Nutr Res Pract.
2010 Jun;4(3):177-182.
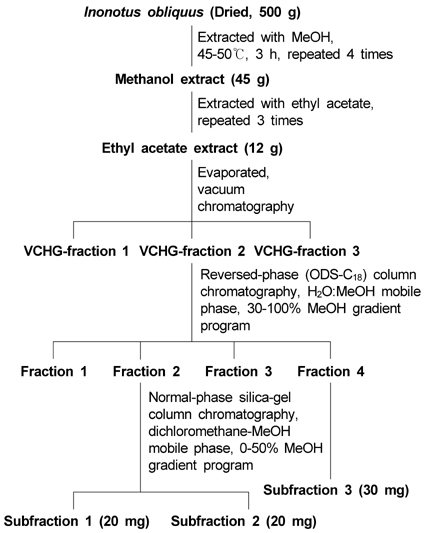
Anticancer activity of subfractions containing pure compounds of Chaga mushroom (Inonotus obliquus) extract in human cancer cells and in Balbc/c mice bearing Sarcoma-180 cells
- Affiliations
-
- 1Department of Food Science and Biotechnology, School of Biotechnology and Bioengineering, Kangwon National University, 192-1 Hyoja-dong, Chuncheon 200-701, Korea. mijachung@kangwon.ac.kr
- 2Department of Food and Nutrition, Hallym University, Chuncheon 200-702, Korea.
- 3Department Food Science and Nutrition, Dankook University, Gyunggi 448-701, Korea.
Abstract
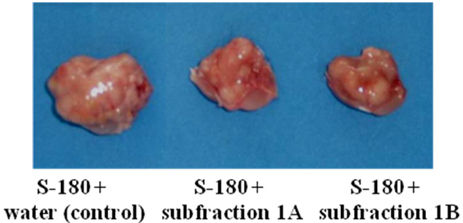
- The Chaga mushroom (Inonotus obliquus) has been used in folk medicine to treat cancers. However, limited information exists on the underlying anticancer effects of the major component of I. obliquus in vivo. We hypothesize that the pure compounds (3beta-hydroxy-lanosta-8,24-dien-21-al, inotodiol and lanosterol, respectively) separated from I. obliquus would inhibit tumor growth in Balbc/c mice bearing Sarcoma-180 cells (S-180) in vivo and growth of human carcinoma cells in vitro. To test this hypothesis, the growth inhibition of each subfraction isolated from I. obliquus on human carcinoma cell lines (lung carcinoma A-549 cells, stomach adenocarcinoma AGS cells, breast adenocarcinoma MCF-7 cells, and cervical adenocarcinoma HeLa cells) was tested in vitro. Then, after S-180 implantation, the mice were fed a normal chow supplemented with 0, 0.1 or 0.2 mg of subfraction 1, 2 or 3 per mouse per day. All of the subfractions isolated from I. obliquus showed significant cytotoxic activity against the selected cancer cell lines in vitro. Subfraction 1 was more active than subfraction 2 and subfraction 3 against the A549, AGS and MCF-7 cancer cell lines in vitro. In in vivo results, subfraction 1 isolated from I. obliquus at concentrations of 0.1 and 0.2 mg/mouse per day significantly decreased tumor volume by 23.96% and 33.71%, respectively, as compared with the control. Subfractions 2 and 3 also significantly inhibited tumor growth in mice bearing S-180 as compared with the control mouse tumor. Subfraction 1 isolated from I. obliquus showed greater inhibition of tumor growth than subfractions 2 and 3, which agrees well with the in vitro results. The results suggest that I. obliquus and its compounds in these subfractions isolated from I. obliquus could be used as natural anticancer ingredients in the food and/or pharmaceutical industry.
Keyword
MeSH Terms
Figure
Reference
-
1. Sporn MB. Prevention of cancer in the next millennium: report of the chemoprevention working group to the American association for cancer research. Cancer Res. 1999. 59:4743–4758.2. Roland JF, Chmielewicz ZF, Weiner BA, Gross AM, Boening OP, Luck JV, Bardos TJ, Reilly HC, Sugiura K, Stock CC, Lucas EH, Byerrum RU, Scevena JA. Calvacin: A new antitumor agent. Science. 1960. 132:1897.
Article3. Gregory FJ, Healy EM, Agersborg HP Jr, Warren GH. Studies on antitumor substances produced by Basidiomycetes. Mycologia. 1966. 58:80–90.
Article4. Saar M. Fungi in Khanty folk medicine. J Ethnopharmacol. 1991. 31:175–179.
Article5. Ham SS, Oh SW, Kim YK, Shin KS, Chang HY, Chung GH. Antimutagenic and cytotoxic effects of ethanol extract from the Inonotus obliquus. J Korean Soc Food Sci Nutr. 2003. 32:1088–1094.6. Ham SS, Oh SW, Kim YK, Shin KS, Chang HY, Chung GH. Antioxidant and genotoxic inhibition activity of ethanol extract from the Inonotus obliquus. J Korean Soc Food Sci Nutr. 2003. 32:1071–1075.7. Kahlos K, Hiltunen R, Schantz MV. 3β-Hydroxy-lanosta-8,24-dien-21-al, a new triterpene from Inontus obliquus. Planta Med. 1984. 50:197–198.
Article8. Ichmura T, Watanave O, Maruyama S. Inhibition of HIV-1 protease by water-soluble lignin-like substance from an edible mushroom Fuscoporia obliqua. Biosci Biotechnol Biochem. 1998. 62:575–577.9. Nakata T, Yamada T, Taji S, Ohishi H, Wada SI, Tokuda H, Sakuma K, Tanaka R. Structure determination of inonotsuoxides A and B and in vivo anti-tumor promoting activity of inotodiol from the sclerotia of Inonotus obliquus. Bioorg Med Chem. 2007. 15:257–264.
Article10. Lee SH, Hwang HS, Yun JW. Antitumor activity of water extract of a mushroom, Inonotus obliquus, against HT-29 human colon cancer cells. Phytother Res. 2009. 23:1784–1789.
Article11. Ham SS, Kim SH, Moon SY, Chung MJ, Cui CB, Han EK, Chung CK, Choe M. Antimutagenic effects of subfractions of Chaga mushroom (Inonotus obliquus) extract. Mutat Res. 2009. 672:55–59.
Article12. Chung MJ, Walker PA, Brown RW, Hogstrand C. Zinc-mediated gene expression offers protection against H2O2-induced cytotoxicity. Toxicol Appl Pharmacol. 2005. 205:225–236.
Article13. Ham YA, Choi HJ, Kim SH, Chung MJ, Ham SS. Antimutagenic and antitumor effects of Adenophora triphylla extracts. J Korean Soc Food Sci Nutr. 2009. 38:25–31.14. Maeda N, Kokai Y, Ohtani S, Hada T, Yoshida H, Mizushina Y. Inhibitory effects of preventive and curative orally administered spinach glycoglycerolipid fraction on the tumor growth of sarcoma and colon in mouse graft models. Food Chem. 2009. 112:205–210.
Article15. Oh HT, Chung MJ, Kim SH, Choi HJ, Ham SS. Masou salmon (Oncorhynchus masou) ethanol extract decrease 3-hydroxy-3-methylglutaryl coenzyme A reductase expression in diet-induced obese mice. Nutr Res. 2009. 29:123–129.
Article16. Kahlos K, Hiltunen R. Identification of some lanostane type triterpenes from Inonotus obliquus. Acta Pharmaceutica Fennica. 1983. 92:220–222.17. Mizuno T, Zhuang C, Abe K, Okamoto H, Kiho T, Ukai S, Leclerc S, Meijer L. Antitumor and hypoglycemic activities of polysaccharides from the Sclerotia and Mycelia of Inonotus obliquus (Pers: Fe.) Pll. (Aphyllophoromycetideae). International Journal of Medicinal Mushrooms. 1999. 1:301–316.
Article18. Kahlos T, Zhuangas L, Hitunen R. Antitumor activity of some compounds and fractions from an n-hexene extract of Inonotus obliquus. Acta Pharmaceutica Fennica. 1987. 96:33–40.19. Nomura M, Takahashi T, Uesugi A, Tanaka R, Kobayashi S. Inotodiol, a lanostane triterpenoid, from Inonotus obliquus inhibits cell proliferation through caspase-3-dependent apoptosis. Anticancer Res. 2008. 28:2691–2696.20. Youn MJ, Kim JK, Park SY, Kim Y, Park C, Kim ES, Park Kl, So HS, Park R. Potential anticancer properties of the water extract of lnonotus obliquus by induction of apoptosis in melanoma B16-F10 cells. J Ethnopharmacol. 2009. 121:221–228.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Immunomodulatory Activity of the Water Extract from Medicinal Mushroom Inonotus obliquus
- Ethanol extract of Innotus obliquus (Chaga mushroom) induces G1 cell cycle arrest in HT-29 human colon cancer cells
- Effects of Inonotus Obliqua Extract on Blood Glucose Levels in Genetically Diabetic Mice
- Effect of Inonotus Obliques Extracts on Proliferation and Caspase-3 Activity in Human Castro-Intestinal Cancer Cell Lines
- Antitumor and Immunostimulating Activities of Elfvingia applanata Hot Water Extract on Sarcoma 180 Tumor-bearing ICR Mice