Nutr Res Pract.
2012 Oct;6(5):396-404.
Inhibitory effects of calcium against intestinal cancer in human colon cancer cells and ApcMin/+ mice
- Affiliations
-
- 1Department of Food and Nutrition, Chungbuk National University, 52 Naesudong-ro, Heungdeok-gu, Cheongju 361-763, Korea. jujih@chungbuk.ac.kr
- 2Susan Lehman Cullman Laboratory for Cancer Research, Department of Chemical Biology, Ernest Mario School of Pharmacy, Rutgers, The State University of New Jersey, Piscataway, New Jersey 08854, USA.
Abstract
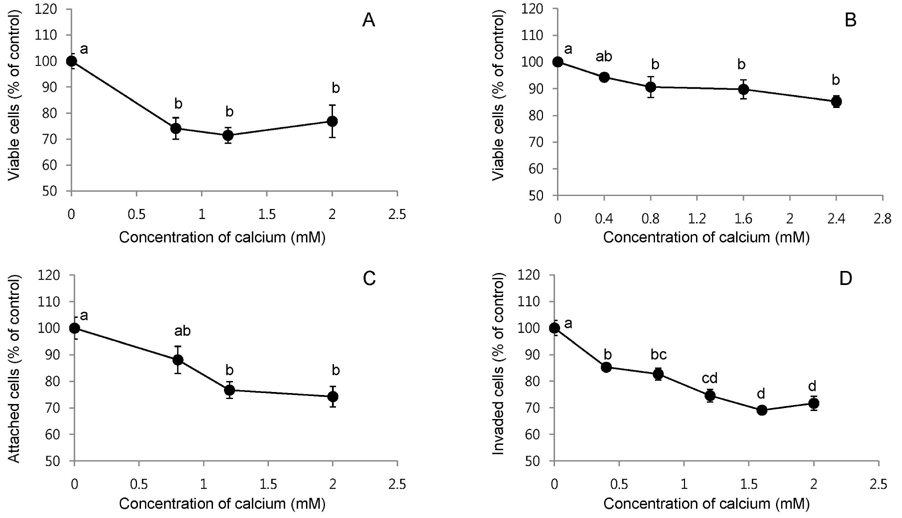
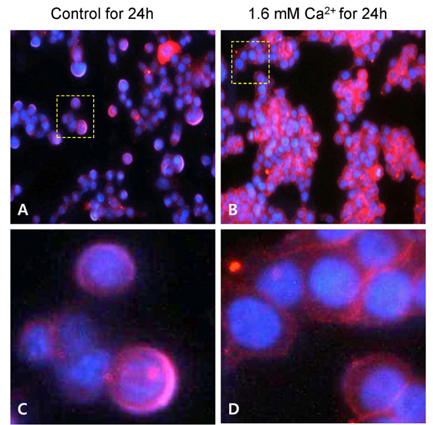
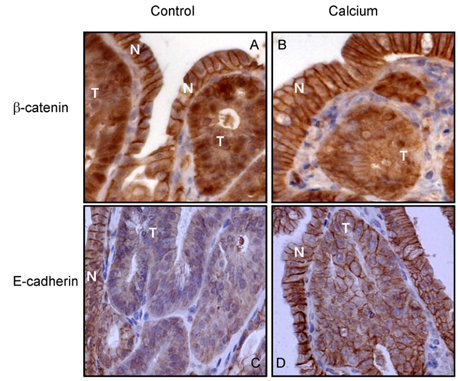
- The aim of the study was to investigate the inhibitory effects of calcium against intestinal cancer in vitro and in vivo. We first investigated the effects of calcium treatment in HCT116 and HT29 human colon cancer cells. At the concentration range of 0.8-2.4 mM, calcium significantly inhibited cell growth (by 9-29%), attachment (by 12-26%), invasion (by 15-31%), and migration (by 19-61%). An immunofluorescence microscope analysis showed that the treatment with calcium (1.6 mM) for 24 h increased plasma membrane beta-catenin but decreased nuclear beta-catenin levels in HT29 cells. We then investigated the effect of dietary calcium on intestinal tumorigenesis in ApcMin/+ mice. Mice received dietary treatment starting at 6 weeks of age for the consecutive 8 weeks. The basal control diet contained high-fat (20% mixed lipids by weight) and low-calcium (1.4 mg/g diet) to mimic the average Western diet, while the treatment diet contained an enriched level of calcium (5.2 mg calcium/g diet). The dietary calcium treatment decreased the total number of small intestinal tumors (by 31.4%; P < 0.05). The largest decrease was in tumors which were > or = 2 mm in diameter, showing a 75.6% inhibition in the small intestinal tumor multiplicity (P < 0.001). Immunohistochemical analysis showed significantly reduced nuclear staining of beta-catenin (expressed as nuclear positivity), but increased plasma membrane staining of beta-catenin, in the adenomas from the calcium-treated groups in comparison to those from the control group (P < 0.001). These results demonstrate intestinal cancer inhibitory effects of calcium both in human colon cancer cells and Apc Min/+ mice. The decreased beta-catenin nuclear localization caused by the calcium treatment may contribute to the inhibitory action.
Keyword
MeSH Terms
Figure
Reference
-
1. Boyle P, Levin B. World Cancer Report 2008. 2008. Geneva: WHO Press;11–55.2. McCullough ML, Giovannucci EL. Diet and cancer prevention. Oncogene. 2004. 23:6349–6364.
Article3. Straub DA. Calcium supplementation in clinical practice: a review of forms, doses, and indications. Nutr Clin Pract. 2007. 22:286–296.
Article4. Chia V, Newcomb PA. Calcium and colorectal cancer: some questions remain. Nutr Rev. 2004. 62:115–120.
Article5. Lamprecht SA, Lipkin M. Chemoprevention of colon cancer by calcium, vitamin D and folate: molecular mechanisms. Nat Rev Cancer. 2003. 3:601–614.
Article6. Half E, Arber N. Colon cancer: preventive agents and the present status of chemoprevention. Expert Opin Pharmacother. 2009. 10:211–219.
Article7. Carroll C, Cooper K, Papaioannou D, Hind D, Pilgrim H, Tappenden P. Supplemental calcium in the chemoprevention of colorectal cancer: a systematic review and meta-analysis. Clin Ther. 2010. 32:789–803.
Article8. Baron JA, Beach M, Mandel JS, van Stolk RU, Haile RW, Sandler RS, Rothstein R, Summers RW, Snover DC, Beck GJ, Bond JH, Greenberg ER. Calcium Polyp Prevention Study Group. Calcium supplements for the prevention of colorectal adenomas. N Engl J Med. 1999. 340:101–107.
Article9. Wallace K, Baron JA, Cole BF, Sandler RS, Karagas MR, Beach MA, Haile RW, Burke CA, Pearson LH, Mandel JS, Rothstein R, Snover DC. Effect of calcium supplementation on the risk of large bowel polyps. J Natl Cancer Inst. 2004. 96:921–925.
Article10. Kenar L, Karayilanoglu T, Aydin A, Serdar M, Kose S, Erbil MK. Protective effects of diets supplemented with omega-3 polyunsaturated fatty acids and calcium against colorectal tumor formation. Dig Dis Sci. 2008. 53:2177–2182.
Article11. Belbraouet S, Felden F, Pelletier X, Gastin I, Lambert D, Floquet J, Guéant JL, Debry G. Dietary calcium salts as protective agents and laminin P1 as a biochemical marker in chemically induced colon carcinogenesis in rats. Cancer Detect Prev. 1996. 20:294–299.12. Ranhotra GS, Gelroth JA, Glaser BK, Schoening P, Brown SE. Cellulose and calcium lower the incidence of chemically-induced colon tumors in rats. Plant Foods Hum Nutr. 1999. 54:295–303.13. Pence BC, Dunn DM, Zhao C, Landers M, Wargovich MJ. Chemopreventive effects of calcium but not aspirin supplementation in cholic acid-promoted colon carcinogenesis: correlation with intermediate endpoints. Carcinogenesis. 1995. 16:757–765.
Article14. Viñas-Salas J, Biendicho-Palau P, Piñol-Felis C, Miguelsanz-Garcia S, Perez-Holanda S. Calcium inhibits colon carcinogenesis in an experimental model in the rat. Eur J Cancer. 1998. 34:1941–1945.
Article15. Dwivedi C, Oredipe OA, Barth RF, Downie AA, Webb TE. Effects of the experimental chemopreventative agent, glucarate, on intestinal carcinogenesis in rats. Carcinogenesis. 1989. 10:1539–1541.
Article16. Wargovich MJ, Allnutt D, Palmer C, Anaya P, Stephens LC. Inhibition of the promotional phase of azoxymethane-induced colon carcinogenesis in the F344 rat by calcium lactate: effect of simulating two human nutrient density levels. Cancer Lett. 1990. 53:17–25.
Article17. Karkare MR, Clark TD, Glauert HP. Effect of dietary calcium on colon carcinogenesis induced by a single injection of 1,2-dimethylhydrazine in rats. J Nutr. 1991. 121:568–577.
Article18. Adell-Carceller R, Segarra-Soria M, Gibert-Jerez J, Salvador Sanchís JL, Lázaro-Santander R, Escrig-Sos J, Ruiz-Castillo J. Inhibitory effect of calcium on carcinogenesis at the site of colonic anastomosis: an experimental study. Dis Colon Rectum. 1997. 40:1376–1381.
Article19. Pereira MA, Barnes LH, Rassman VL, Kelloff GV, Steele VE. Use of azoxymethane-induced foci of aberrant crypts in rat colon to identify potential cancer chemopreventive agents. Carcinogenesis. 1994. 15:1049–1054.
Article20. Liu Z, Tomotake H, Wan G, Watanabe H, Kato N. Combined effect of dietary calcium and iron on colonic aberrant crypt foci, cell proliferation and apoptosis, and fecal bile acids in 1,2-dimethylhydrazine-treated rats. Oncol Rep. 2001. 8:893–897.
Article21. Mølck AM, Poulsen M, Meyer O. The combination of 1alpha,25 (OH2)-vitamin D3, calcium and acetylsalicylic acid affects azoxymethane-induced aberrant crypt foci and colorectal tumours in rats. Cancer Lett. 2002. 186:19–28.
Article22. Wargovich MJ, Jimenez A, McKee K, Steele VE, Velasco M, Woods J, Price R, Gray K, Kelloff GJ. Efficacy of potential chemopreventive agents on rat colon aberrant crypt formation and progression. Carcinogenesis. 2000. 21:1149–1155.
Article23. Li H, Kramer PM, Lubet RA, Steele VE, Kelloff GJ, Pereira MA. Effect of calcium on azoxymethane-induced aberrant crypt foci and cell proliferation in the colon of rats. Cancer Lett. 1998. 124:39–46.
Article24. Nelson RL, Tanure JC, Andrianopoulos G. The effect of dietary milk and calcium on experimental colorectal carcinogenesis. Dis Colon Rectum. 1987. 30:947–949.
Article25. Sitrin MD, Halline AG, Abrahams C, Brasitus TA. Dietary calcium and vitamin D modulate 1,2-dimethylhydrazine-induced colonic carcinogenesis in the rat. Cancer Res. 1991. 51:5608–5613.26. Beaty MM, Lee EY, Glauert HP. Influence of dietary calcium and vitamin D on colon epithelial cell proliferation and 1,2-dimethylhydrazine-induced colon carcinogenesis in rats fed high fat diets. J Nutr. 1993. 123:144–152.
Article27. Pence BC, Dunn DM, Zhao C, Patel V, Hunter S, Landers M. Protective effects of calcium from nonfat dried milk against colon carcinogenesis in rats. Nutr Cancer. 1996. 25:35–45.
Article28. Huerta S, Irwin RW, Heber D, Go VL, Moatamed F, Huerta S, Ou C, Harris DM. Intestinal polyp formation in the Apcmin mouse: effects of levels of dietary calcium and altered vitamin D homeostasis. Dig Dis Sci. 2003. 48:870–876.29. Ding S, McEntee MF, Whelan J, Zemel M. Adiposity-related protection of intestinal tumorigenesis: interaction with dietary calcium. Nutr Cancer. 2007. 58:153–161.
Article30. Newmark HL, Yang K, Lipkin M, Kopelovich L, Liu Y, Fan K, Shinozaki H. A Western-style diet induces benign and malignant neoplasms in the colon of normal C57Bl/6 mice. Carcinogenesis. 2001. 22:1871–1875.
Article31. Newmark HL, Yang K, Kurihara N, Fan K, Augenlicht LH, Lipkin M. Western-style diet-induced colonic tumors and their modulation by calcium and vitamin D in C57Bl/6 mice: a preclinical model for human sporadic colon cancer. Carcinogenesis. 2009. 30:88–92.
Article32. Liu Y, Ju J, Xiao H, Simi B, Hao X, Reddy BS, Huang MT, Newmark H, Yang CS. Effects of combination of calcium and aspirin on azoxymethane-induced aberrant crypt foci formation in the colons of mice and rats. Nutr Cancer. 2008. 60:660–665.
Article33. Van Aken E, De Wever O, Correia da Rocha AS, Mareel M. Defective E-cadherin/catenin complexes in human cancer. Virchows Arch. 2001. 439:725–751.
Article34. Hao X, Frayling IM, Willcocks TC, Han W, Tomlinson IP, Pignatelli MN, Pretlow TP, Talbot IC. Beta-catenin expression and allelic loss at APC in sporadic colorectal carcinogenesis. Virchows Arch. 2002. 440:362–366.
Article35. Hao X, Palazzo JP, Ilyas M, Tomlinson I, Talbot IC. Reduced expression of molecules of the cadherin/catenin complex in the transition from colorectal adenoma to carcinoma. Anticancer Res. 1997. 17:2241–2247.36. Bhagavathula N, Hanosh AW, Nerusu KC, Appelman H, Chakrabarty S, Varani J. Regulation of E-cadherin and beta-catenin by Ca2+ in colon carcinoma is dependent on calcium-sensing receptor expression and function. Int J Cancer. 2007. 121:1455–1462.
Article37. Tu CL, Chang W, Xie Z, Bikle DD. Inactivation of the calcium sensing receptor inhibits E-cadherin-mediated cell-cell adhesion and calcium-induced differentiation in human epidermal keratinocytes. J Biol Chem. 2008. 283:3519–3528.
Article38. Chakrabarty S, Radjendirane V, Appelman H, Varani J. Extracellular calcium and calcium sensing receptor function in human colon carcinomas: promotion of E-cadherin expression and suppression of beta-catenin/TCF activation. Cancer Res. 2003. 63:67–71.39. Shtutman M, Levina E, Ohouo P, Baig M, Roninson IB. Cell adhesion molecule L1 disrupts E-cadherin-containing adherens junctions and increases scattering and motility of MCF7 breast carcinoma cells. Cancer Res. 2006. 66:11370–11380.
Article40. Boivin GP, Washington K, Yang K, Ward JM, Pretlow TP, Russell R, Besselsen DG, Godfrey VL, Doetschman T, Dove WF, Pitot HC, Halberg RB, Itzkowitz SH, Groden J, Coffey RJ. Pathology of mouse models of intestinal cancer: consensus report and recommendations. Gastroenterology. 2003. 124:762–777.
Article41. Miyoshi Y, Nagase H, Ando H, Horii A, Ichii S, Nakatsuru S, Aoki T, Miki Y, Mori T, Nakamura Y. Somatic mutations of the APC gene in colorectal tumors: mutation cluster region in the APC gene. Hum Mol Genet. 1992. 1:229–233.
Article42. Corpet DE, Pierre F. Point: From animal models to prevention of colon cancer. Systematic review of chemoprevention in min mice and choice of the model system. Cancer Epidemiol Biomarkers Prev. 2003. 12:391–400.43. Ju J, Hong J, Zhou JN, Pan Z, Bose M, Liao J, Yang GY, Liu YY, Hou Z, Lin Y, Ma J, Shih WJ, Carothers AM, Yang CS. Inhibition of intestinal tumorigenesis in Apcmin/+ mice by (-)-epigallocatechin-3-gallate, the major catechin in green tea. Cancer Res. 2005. 65:10623–10631.
Article44. Sang S, Ju J, Lambert JD, Lin Y, Hong J, Bose M, Wang S, Bai N, He K, Reddy BS, Ho CT, Li F, Yang CS. Wheat bran oil and its fractions inhibit human colon cancer cell growth and intestinal tumorigenesis in Apc(min/+) mice. J Agric Food Chem. 2006. 54:9792–9797.
Article45. Hao X, Sun Y, Yang CS, Bose M, Lambert JD, Ju J, Lu G, Lee MJ, Park S, Husain A, Wang S. Inhibition of intestinal tumorigenesis in Apc(min/+) mice by green tea polyphenols (polyphenon E) and individual catechins. Nutr Cancer. 2007. 59:62–69.
Article46. Carafoli E. Intracellular calcium homeostasis. Annu Rev Biochem. 1987. 56:395–433.
Article47. Newmark HL, Heaney RP, Lachance PA. Should calcium and vitamin D be added to the current enrichment program for cereal-grain products? Am J Clin Nutr. 2004. 80:264–270.
Article48. Newmark HL. Nutrient density: an important and useful tool for laboratory animal studies. Carcinogenesis. 1987. 8:871–873.
Article49. Standing Committee on the Scientific Evaluation of Dietary Reference Intakes, Food and Nutrition Board, Institute of Medicine. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride. 1997. Washington, D.C: National Academy Press.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Withaferin-A Inhibits Colon Cancer Cell Growth by Blocking STAT3 Transcriptional Activity
- Regional Distribution and Relative Frequency of Gastrointestinal Endocrine Cells in Large Intestines of C57BL/6 Mice
- β-carotene regulates cancer stemness in colon cancer in vivo and in vitro
- Cancer-preventive Properties of an Anthocyanin-enriched Sweet Potato in the APCMIN Mouse Model
- A Case of Colon Cancer Associated with Colonic Tuberculosis