Pediatr Allergy Respir Dis.
2012 Jun;22(2):180-187.
Role of Fractional Exhaled Nitric Oxide in Predicting Development of Allergic Rhinits in Children with Bronchial Asthma
- Affiliations
-
- 1Department of Pediatrics, Kyung Hee University School of Medicine, Seoul, Korea. yhrha@khu.ac.kr
Abstract
- PURPOSE
Fractional exhaled nitric oxide (FeNO) has been widely reported as a marker for airway inflammationin, and FeNO have shown increased levels in the non-asthmatic patients with other atopy related diseases, such as rhinitis. Bronchial hyperresponsiveness is a characteristic feature of asthma, which is often associated with airway inflammationin allergic rhinitis. It has been suggested that asthma is associated with rhinitis, which is a link between the upper and the lower airways, beyond allergy associated inflammation in the respiratory tract. To evaluate the usefulness of FeNO measurement, as a risk factor of allergic and nonallergic rhinitis in children with asthma.
METHODS
Fifty-three children included in this study were diagnosed as asthma from April through August 2005 in the Department of Pediatrics, Kyung Hee University School of Medicine. They conducted FeNO monitoring, total eosinophil count and serum immunoglobulin E at that time. We put a question to the participants' parents about the doctor diagnosed participants' manifestations (sneezing, nasal congestion, nasal itching, rhinorrhea), via a telephone interview survey or at the outpatient department in July 2011. Statistical analyses were performed using IBM SPSS ver. 18.0.
RESULTS
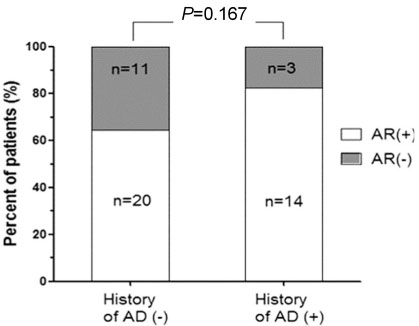
The overall prevalence of rhinitis, among the 53 children, was 67.9%. The means of FeNO differed significantly between the two groups, being higher in children who have rhinitis symptoms and lower in asymptomatic children. (mean+/-SD, 29.4+/-24.6 to 13.6+/-11.8 parts per billion; P=0.003; T-test)
CONCLUSION
To take the measurement of FeNO value in asthmatics may be a tool in the predictor of the causes of rhinitis.
MeSH Terms
-
Aluminum Hydroxide
Asthma
Carbonates
Child
Eosinophils
Estrogens, Conjugated (USP)
Humans
Hypersensitivity
Immunoglobulin E
Immunoglobulins
Inflammation
Interviews as Topic
Nitric Oxide
Outpatients
Parents
Pediatrics
Prevalence
Pruritus
Respiratory System
Rhinitis
Rhinitis, Allergic, Perennial
Risk Factors
Aluminum Hydroxide
Carbonates
Estrogens, Conjugated (USP)
Immunoglobulin E
Immunoglobulins
Nitric Oxide
Figure
Reference
-
1. Schoenwetter WF, Dupclay L Jr, Appajosyula S, Botteman MF, Pashos CL. Economic impact and quality-of-life burden of allergic rhinitis. Curr Med Res Opin. 2004. 20:305–317.
Article2. Strachan D, Sibbald B, Weiland S, Ait-Khaled N, Anabwani G, Anderson HR, et al. Worldwide variations in prevalence of symptoms of allergic rhinoconjunctivitis in children: the International Study of Asthma and Allergies in Childhood (ISAAC). Pediatr Allergy Immunol. 1997. 8:161–176.
Article3. Bachert C, Vignola AM, Gevaert P, Leynaert B, Van Cauwenberge P, Bousquet J. Allergic rhinitis, rhinosinusitis, and asthma: one airway disease. Immunol Allergy Clin North Am. 2004. 24:19–43.
Article4. Passalacqua G, Ciprandi G, Canonica GW. The nose-lung interaction in allergic rhinitis and asthma: united airways disease. Curr Opin Allergy Clin Immunol. 2001. 1:7–13.
Article5. Settipane RJ, Hagy GW, Settipane GA. Long-term risk factors for developing asthma and allergic rhinitis: a 23-year follow-up study of college students. Allergy Proc. 1994. 15:21–25.
Article6. Kocabas CN, Civelek E, Sackesen C, Orhan F, Tuncer A, Adalioglu G, et al. Burden of rhinitis in children with asthma. Pediatr Pulmonol. 2005. 40:235–240.
Article7. Braunstahl GJ, Overbeek SE, Kleinjan A, Prins JB, Hoogsteden HC, Fokkens WJ. Nasal allergen provocation induces adhesion molecule expression and tissue eosinophilia in upper and lower airways. J Allergy Clin Immunol. 2001. 107:469–476.
Article8. Reed CE, Marcoux JP, Welsh PW. Effects of topical nasal treatment on asthma symptoms. J Allergy Clin Immunol. 1988. 81(5 Pt 2):1042–1047.
Article9. Marcucci F, Passalacqua G, Canonica GW, Frati F, Salvatori S, Di cara G, et al. Lower airway inflammation before and after house dust mite nasal challenge: an age and allergen exposure-related phenomenon. Respir Med. 2007. 101:1600–1608.
Article10. Alving K, Weitzberg E, Lundberg JM. Increased amount of nitric oxide in exhaled air of asthmatics. Eur Respir J. 1993. 6:1368–1370.11. Strunk RC, Szefler SJ, Phillips BR, Zeiger RS, Chinchilli VM, Larsen G, et al. Relationship of exhaled nitric oxide to clinical and inflammatory markers of persistent asthma in children. J Allergy Clin Immunol. 2003. 112:883–892.
Article12. Scott M, Raza A, Karmaus W, Mitchell F, Grundy J, Kurukulaaratchy RJ, et al. Influence of atopy and asthma on exhaled nitric oxide in an unselected birth cohort study. Thorax. 2010. 65:258–262.
Article13. Hervas D, Milan JM, Garde J. Differences in exhaled nitric oxide in atopic children. Allergol Immunopathol (Madr). 2008. 36:331–335.
Article14. Cardinale F, de Benedictis FM, Muggeo V, Giordano P, Loffredo MS, Iacoviello G, et al. Exhaled nitric oxide, total serum IgE and allergic sensitization in childhood asthma and allergic rhinitis. Pediatr Allergy Immunol. 2005. 16:236–242.
Article15. Reijonen TM, Kotaniemi-Syrjanen A, Korhonen K, Korppi M. Predictors of asthma three years after hospital admission for wheezing in infancy. Pediatrics. 2000. 106:1406–1412.
Article16. American Thoracic Society. European Respiratory Society. ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am J Respir Crit Care Med. 2005. 171:912–930.17. Brown WG, Halonen MJ, Kaltenborn WT, Barbee RA. The relationship of respiratory allergy, skin test reactivity, and serum IgE in a community population sample. J Allergy Clin Immunol. 1979. 63:328–335.
Article18. Liu AH, Jaramillo R, Sicherer SH, Wood RA, Bock SA, Burks AW, et al. National prevalence and risk factors for food allergy and relationship to asthma: results from the National Health and Nutrition Examination Survey 2005-2006. J Allergy Clin Immunol. 2010. 126:798–806.e13.
Article19. Arnal JF, Didier A, Rami J, M'Rini C, Charlet JP, Serrano E, et al. Nasal nitric oxide is increased in allergic rhinitis. Clin Exp Allergy. 1997. 27:358–362.
Article20. Kharitonov SA, Barnes PJ. Exhaled markers of pulmonary disease. Am J Respir Crit Care Med. 2001. 163:1693–1722.
Article21. Zvezdin B, Milutinov S, Tanaskovic I, Kojicic M, Kolarov V, Hromis S, et al. The frequency of sensitization to inhalatory allergens and concomitant rhinitis in asthmatic patients. Vojnosanit Pregl. 2011. 68:309–313.
Article22. Caudri D, Wijga AH, Hoekstra MO, Kerkhof M, Koppelman GH, Brunekreef B, et al. Prediction of asthma in symptomatic preschool children using exhaled nitric oxide, Rint and specific IgE. Thorax. 2010. 65:801–807.
Article23. Chiron R, Vachier I, Khanbabaee G, Molinari N, Varrin M, Godard P, et al. Impact of rhinitis on asthma control in children: association with FeNO. J Asthma. 2010. 47:604–608.
Article24. Peroni DG, Piacentini GL, Alfonsi L, Zerman L, Di Blasi P, Visona' G, et al. Rhinitis in pre-school children: prevalence, association with allergic diseases and risk factors. Clin Exp Allergy. 2003. 33:1349–1354.
Article25. Navarro A, Valero A, Julia B, Quirce S. Coexistence of asthma and allergic rhinitis in adult patients attending allergy clinics: ONEAIR study. J Investig Allergol Clin Immunol. 2008. 18:233–238.26. Prieto L, Gutierrez V, Uixera S. Exhaled nitric oxide and bronchial responsiveness to adenosine 5'-monophosphate in subjects with allergic rhinitis. Chest. 2002. 121:1853–1859.
Article27. Avital A, Uwyyed K, Berkman N, Bar-Yishay E, Godfrey S, Springer C. Exhaled nitric oxide is age-dependent in asthma. Pediatr Pulmonol. 2003. 36:433–438.
Article28. Price D, Zhang Q, Kocevar VS, Yin DD, Thomas M. Effect of a concomitant diagnosis of allergic rhinitis on asthma-related health care use by adults. Clin Exp Allergy. 2005. 35:282–287.
Article29. Bousquet J, Gaugris S, Kocevar VS, Zhang Q, Yin DD, Polos PG, et al. Increased risk of asthma attacks and emergency visits among asthma patients with allergic rhinitis: a subgroup analysis of the investigation of montelukast as a partner agent for complementary therapy [corrected]. Clin Exp Allergy. 2005. 35:723–727.
Article30. Corren J, Manning BE, Thompson SF, Hennessy S, Strom BL. Rhinitis therapy and the prevention of hospital care for asthma: a case-control study. J Allergy Clin Immunol. 2004. 113:415–419.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Measurements of fractional exhaled nitric oxide in pediatric asthma
- Utility of Fractional Exhaled Nitric Oxide in the Diagnosis of Asthma and the Assessment of Asthma Control
- Clinical application of fractional exhaled nitric oxide in pediatric allergic rhinitis
- Changes in levels of fractional exhaled and nasal nitric oxide after treatment in allergic rhinitis
- Use of the exhaled nitric oxide for management of asthma and respiratory diseases