Restor Dent Endod.
2012 Nov;37(4):194-200.
Evaluation of the rat tissue reaction to experimental new resin cement and mineral trioxide aggregate cement
- Affiliations
-
- 1Department of Conservative Dentistry, Ulsan University Asan Medical Center, Seoul, Korea. kmr333@amc.seoul.kr
Abstract
OBJECTIVES
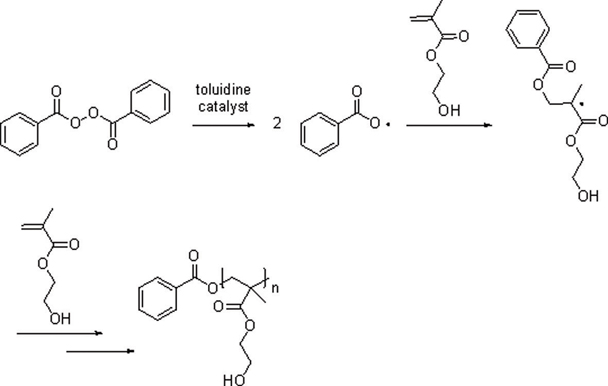
New resin cement (NRC) has been developed as a root repairing material and the material is composed of organic resin matrix and inorganic powders. The aim of this study was to compare the rat subcutaneous tissue response to NRC and mineral trioxide aggregate (MTA) cement and to investigate the tissue toxicity of both materials.
MATERIALS AND METHODS
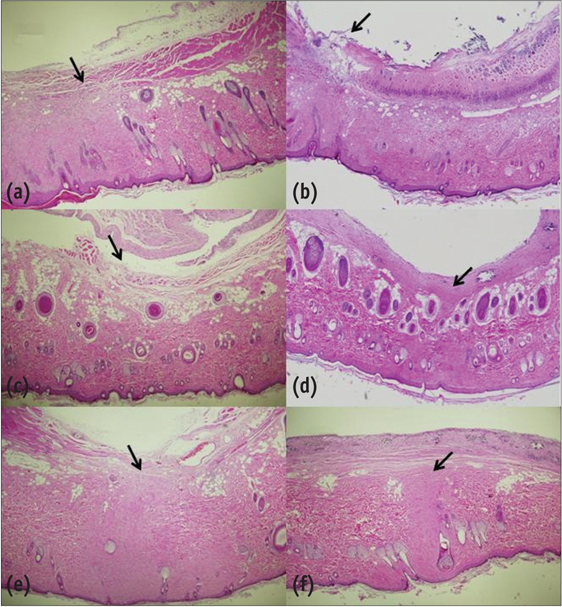
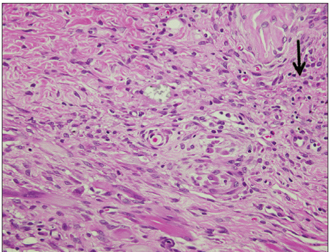
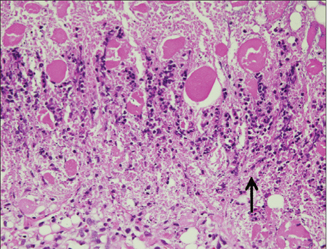
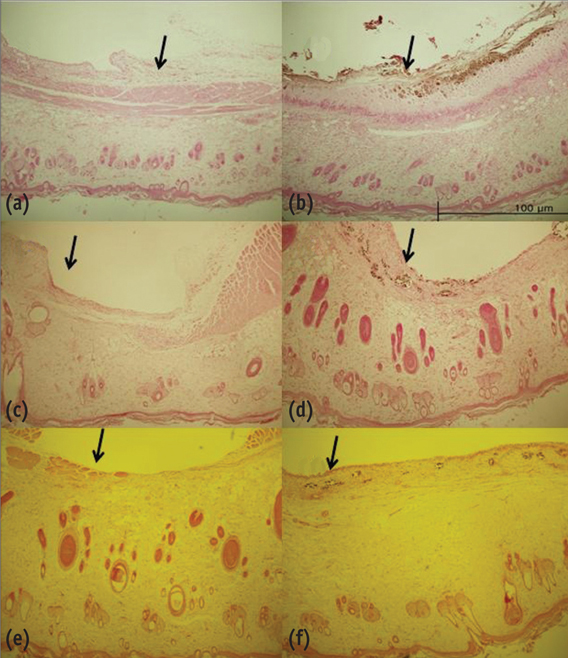
Sixty rats received two polyethylene tube-implants in dorsal subcutaneous regions, MTA and NRC specimens. Twenty rats were sacrificed respectively at 1, 4 and 8 wk after implantation and sectioned to 5 microm thickness and stained with Hematoxylin-Eosin (H-E) or von-Kossa staining. The condition of tissue adjacent to the implanted materials and the extent of inflammation to each implant were evaluated by two examiners who were unaware of the type of implanted materials in the tissues. Data were statistically analyzed with paired t-test (p < 0.05).
RESULTS
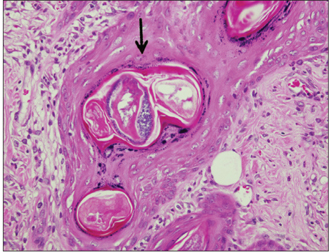
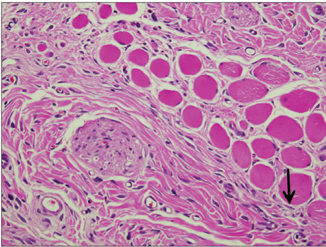
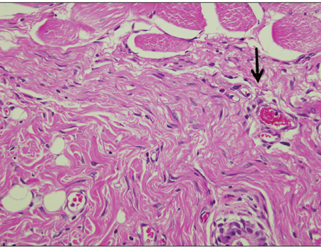
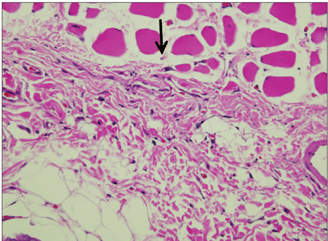
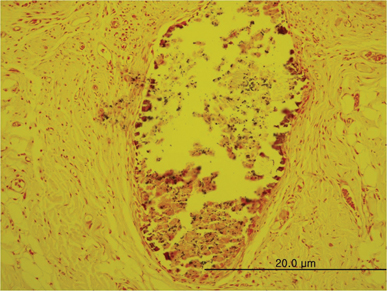
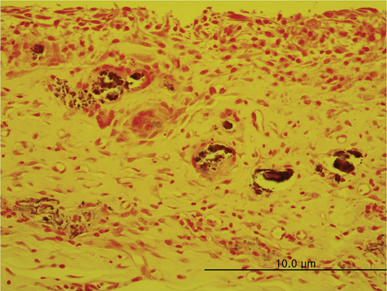
In specimens implanted with both NRC and MTA, severe inflammatory reactions were present at one wk, which decreased with time. At eighth wk, MTA implanted tissue showed mild inflammatory reaction, while there were moderate inflammatory reactions in NRC implanted tissue, respectively. In NRC group, von-Kossa staining showed more calcification materials than MTA group at eighth wk.
CONCLUSIONS
It was concluded that the calcium reservoir capability of NRC may contribute to mineralization of the tissues.
MeSH Terms
Figure
Reference
-
1. Imura N, Pinheiro ET, Gomes BP, Zaia AA, Ferraz CC, Souza-Filho FJ. The outcome of endodontic treatment: a retrospective study of 2000 cases performed by a specialist. J Endod. 2007. 33:1278–1282.
Article2. McDonald NJ, Hovland EJ. Walton RE, Torabinejad M, editors. Surgical endodontics. Principles and practice of endodontics. 1996. 2nd ed. Philadelphia, PA: W.B. Saunders Co.;401–422.3. Lee SJ, Monsef M, Torabinejad M. Sealing ability of a mineral trioxide aggregate for repair of lateral root perforations. J Endod. 1993. 19:541–544.
Article4. Lee YL, Lin FH, Wang WH, Ritchie HH, Lan WH, Lin CP. Effects of EDTA on the hydration mechanism of mineral trioxide aggregate. J Dent Res. 2007. 86:534–538.
Article5. Parirokh M, Torabinejad M. Mineral trioxide aggregate: a comprehensive literature review-Part III: Clinical applications, drawbacks, and mechanism of action. J Endod. 2010. 36:400–413.
Article6. Johnson BR. Considerations in the selection of a root-end filling material. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999. 87:398–404.
Article7. Kim M, Ko H, Yang W, Lee Y, Kim S, Mante FK. A new resin-bonded retrograde filling material. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009. 108:e111–e116.
Article8. Roberts HW, Toth JM, Berzins DW, Charlton DG. Mineral trioxide aggregate material use in endodontic treatment: a review of the literature. Dent Mater. 2008. 24:149–164.
Article9. Gomes-Filho JE, Rodrigues G, Watanabe S, Estrada Bernabé PF, Lodi CS, Gomes AC, Faria MD, Domingos Dos Santos A, Silos Moraes JC. Evaluation of the tissue reaction to fast endodontic cement (CER) and Angelus MTA. J Endod. 2009. 35:1377–1380.
Article10. Yaltirik M, Ozbas H, Bilgic B, Issever H. Reactions of connective tissue to mineral trioxide aggregate and amalgam. J Endod. 2004. 30:95–99.
Article11. Holland R, de Souza V, Nery MJ, Otoboni Filho JA, Bernabé PF, Dezan Júnior E. Reaction of rat connective tissue to implanted dentin tubes filled with mineral trioxide aggregate or calcium hydroxide. J Endod. 1999. 25:161–166.
Article12. Morse DR, Wilcko JM, Pullon PA, Furst ML, Passo SA. A comparative tissue toxicity evaluation of the liquid components of gutta-percha root canal sealers. J Endod. 1981. 7:545–550.
Article13. Hammad HM, Hamadah MA, Al-Omari WM. Histological evaluation of rat tissue response to GMTA, Retroplast, and Geristore retrograde filling materials. Aust Endod J. 2011. 37:18–25.
Article14. Torneck CD. Reaction of rat connective tissue to polyethylene tube implants. I. Oral Surg Oral Med Oral Pathol. 1966. 21:379–387.15. Olsson B, Sliwkowski A, Langeland K. Subcutaneous implantation for the biological evaluation of endodontic materials. J Endod. 1981. 7:355–367.
Article16. Parirokh M, Mirsoltani B, Raoof M, Tabrizchi H, Haghdoost AA. Comparative study of subcutaneous tissue responses to a novel root-end filling material and white and grey mineral trioxide aggregate. Int Endod J. 2011. 44:283–289.
Article17. Shahi S, Rahimi S, Lotfi M, Yavari H, Gaderian A. A comparative study of the biocompatibility of three root-end filling materials in rat connective tissue. J Endod. 2006. 32:776–780.
Article18. Mohammadi Z, Yazdizadeh M. Obturation of immature non-vital tooth using MTA. Case report. N Y State Dent J. 2011. 77:33–35.19. Witherspoon DE, Small JC, Regan JD, Nunn M. Retrospective analysis of open apex teeth obturated with mineral trioxide aggregate. J Endod. 2008. 34:1171–1176.
Article20. Jung IY, Kim ES, Lee CY, Lee SJ. Continued development of the root separated from the main root. J Endod. 2011. 37:711–714.21. Torabinejad M, Pitt Ford TR, McKendry DJ, Abedi HR, Miller DA, Kariyawasam SP. Histologic assessment of mineral trioxide aggregate as a root-end filling in monkeys. J Endod. 1997. 23:225–228.
Article22. Foreman PC, Barnes IE. Review of calcium hydroxide. Int Endod J. 1990. 23:283–297.23. Asgary S, Eghbal MJ, Parirokh M, Ghoddusi J. Effect of two storage solutions on surface topography of two root-end fillings. Aust Endod J. 2009. 35:147–152.
Article24. Zayzafoon M. Calcium/calmodulin signaling controls osteoblast growth and differentiation. J Cell Biochem. 2006. 97:56–70.
Article25. Paula-Silva FW, Ghosh A, Arzate H, Kapila S, da Silva LA, Kapila YL. Calcium hydroxide promotes cementogenesis and induces cementoblastic differentiation of mesenchymal periodontal ligament cells in a CEMP1- and ERK-dependent manner. Calcif Tissue Int. 2010. 87:144–157.
Article26. Sheehan D, Hrapchak B. Theory and practice of histotechnology. 1980. 2nd ed. Columbus, Ohio: Battelle Press;226–227.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Mineral trioxied aggregate and its substitutes
- A literature review on cementation of implant prosthesis
- The study on the color stability of resin cement used in all ceramic crown
- Biomechanical three-dimensional finite element analysis of monolithic zirconia crown with different cement type
- Physical and chemical properties of experimental mixture of mineral trioxide aggregate and glass ionomer cement