Tuberc Respir Dis.
2015 Apr;78(2):78-84. 10.4046/trd.2015.78.2.78.
Outcomes and Use of Therapeutic Drug Monitoring in Multidrug-Resistant Tuberculosis Patients Treated in Virginia, 2009-2014
- Affiliations
-
- 1Division of Infectious Diseases and International Health, University of Virginia, Charlottesville, VA, USA. skh8r@virginia.edu
- 2Tuberculosis Control and Newcomer Health, Virginia Department of Health, Richmond, VA, USA.
- 3College of Pharmacy and Emerging Pathogens Institute, University of Florida, Gainesville, FL, USA.
- 4Southeastern National Tuberculosis Center and the University of Miami, Miami, FL, USA.
- KMID: 2320598
- DOI: http://doi.org/10.4046/trd.2015.78.2.78
Abstract
- BACKGROUND
Reports of therapeutic drug monitoring (TDM) for second-line medications to treat multidrug-resistant tuberculosis (MDR-TB) remain limited.
METHODS
A retrospective cohort from the Virginia state tuberculosis (TB) registry, 2009-2014, was analyzed for TDM usage in MDR-TB. Drug concentrations, measured at time of estimated peak (Cmax), were compared to expected ranges.
RESULTS
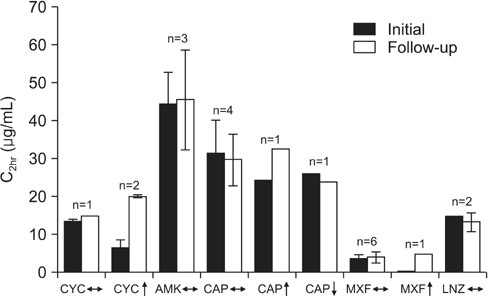
Of 10 patients with MDR-TB, 8 (80%) had TDM for at least one drug (maximum 6 drugs). Second-line drugs tested were cycloserine in seven patients (mean C2hr, 16.6+/-10.2 microg/mL; 4 [57%] below expected range); moxifloxacin in five (mean C2hr, 3.2+/-1.5 microg/mL; 1 [20%] below); capreomycin in five (mean C2hr, 21.5+/-14.0 microg/mL; 3 [60%] below); para-aminosalicylic acid in five (mean C6hr, 65.0+/-29.1 microg/mL; all within or above); linezolid in three (mean C2hr, 11.4+/-4.1 microg/mL, 1 [33%] below); amikacin in two (mean C2hr, 35.3+/-3.7 microg/mL; 1 [50%] below); ethionamide in one (C2hr, 1.49 microg/mL, within expected). Two patients died: a 38-year-old woman with human immunodeficiency virus/acquired immune deficiency syndrome and TB meningitis without TDM, and a 76-year-old man with fluoroquinolone-resistant (pre-extensively drug-resistant) pulmonary TB and low linezolid and capreomycin concentrations.
CONCLUSION
Individual pharmacokinetic variability was common. A more standardized approach to TDM for MDR-TB may limit over-testing and maximize therapeutic gain.
Keyword
MeSH Terms
Figure
Reference
-
1. Shenoi S, Heysell S, Moll A, Friedland G. Multidrug-resistant and extensively drug-resistant tuberculosis: consequences for the global HIV community. Curr Opin Infect Dis. 2009; 22:11–17.2. Gandhi NR, Nunn P, Dheda K, Schaaf HS, Zignol M, van Soolingen D, et al. Multidrug-resistant and extensively drug-resistant tuberculosis: a threat to global control of tuberculosis. Lancet. 2010; 375:1830–1843.3. Marks SM, Flood J, Seaworth B, Hirsch-Moverman Y, Armstrong L, Mase S, et al. Treatment practices, outcomes, and costs of multidrug-resistant and extensively drug-resistant tuberculosis, United States, 2005-2007. Emerg Infect Dis. 2014; 20:812–821.4. Rajbhandary SS, Marks SM, Bock NN. Costs of patients hospitalized for multidrug-resistant tuberculosis. Int J Tuberc Lung Dis. 2004; 8:1012–1016.5. Althomsons SP, Cegielski JP. Impact of second-line drug resistance on tuberculosis treatment outcomes in the United States: MDR-TB is bad enough. Int J Tuberc Lung Dis. 2012; 16:1331–1334.6. Pasipanodya JG, McIlleron H, Burger A, Wash PA, Smith P, Gumbo T. Serum drug concentrations predictive of pulmonary tuberculosis outcomes. J Infect Dis. 2013; 208:1464–1473.7. Heysell SK, Moore JL, Keller SJ, Houpt ER. Therapeutic drug monitoring for slow response to tuberculosis treatment in a state control program, Virginia, USA. Emerg Infect Dis. 2010; 16:1546–1553.8. Heysell SK, Moore JL, Staley D, Dodge D, Houpt ER. Early Therapeutic drug monitoring for isoniazid and rifampin among diabetics with newly diagnosed tuberculosis in Virginia, USA. Tuberc Res Treat. 2013; 2013:129723.9. Chan ED, Laurel V, Strand MJ, Chan JF, Huynh ML, Goble M, et al. Treatment and outcome analysis of 205 patients with multidrug-resistant tuberculosis. Am J Respir Crit Care Med. 2004; 169:1103–1109.10. Peloquin CA. Therapeutic drug monitoring in the treatment of tuberculosis. Drugs. 2002; 62:2169–2183.11. California Department of Public Health. Curry International TB Center. Drug-resistant tuberculosis: a survival guide for clinicians. 2nd edition. Oakland: Curry International TB Center;2008. p. 66–67.12. Mpagama SG, Ndusilo N, Stroup S, Kumburu H, Peloquin CA, Gratz J, et al. Plasma drug activity in patients on treatment for multidrug-resistant tuberculosis. Antimicrob Agents Chemother. 2014; 58:782–788.13. Alsultan A, Peloquin CA. Therapeutic drug monitoring in the treatment of tuberculosis: an update. Drugs. 2014; 74:839–854.14. Casali N, Nikolayevskyy V, Balabanova Y, Ignatyeva O, Kontsevaya I, Harris SR, et al. Microevolution of extensively drugresistant tuberculosis in Russia. Genome Res. 2012; 22:735–745.15. Zaunbrecher MA, Sikes RD Jr, Metchock B, Shinnick TM, Posey JE. Overexpression of the chromosomally encoded aminoglycoside acetyltransferase eis confers kanamycin resistance in Mycobacterium tuberculosis. Proc Natl Acad Sci U S A. 2009; 106:20004–20009.16. Mpagama SG, Houpt ER, Stroup S, Kumburu H, Gratz J, Kibiki GS, et al. Application of quantitative second-line drug susceptibility testing at a multidrug-resistant tuberculosis hospital in Tanzania. BMC Infect Dis. 2013; 13:432.17. Rowlinson MC. ighth National Conference on Laboratory Aspects of Tuberculosis. Association of Public Health Laboratories. MICs in TB susceptibility testing: challenges and solutions for implementation. San Diego, CA, USA: 2013. 08. 19-21.18. Banu S, Rahman SM, Khan MS, Ferdous SS, Ahmed S, Gratz J, et al. Discordance across several methods for drug susceptibility testing of drug-resistant Mycobacterium tuberculosis isolates in a single laboratory. J Clin Microbiol. 2014; 52:156–163.19. Lee J, Armstrong DT, Ssengooba W, Park JA, Yu Y, Mumbowa F, et al. Sensititre MYCOTB MIC plate for testing Mycobacterium tuberculosis susceptibility to first- and second-line drugs. Antimicrob Agents Chemother. 2014; 58:11–18.20. Chigutsa E, Meredith S, Wiesner L, Padayatchi N, Harding J, Moodley P, et al. Population pharmacokinetics and pharmacodynamics of ofloxacin in South African patients with multidrug-resistant tuberculosis. Antimicrob Agents Chemother. 2012; 56:3857–3863.21. Hung WY, Yu MC, Chiang YC, Chang JH, Chiang CY, Chang CC, et al. Serum concentrations of cycloserine and outcome of multidrug-resistant tuberculosis in Northern Taiwan. Int J Tuberc Lung Dis. 2014; 18:601–606.22. Lee M, Lee J, Carroll MW, Choi H, Min S, Song T, et al. Linezolid for treatment of chronic extensively drug-resistant tuberculosis. N Engl J Med. 2012; 367:1508–1518.23. Peloquin CA, Berning SE, Huitt GA, Childs JM, Singleton MD, James GT. Once-daily and twice-daily dosing of p-aminosalicylic acid granules. Am J Respir Crit Care Med. 1999; 159:932–934.24. Heysell SK, Houpt ER. Optimizing second-line therapy for drug-resistant tuberculosis: the additive value of sequencing for multiple resistance loci. Antimicrob Agents Chemother. 2011; 55:3968–3969.25. Campbell PJ, Morlock GP, Sikes RD, Dalton TL, Metchock B, Starks AM, et al. Molecular detection of mutations associated with first- and second-line drug resistance compared with conventional drug susceptibility testing of Mycobacterium tuberculosis. Antimicrob Agents Chemother. 2011; 55:2032–2041.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Medical Management of Drug-Resistant Tuberculosis
- Multidrug-resistant Tuberculosis Spondylitis: A Case Report
- Pediatric tuberculosis and drug resistance
- Diagnosis and treatment of multidrug-resistant tuberculosis
- Drug Resistance Patterns of Multidrug- and Extensively Drug-Resistant Tuberculosis in Korea: Amplification of Resistance to Oral Second-line Drugs