Korean J Physiol Pharmacol.
2016 Sep;20(5):477-485. 10.4196/kjpp.2016.20.5.477.
Histone deacetylase inhibitor, CG200745, attenuates cardiac hypertrophy and fibrosis in DOCA-induced hypertensive rats
- Affiliations
-
- 1Department of Pharmacology, Kyungpook National University School of Medicine, Daegu 41944, Korea. inkim@knu.ac.kr
- 2Cardiovascular Research Institute, Kyungpook National University School of Medicine, Daegu 41944, Korea.
- 3Cell and Matrix Research Institute, Kyungpook National University School of Medicine, Daegu 41944, Korea.
- 4BK21 Plus KNU Biomedical Convergence Program, Department of Biomedical Science, Kyungpook National University School of Medicine, Daegu 41944, Korea.
- 5Department of Physiology, Kyungpook National University School of Medicine, Daegu 41944, Korea.
- 6Translational Research Center, CrystalGenomics, Inc., Seongnam 13488, Korea.
- KMID: 2350504
- DOI: http://doi.org/10.4196/kjpp.2016.20.5.477
Abstract
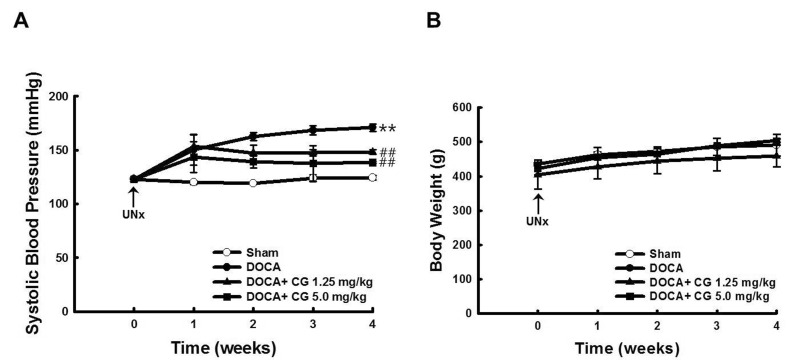
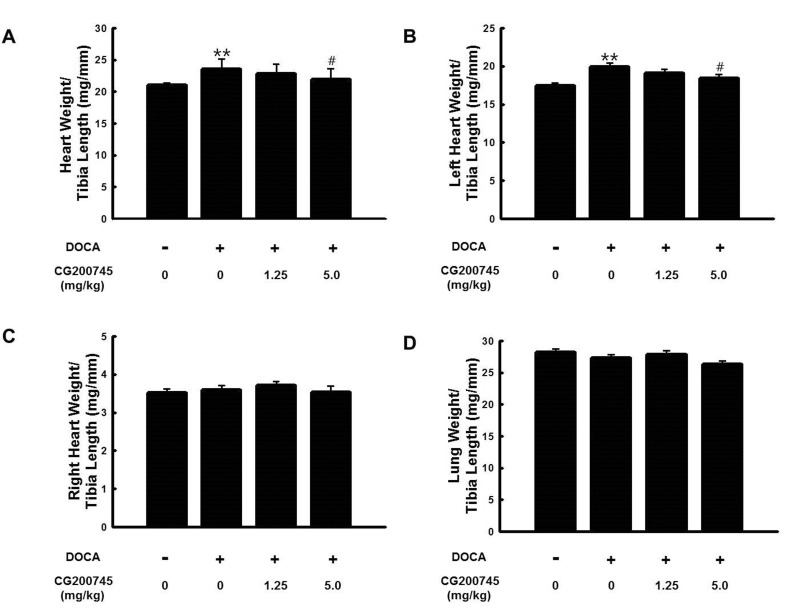
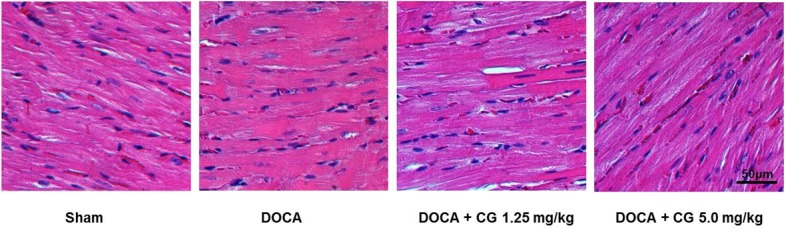
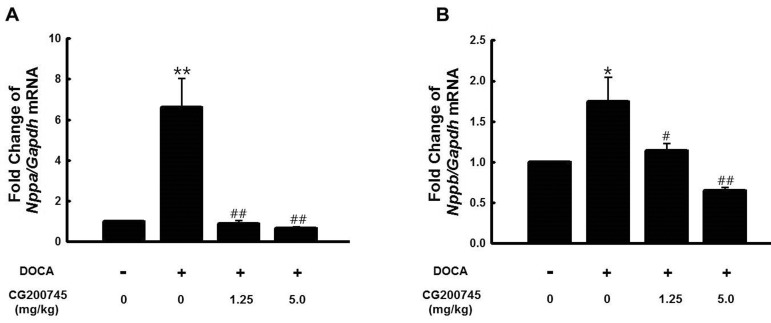
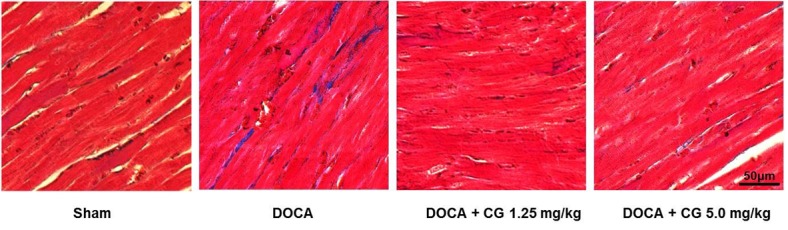
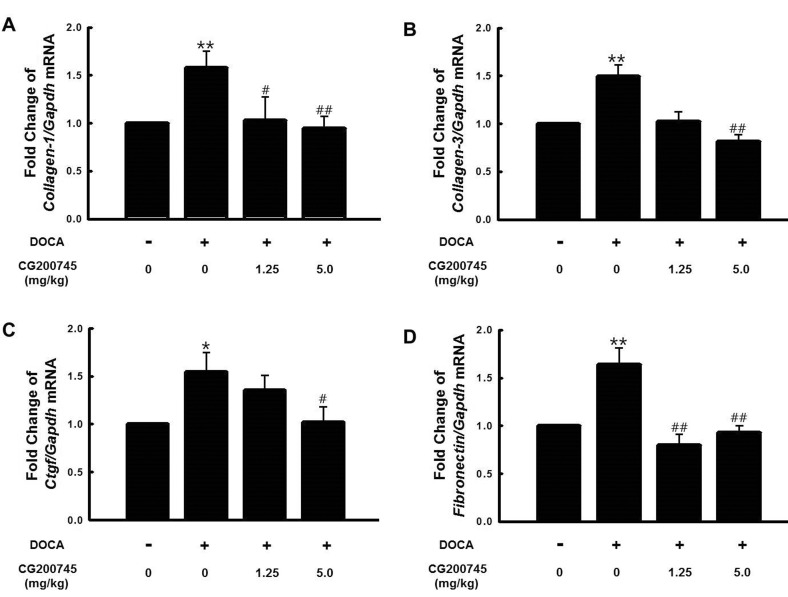
- CG200745 is a novel inhibitor of histone deacetylases (HDACs), initially developed for treatment of various hematological and solid cancers. Because it is water-soluble, it can be administered orally. We hypothesized that the HDAC inhibitor, CG200745, attenuates cardiac hypertrophy and fibrosis in deoxycorticosterone acetate (DOCA)-induced hypertensive rats. For establishment of hypertension, 40 mg/kg of DOCA was subcutaneously injected four times weekly into Sprague-Dawley rats. All the rats used in this study including those in the sham group had been unilaterally nephrectomized and allowed free access to drinking water containing 1% NaCl. Systolic blood pressure was measured by the tail-cuff method. Blood chemistry including sodium, potassium, glucose, triglyceride, and cholesterol levels was analyzed. Sections of the heart were visualized after trichrome and hematoxylin and eosin stain. The expression of hypertrophic genes such as atrial natriuretic peptide A (Nppa) and atrial natriuretic peptide B (Nppb) in addition to fibrotic genes such as Collagen-1, Collagen-3, connective tissue growth factor (Ctgf), and Fibronectin were measured by quantitative real-time PCR (qRT-PCR). Injection of DOCA increased systolic blood pressure, heart weight, and cardiac fibrosis, which was attenuated by CG200745. Neither DOCA nor CG200745 affected body weight, vascular contraction and relaxation responses, and blood chemistry. Injection of DOCA increased expression of both hypertrophic and fibrotic genes, which was abrogated by CG200745. These results indicate that CG200745 attenuates cardiac hypertrophy and fibrosis in DOCA-induced hypertensive rats.
Keyword
MeSH Terms
-
Animals
Blood Pressure
Body Weight
Cardiomegaly*
Chemistry
Cholesterol
Connective Tissue Growth Factor
Desoxycorticosterone
Desoxycorticosterone Acetate
Drinking Water
Eosine Yellowish-(YS)
Fibronectins
Fibrosis*
Glucose
Heart
Hematoxylin
Histone Deacetylase Inhibitors*
Histone Deacetylases*
Histones*
Hypertension
Methods
Potassium
Rats*
Rats, Sprague-Dawley
Real-Time Polymerase Chain Reaction
Relaxation
Sodium
Triglycerides
Cholesterol
Connective Tissue Growth Factor
Desoxycorticosterone
Drinking Water
Eosine Yellowish-(YS)
Fibronectins
Glucose
Hematoxylin
Histone Deacetylase Inhibitors
Histone Deacetylases
Histones
Potassium
Sodium
Figure
Cited by 2 articles
-
The effect of melatonin on cardio fibrosis in juvenile rats with pressure overload and deregulation of HDACs
Yao Wu, Feifei Si, Li Luo, Fengchuan Jing, Kunfeng Jiang, Jiwei Zhou, Qijian Yi
Korean J Physiol Pharmacol. 2018;22(6):607-616. doi: 10.4196/kjpp.2018.22.6.607.Sulforaphane Ameliorates Diabetes-Induced Renal Fibrosis through Epigenetic Up-Regulation of BMP-7
Lili Kong, Hongyue Wang, Chenhao Li, Huiyan Cheng, Yan Cui, Li Liu, Ying Zhao
Diabetes Metab J. 2021;45(6):909-920. doi: 10.4093/dmj.2020.0168.
Reference
-
1. Yip GW, Fung JW, Tan YT, Sanderson JE. Hypertension and heart failure: a dysfunction of systole, diastole or both? J Hum Hypertens. 2009; 23:295–306. PMID: 19037230.
Article2. Writing Group Members. Lloyd-Jones D, Adams RJ, Brown TM, Carnethon M, Dai S, De Simone G, Ferguson TB, Ford E, Furie K, Gillespie C, Go A, Greenlund K, Haase N, Hailpern S, Ho PM, Howard V, Kissela B, Kittner S, Lackland D, Lisabeth L, Marelli A, McDermott MM, Meigs J, Mozaffarian D, Mussolino M, Nichol G, Roger VL, Rosamond W, Sacco R, Sorlie P, Roger VL, Thom T, Wasserthiel-Smoller S, Wong ND, Wylie-Rosett J; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics--2010 update: a report from the American Heart Association. Circulation. 2010; 121:e46–e215. PMID: 20019324.3. Bush EW, McKinsey TA. Protein acetylation in the cardiorenal axis: the promise of histone deacetylase inhibitors. Circ Res. 2010; 106:272–284. PMID: 20133912.4. Smith KT, Workman JL. Introducing the acetylome. Nat Biotechnol. 2009; 27:917–919. PMID: 19816449.
Article5. Norris KL, Lee JY, Yao TP. Acetylation goes global: the emergence of acetylation biology. Sci Signal. 2009; 2:pe76. PMID: 19920250.
Article6. Gregoretti IV, Lee YM, Goodson HV. Molecular evolution of the histone deacetylase family: functional implications of phylogenetic analysis. J Mol Biol. 2004; 338:17–31. PMID: 15050820.
Article7. Smith KT, Workman JL. Histone deacetylase inhibitors: anticancer compounds. Int J Biochem Cell Biol. 2009; 41:21–25. PMID: 18845268.
Article8. Kang SH, Seok YM, Song MJ, Lee HA, Kurz T, Kim I. Histone deacetylase inhibition attenuates cardiac hypertrophy and fibrosis through acetylation of mineralocorticoid receptor in spontaneously hypertensive rats. Mol Pharmacol. 2015; 87:782–791. PMID: 25667225.
Article9. Lee HA, Lee DY, Cho HM, Kim SY, Iwasaki Y, Kim IK. Histone deacetylase inhibition attenuates transcriptional activity of mineralocorticoid receptor through its acetylation and prevents development of hypertension. Circ Res. 2013; 112:1004–1012. PMID: 23421989.
Article10. Kee HJ, Bae EH, Park S, Lee KE, Suh SH, Kim SW, Jeong MH. HDAC inhibition suppresses cardiac hypertrophy and fibrosis in DOCA-salt hypertensive rats via regulation of HDAC6/HDAC8 enzyme activity. Kidney Blood Press Res. 2013; 37:229–239. PMID: 23868068.
Article11. Cardinale JP, Sriramula S, Pariaut R, Guggilam A, Mariappan N, Elks CM, Francis J. HDAC inhibition attenuates inflammatory, hypertrophic, and hypertensive responses in spontaneously hypertensive rats. Hypertension. 2010; 56:437–444. PMID: 20679181.
Article12. Jang EJ, Seok YM, Lee JI, Cho HM, Sohn UD, Kim IK. 3',4'-Dimethoxythioflavone induces endothelium-dependent vasorelaxation through activation of epidermal growth factor receptor. Naunyn Schmiedebergs Arch Pharmacol. 2013; 386:339–350. PMID: 23232926.
Article13. Lv L, Tang YP, Han X, Wang X, Dong Q. Therapeutic application of histone deacetylase inhibitors for stroke. Cent Nerv Syst Agents Med Chem. 2011; 11:138–149. PMID: 21521169.
Article14. McKinsey TA. Targeting inflammation in heart failure with histone deacetylase inhibitors. Mol Med. 2011; 17:434–441. PMID: 21267510.
Article15. Iyer A, Fenning A, Lim J, Le GT, Reid RC, Halili MA, Fairlie DP, Brown L. Antifibrotic activity of an inhibitor of histone deacetylases in DOCA-salt hypertensive rats. Br J Pharmacol. 2010; 159:1408–1417. PMID: 20180942.
Article16. Ooi JY, Tuano NK, Rafehi H, Gao XM, Ziemann M, Du XJ, El-Osta A. HDAC inhibition attenuates cardiac hypertrophy by acetylation and deacetylation of target genes. Epigenetics. 2015; 10:418–430. PMID: 25941940.
Article17. Lemon DD, Horn TR, Cavasin MA, Jeong MY, Haubold KW, Long CS, Irwin DC, McCune SA, Chung E, Leinwand LA, McKinsey TA. Cardiac HDAC6 catalytic activity is induced in response to chronic hypertension. J Mol Cell Cardiol. 2011; 51:41–50. PMID: 21539845.
Article18. Eom GH, Cho YK, Ko JH, Shin S, Choe N, Kim Y, Joung H, Kim HS, Nam KI, Kee HJ, Kook H. Casein kinase-2α1 induces hypertrophic response by phosphorylation of histone deacetylase 2 S394 and its activation in the heart. Circulation. 2011; 123:2392–2403. PMID: 21576649.
Article19. Nural-Guvener HF, Zakharova L, Nimlos J, Popovic S, Mastroeni D, Gaballa MA. HDAC class I inhibitor, Mocetinostat, reverses cardiac fibrosis in heart failure and diminishes CD90+ cardiac myofibroblast activation. Fibrogenesis Tissue Repair. 2014; 7:10. PMID: 25024745.
Article20. Williams SM, Golden-Mason L, Ferguson BS, Schuetze KB, Cavasin MA, Demos-Davies K, Yeager ME, Stenmark KR, McKinsey TA. Class I HDACs regulate angiotensin II-dependent cardiac fibrosis via fibroblasts and circulating fibrocytes. J Mol Cell Cardiol. 2014; 67:112–125. PMID: 24374140.
Article21. Tao H, Yang JJ, Hu W, Shi KH, Li J. HDAC6 Promotes Cardiac Fibrosis Progression through Suppressing RASSF1A Expression. Cardiology. 2016; 133:18–26. PMID: 26401643.
Article22. Chun SM, Lee JY, Choi J, Lee JH, Hwang JJ, Kim CS, Suh YA, Jang SJ. Epigenetic modulation with HDAC inhibitor CG200745 induces anti-proliferation in non-small cell lung cancer cells. PLoS One. 2015; 10:e0119379. PMID: 25781604.
Article23. Hwang JJ, Kim YS, Kim T, Kim MJ, Jeong IG, Lee JH, Choi J, Jang S, Ro S, Kim CS. A novel histone deacetylase inhibitor, CG200745, potentiates anticancer effect of docetaxel in prostate cancer via decreasing Mcl-1 and Bcl-XL. Invest New Drugs. 2012; 30:1434–1442. PMID: 21773733.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Role of tyrosine kinases in vascular contraction in deoxycorticosterone acetate-salt hypertensive rats
- YAK577 Attenuates Cardiac Remodeling and Fibrosis in Isoproterenol-Infused Heart Failure Mice by Downregulating MMP12
- Expression of Class I and Class II a/b Histone Deacetylase is Dysregulated in Hypertensive Animal Models
- Potential of histone deacetylase 6 inhibitors in alleviating chemotherapy-induced peripheral neuropathy
- Time-Related Alterations of Endogenous Ouabain in DOCA-Salt Hypertensive Rats