profiling of VEZT and G3BP1 can be used for diagnosis of gastric cancer, and specifically, VEZT gene could be considered as a biomarker for the detection of gastric cancer progression.
Keyword
MeSH Terms
Figure
Reference
-
1. McLean MH, El-Omar EM. Genetics of gastric cancer. Nat Rev Gastroenterol Hepatol. 2014; 11:664–674.2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015; 65:5–29.3. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87–108.4. Lagergren J, Andersson G, Talbäck M, Drefahl S, Bihagen E, Härkönen J, et al. Marital status, education, and income in relation to the risk of esophageal and gastric cancer by histological type and site. Cancer. 2016; 122:207–212.5. Arnold M, Karim-Kos HE, Coebergh JW, Byrnes G, Antilla A, Ferlay J, et al. Recent trends in incidence of five common cancers in 26 European countries since 1988: Analysis of the European Cancer Observatory. Eur J Cancer. 2015; 51:1164–1187.6. Fu DG. Epigenetic alterations in gastric cancer. [Review]. Mol Med Rep. 2015; 12:3223–3230.7. Carcas LP. Gastric cancer review. J Carcinog. 2014; 13:14.8. Jin Z, Jiang W, Wang L. Biomarkers for gastric cancer: progression in early diagnosis and prognosis. [Review]. Oncol Lett. 2015; 9:1502–1508.9. Downward J. Targeting RAS signalling pathways in cancer therapy. Nat Rev Cancer. 2003; 3:11–22.10. Min L, Ruan Y, Shen Z, Jia D, Wang X, Zhao J, et al. Overexpression of Ras-GTPase-activating protein SH3 domain-binding protein 1 correlates with poor prognosis in gastric cancer patients. Histopathology. 2015; 67:677–688.11. Guitard E, Parker F, Millon R, Abecassis J, Tocqué B. G3BP is overexpressed in human tumors and promotes S phase entry. Cancer Lett. 2001; 162:213–221.12. Taniuchi K, Nishimori I, Hollingsworth MA. The N-terminal domain of G3BP enhances cell motility and invasion by posttranscriptional regulation of BART. Mol Cancer Res. 2011; 9:856–866.13. Barnes CJ, Li F, Mandal M, Yang Z, Sahin AA, Kumar R. Heregulin induces expression, ATPase activity, and nuclear localization of G3BP, a Ras signaling component, in human breast tumors. Cancer Res. 2002; 62:1251–1255.14. Winslow S, Leandersson K, Larsson C. Regulation of PMP22 mRNA by G3BP1 affects cell proliferation in breast cancer cells. Mol Cancer. 2013; 12:156.15. Zhang HZ, Liu JG, Wei YP, Wu C, Cao YK, Wang M. Expression of G3BP and RhoC in esophageal squamous carcinoma and their effect on prognosis. World J Gastroenterol. 2007; 13:4126–4130.16. Parker F, Maurier F, Delumeau I, Duchesne M, Faucher D, Debussche L, et al. A Ras-GTPase-activating protein SH3-domain-binding protein. Mol Cell Biol. 1996; 16:2561–2569.17. Kennedy D, French J, Guitard E, Ru K, Tocque B, Mattick J. Characterization of G3BPs: tissue specific expression, chromosomal localisation and rasGAP(120) binding studies. J Cell Biochem. 2001; 84:173–187.18. Tourrière H, Chebli K, Zekri L, Courselaud B, Blanchard JM, Bertrand E, et al. The RasGAP-associated endoribonuclease G3BP assembles stress granules. J Cell Biol. 2003; 160:823–831.19. Gallouzi IE, Parker F, Chebli K, Maurier F, Labourier E, Barlat I, et al. A novel phosphorylation-dependent RNase activity of GAP-SH3 binding protein: a potential link between signal transduction and RNA stability. Mol Cell Biol. 1998; 18:3956–3965.20. Miao R, Guo X, Zhi Q, Shi Y, Li L, Mao X, et al. VEZT, a novel putative tumor suppressor, suppresses the growth and tumorigenicity of gastric cancer. PLoS One. 2013; 8:e74409.21. Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004; 116:281–297.22. Li YS, Chen YZ, Guo XB, Liu X, Li LP. VEZT as a novel independent prognostic factor in gastric cancer. Cancer Biomark. 2015; 15:375–380.23. Bahloul A, Simmler MC, Michel V, Leibovici M, Perfettini I, Roux I, et al. Vezatin, an integral membrane protein of adherens junctions, is required for the sound resilience of cochlear hair cells. EMBO Mol Med. 2009; 1:125–138.24. Guo X, Jing C, Li L, Zhang L, Shi Y, Wang J, et al. Down-regulation of VEZT gene expression in human gastric cancer involves promoter methylation and miR-43c. Biochem Biophys Res Commun. 2011; 404:622–627.25. Ortega ÁD, Willers IM, Sala S, Cuezva JM. Human G3BP1 interacts with β-F1-ATPase mRNA and inhibits its translation. J Cell Sci. 2010; 123:2685–2696.26. Izadi A, Moslemi E, Poorhosseini SM, Yassaee VR, Kheiri HR, Elikai HR. UBD identify in paraffin tissues in patients with colorectal cancer. J Isfahan Med Sch. 2014; 32:1–10.27. Kim J, Cho YA, Choi WJ, Jeong SH. Gene-diet interactions in gastric cancer risk: a systematic review. World J Gastroenterol. 2014; 20:9600–9610.28. Somasekharan SP, El-Naggar A, Leprivier G, Cheng H, Hajee S, Grunewald TG, et al. YB-1 regulates stress granule formation and tumor progression by translationally activating G3BP1 . J Cell Biol. 2015; 208:913–929.29. Annibaldi A, Dousse A, Martin S, Tazi J, Widmann C. Revisiting G3BP1 as a RasGAP binding protein: sensitization of tumor cells to chemotherapy by the RasGAP 317-326 sequence does not involve G3BP1 . PLoS One. 2011; 6:e29024.30. Kim MM, Wiederschain D, Kennedy D, Hansen E, Yuan ZM. Modulation of p53 and MDM2 activity by novel interaction with Ras-GAP binding proteins (G3BP). Oncogene. 2007; 26:4209–4215.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Cyclooxygenase-2 Expression on Lymphangiogenesis and Lymph Node Metastasis in Gastric Cancer Tissues
- Expression of Cyclooxygenase-2 Protein in Gastric Carcinogenesis
- MUC2 Expression Is Correlated with Tumor Differentiation and Inhibits Tumor Invasion in Gastric Carcinomas: A Systematic Review and Meta-analysis
- Clinicopathological Significance of p53 and HSP27 in Gastric-cancer Patients
- Prognostic Value of p53 and Proliferating Cell Nuclear Antigen ( PCNA ) in Stage 3 Gastric Carcinoma