Nutr Res Pract.
2011 Oct;5(5):375-380.
Inorganic sulfur reduces the motility and invasion of MDA-MB-231 human breast cancer cells
- Affiliations
-
- 1Department of Food Science and Nutrition, Dankook University, 126 Jukjeon-dong, Suji-gu, Yongin-si, Gyunggi 448-701, Korea. wkkim@dankook.ac.kr
- 2Department of Food and Nutrition, Beawha Women's University, Seoul 110-735, Korea.
Abstract
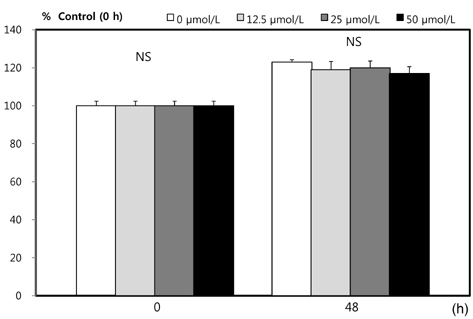
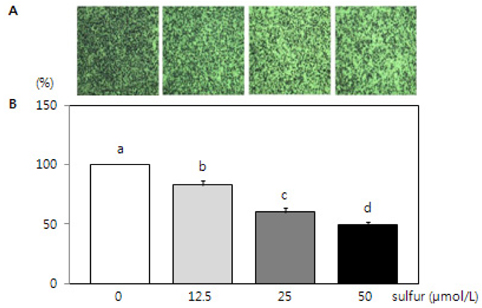
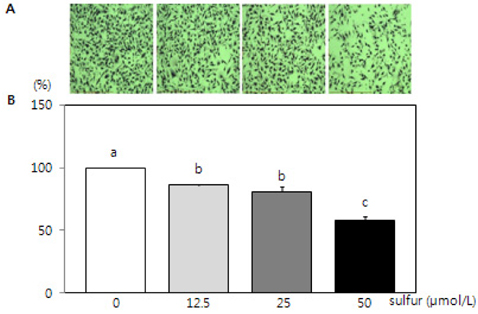
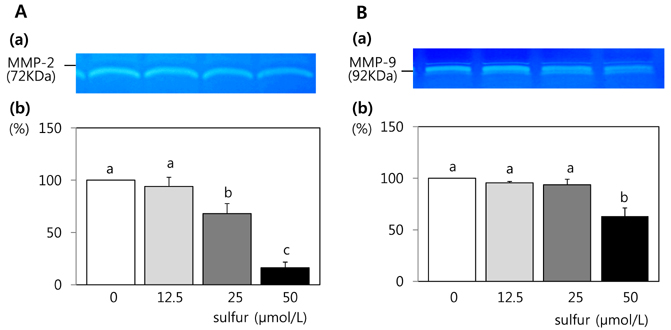
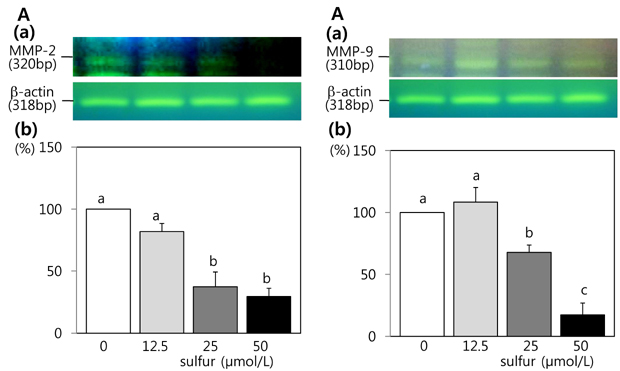
- This study investigated the effects of inorganic sulfur on metastasis in MDA-MB-231 human breast cancer cells. MDA-MB-231 cells were cultured in the absence or presence of various concentrations (12.5, 25, or 50 micromol/L) of inorganic sulfur. Cell motility, invasion, and the activity and mRNA expression of matrix metalloproteases (MMPs) were examined. Numbers of viable MDA-MB-231 cells did not differ by inorganic sulfur treatment from 0 to 50 micromol/L within 48 h. Inorganic sulfur significantly decreased cell motility and invasion in the MDA-MB-231 cells in a dose-dependent manner (P < 0.05), as determined using a Boyden chamber assay and a Matrigel chamber. The activities of MMP-2 and MMP-9 were significantly reduced by inorganic sulfur in a dose-dependent manner (P < 0.05). The inorganic sulfur also significantly inhibited MMP-2 and MMP-9 expression in the cells (P < 0.05). These data suggest that inorganic sulfur can suppress cancer cell motility and invasion by inhibiting MMP-2 and MMP-9 activity and gene expression in MDA-MB-231 cells.
MeSH Terms
Figure
Reference
-
1. Are the number of cancer cases increasing or decreasing in the world? World Health Organization [Internet]. cited 2010 June 3. Available from: http://www.who.int/features/qa/15/en/print.html.2. Analysis of cancer patients in 2008. National Health Insurance [Internet]. cited 2010 June 5. Available from: http://www.nhic.or.kr/cms/board/board/Board.jsp?act=VIEW&communityKey=B0070&boardId=20108.3. Types of breast cancer. National Cancer Center [Internet]. cited 2010 June 10. Available from: http://www.cancer.go.kr/cms/cancer/cancer_is/02/1191227_1751.html.4. Yip D, Ahmad A, Karapetis CS, Hawkins CA, Harper PG. Matrix metalloproteinase inhibitors: applications in oncology. Invest New Drugs. 1999. 17:387–399.5. Duffy MJ. The role of proteolytic enzymes in cancer invasion and metastasis. Clin Exp Metastasis. 1992. 10:145–155.
Article6. Iwata H, Kobayashi S, Iwase H, Masaoka A, Fujimoto N, Okada Y. Production of matrix metalloproteinases and tissue inhibitors of metalloproteinases in human breast carcinomas. Jpn J Cancer Res. 1996. 87:602–611.
Article7. Kim KW. Tumor metastasis and angiogenesis. J Life Sci. 1994. 4:19–29.8. Okegawa T, Pong RC, Li Y, Hsieh JT. The role of cell adhesion molecule in cancer progression and its application in cancer therapy. Acta Biochim Pol. 2004. 51:445–457.
Article9. Sin DI, Park JB, Park KK, Cho CH, Oh HK, Choi CH, Cho HJ, Jang YC. Inhibitory effects of type IV collagenase by disulfiram. J Life Sci. 2006. 16:964–971.
Article10. Structure of breast cancer. National Cancer Center [Internet]. cited 2010 June 10. Available from: http://www.cancer.go.kr/cms/cancer/cancer_is/01/1190409_1221.html.11. The Merck Index. Sulfur. 2001. 13th ed. Merck & Co., INC.12. Parcell S. Sulfur in human nutrition and applications in medicine. Altern Med Rev. 2002. 7:22–44.13. Ingenbleek Y. The nutritional relationship linking sulfur to nitrogen in living organisms. J Nutr. 2006. 136:1641S–1651S.
Article14. Michels FG, Smith JT. A comparison of the utilization of organic and inorganic sulfur by the rat. J Nutr. 1965. 87:217–220.
Article15. Sasse CE, Baker DH. Sulfur utilization by the chick with emphasis on the effect of inorganic sulfate on the cystine-methionine interrelationship. J Nutr. 1974. 104:244–251.
Article16. Pappa A, Franco R, Schoneveld O, Galanis A, Sandaltzopoulos R, Panayiotidis MI. Sulfur-containing compounds in protecting against oxidant-mediated lung diseases. Curr Med Chem. 2007. 14:2590–2596.
Article17. Gamet-Payrastre L, Li P, Lumeau S, Cassar G, Dupont MA, Chevolleau S, Gasc N, Tulliez J, Tercé F. Sulforaphane, a naturally occurring isothiocyanate, induces cell cycle arrest and apoptosis in HT29 human colon cancer cells. Cancer Res. 2000. 60:1426–1433.18. Singh AV, Xiao D, Lew KL, Dhir R, Singh SV. Sulforaphane induces caspase-mediated apoptosis in cultured PC-3 human prostate cancer cells and retards growth of PC-3 xenografts in vivo. Carcinogenesis. 2004. 25:83–90.
Article19. Kim EJ, Kang IJ, Cho HJ, Kim WK, Ha YL, Park JH. Conjugated linoleic acid down regulates insulin-like growth factor-1 receptor levels in HT-29 human colon cancer cells. J Nutr. 2003. 133:2675–2681.
Article20. Soel SM, Choi OS, Bang MH, Yoon Park JH, Kim WK. Influence of conjugated linoleic acid isomers on the metastasis of colon cancer cells in vitro and in vivo. J Nutr Biochem. 2007. 18:650–657.
Article21. Lee HS, Seo EY, Kang NE, Kim WK. [6]-Gingerol inhibits metastasis of MDA-MB-231 human breast cancer cells. J Nutr Biochem. 2008. 19:313–319.
Article22. Na MH, Seo EY, Kim WK. Effects of α-lipoic acid on cell proliferation and apoptosis in MDA-MB-231 human breast cells. Nutr Res Pract. 2009. 3:265–271.
Article23. Wu X, Zhu Y, Yan H, Liu B, Li Y, Zhou Q, Xu K. Isothiocyanates induce oxidative stress and suppress the metastasis potential of human non-small cell lung cancer cells. BMC Cancer. 2010. 10:269.
Article24. Ho CC, Lai KC, Hsu SC, Kuo CL, Ma CY, Lin ML, Yang JS, Chung JG. Benzyl isothiocyanate (BITC) inhibits migration and invasion of human gastric cancer AGS cells via suppressing ERK signal pathways. Hum Exp Toxicol. 2011. 30:296–306.
Article25. Hwang ES, Lee HJ. Allyl isothiocyanate and its N-acetylcysteine conjugate suppress metastasis via inhibition of invasion, migration, and matrix metalloproteinase-2/-9 activities in SK-Hep 1 human hepatoma cells. Exp Biol Med (Maywood). 2006. 231:421–430.
Article26. Folkman J, Haudenschild C. Angiogenesis in vitro. Nature. 1980. 288:551–556.
Article27. Woessner JF Jr. Matrix metalloproteinases and their inhibitors in connective tissue remodeling. FASEB J. 1991. 5:2145–2154.
Article28. Werb Z, Vu TH, Rinkenberger JL, Coussens LM. Matrix-degrading proteases and angiogenesis during development and tumor formation. APMIS. 1999. 107:11–18.
Article29. Murphy G, Reynolds JJ, Hembry RM. Metalloproteinases and cancer invasion and metastasis. Int J Cancer. 1989. 44:757–760.30. Kawamoto T, Sato JD, Le A, Polikoff J, Sato GH, Mendelsohn J. Growth stimulation of A431 cells by epidermal growth factor: identification of high-affinity receptors for epidermal growth factor by an anti-receptor monoclonal antibody. Proc Natl Acad Sci U S A. 1983. 80:1337–1341.
Article31. Ura H, Bonfil RD, Reich R, Reddel R, Pfeifer A, Harris CC, Klein-Szanto AJ. Expression of type IV collagenase and procollagen genes and its correlation with the tumorigenic, invasive, and metastatic abilities of oncogene-transformed human bronchial epithelial cells. Cancer Res. 1989. 49:4615–4621.32. Liotta LA, Stetler-Stevenson WG. Tumor invasion and metastasis: an imbalance of positive and negative regulation. Cancer Res. 1991. 51:5054s–5059s.33. Tryggvason K, Höyhtyä M, Pyke C. Type IV collagenases in invasive tumors. Breast Cancer Res Treat. 1993. 24:209–218.
Article34. Davies B, Waxman J, Wasan H, Abel P, Williams G, Krausz T, Neal D, Thomas D, Hanby A, Balkwill F. Levels of matrix metalloproteases in bladder cancer correlate with tumor grade and invasion. Cancer Res. 1993. 53:5365–5369.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inorganic sulfur reduces cell proliferation by inhibiting of ErbB2 and ErbB3 protein and mRNA expression in MDA-MB-231 human breast cancer cells
- Effect of Curcumin on Cancer Invasion and Matrix Metalloproteinase-9 Activity in MDA-MB-231 Human Breast Cancer Cell
- Effects of Epigallocatechin Gallate on Adhesion, Invasion and Matrix Metalloproteinase Activity in MDA-MB-231 Human Breast Cancer Cells
- Effect of Cyanidin on Cell Motility and Invasion in MDA-MB-231 Human Breast Cancer Cells
- Effect of corosolic acid on apoptosis and angiogenesis in MDA-MB-231 human breast cancer cells