Korean Circ J.
2011 Oct;41(10):590-595. 10.4070/kcj.2011.41.10.590.
Angiopoietin-1 Gene Therapy Attenuates Hypertension and Target Organ Damage in Nitric Oxide Synthase Inhibited Spontaneously Hypertensive Rats
- Affiliations
-
- 1College of Pharmacy, Ajou University, Suwon, Korea.
- 2Department of Medicine, Samsung Medical Center, Samsung Biomedical Research Institute, Sungkyunkwan University School of Medicine, Seoul, Korea. dkkim@skku.edu
- 3Biomedical Research Center and Department of Biological Sciences, Korea Advanced Institute of Science and Technology, Daejeon, Korea.
- KMID: 2297918
- DOI: http://doi.org/10.4070/kcj.2011.41.10.590
Abstract
- BACKGROUND AND OBJECTIVES
In our previous study, we found that the gene transfer of a potent derivative of cartilage oligomeric matrix protein Angiopoietin-1 (COMP-Ang-1) substantially prevented hypertension, microvascular rarefaction, and target organ damage in spontaneously hypertensive rats (SHRs). The purpose of the present study was to examine the role of nitric oxide (NO) in the therapeutic effects observed after COMP-Ang-1 gene transfer.
MATERIALS AND METHODS
To exclude the NO-mediated effects in COMP-Ang-1 gene therapy, the SHRs were treated with an NO synthase (NOS) inhibitor, Nw-nitro-L-arginine methyl ester (L-NAME) before the electrophoretic gene transfer.
RESULTS
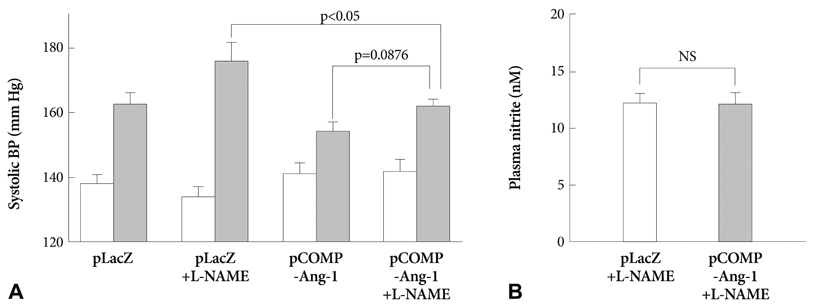
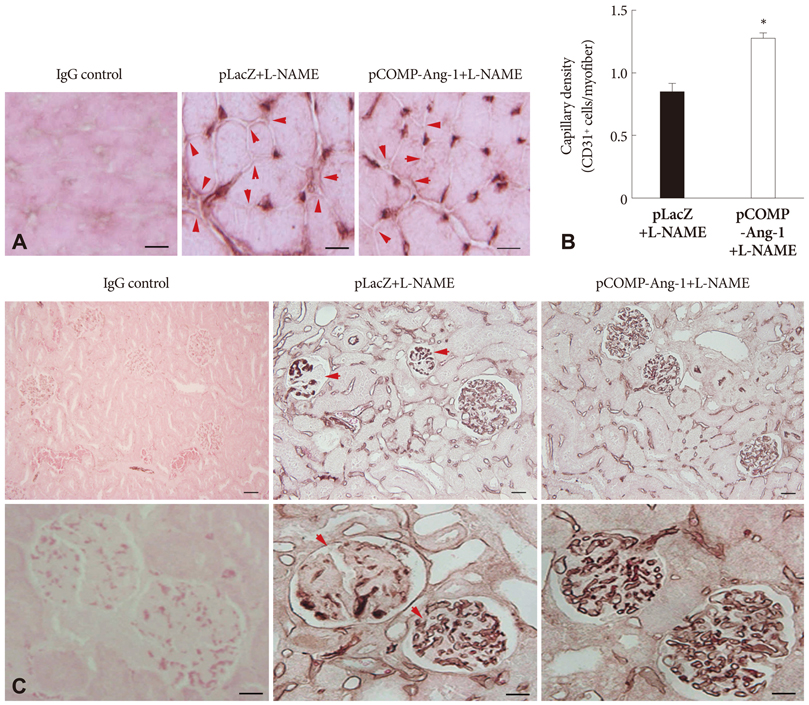
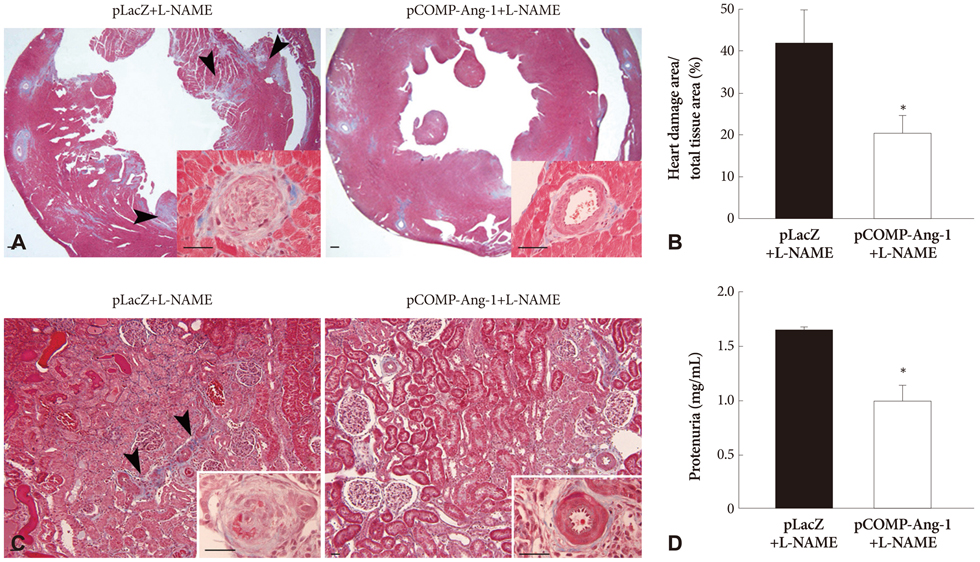
The pretreatment with L-NAME induced a severe and sustained increase in systolic blood pressure (BP) in a LacZ plasmid transferred control SHR. However, the electrophoretic transfer of a COMP-Ang-1 plasmid instead of LacZ plasmid in L-NAME-pretreated SHRs substantially blocked the development of hypertension without any significant difference in comparison with L-NAME-untreated COMP-Ang-1 plasmid transferred groups. In addition, the COMP-Ang-1 plasmid transfer substantially attenuated microvascular rarefaction and arteriole remodeling in the heart and kidney, which might account for the mild histological alterations observed in the COMP-Ang-1 plasmid transferred group, in contrast to the severe fibrosis and necrosis seen in the LacZ plasmid controls.
CONCLUSION
These therapeutic outcomes of COMP-Ang-1 gene transfer even in NOS inhibited SHRs suggested that the antihypertensive effect of COMP-Ang-1 was not merely secondary to NO-mediated vasorelaxation, but it may be associated with its ability to protect the vascular endothelium probably via an NO-independent mechanism which serves to attenuate microvascular rarefaction and target organ damage, and also to prevent hypertension by reducing peripheral vascular resistance.
Keyword
MeSH Terms
-
Angiopoietin-1
Arterioles
Blood Pressure
Cartilage
Endothelium
Endothelium, Vascular
Extracellular Matrix Proteins
Fibrosis
Genetic Therapy
Glycoproteins
Heart
Hypertension
Kidney
Necrosis
NG-Nitroarginine Methyl Ester
Nitric Oxide
Nitric Oxide Synthase
Plasmids
Rats, Inbred SHR
Vascular Resistance
Vasodilation
Angiopoietin-1
Extracellular Matrix Proteins
Glycoproteins
NG-Nitroarginine Methyl Ester
Nitric Oxide
Nitric Oxide Synthase
Figure
Reference
-
1. Cohuet G, Struijker-Boudier H. Mechanisms of target organ damage caused by hypertension: therapeutic potential. Pharmacol Ther. 2006. 111:81–98.2. Levy BI, Ambrosio G, Pries AR, Struijker-Boudier HA. Microcirculation in hypertension: a new target for treatment? Circulation. 2001. 104:735–740.3. Mourad JJ, Laville M. Is hypertension a tissue perfusion disorder? Implications for renal and myocardial perfusion. J Hypertens Suppl. 2006. 24:S10–S16.4. Battegay EJ, de Miguel LS, Petrimpol M, Humar R. Effects of anti-hypertensive drugs on vessel rarefaction. Curr Opin Pharmacol. 2007. 7:151–157.5. Redon J. Antihypertensive treatment: should it be titrated to blood pressure reduction or to target organ damage regression? Curr Opin Nephrol Hypertens. 2005. 14:448–452.6. Lee JS, Song SH, Kim JM, et al. Angiopoietin-1 prevents hypertension and target organ damage through its interaction with endothelial Tie2 receptor. Cardiovasc Res. 2008. 78:572–580.7. Olzinski AR, McCafferty TA, Zhao SQ, et al. Hypertensive target organ damage is attenuated by a p38 MAPK inhibitor: role of systemic blood pressure and endothelial protection. Cardiovasc Res. 2005. 66:170–178.8. Nakamura Y, Ono H, Zhou X, Frohlich ED. Angiotensin type 1 receptor antagonism and ACE inhibition produce similar renoprotection in N(omega)-nitro-L>-arginine methyl ester/spontaneously hypertensive rats. Hypertension. 2001. 37:1262–1267.9. Kubis N, Besnard S, Silvestre JS, et al. Decreased arteriolar density in endothelial nitric oxide synthase knockout mice is due to hypertension, not to the constitutive defect in endothelial nitric oxide synthase enzyme. J Hypertens. 2002. 20:273–280.10. Kubis N, Richer C, Domergue V, Giudicelli JF, Lévy BI. Role of microvascular rarefaction in the increased arterial pressure in mice lacking for the endothelial nitric oxide synthase gene (eNOS3pt-/-). J Hypertens. 2002. 20:1581–1587.11. Kim W, Moon SO, Lee SY, et al. COMP-angiopoietin-1 ameliorates renal fibrosis in a unilateral ureteral obstruction model. J Am Soc Nephrol. 2006. 17:2474–2483.12. Cho CH, Kammerer RA, Lee HJ, et al. Designed angiopoietin-1 variant, COMP-Ang1, protects against radiation-induced endothelial cell apoptosis. Proc Natl Acad Sci U S A. 2004. 101:5553–5558.13. Cho CH, Sung HK, Kim KT, et al. COMP-angiopoietin-1 promotes wound healing through enhanced angiogenesis, lymphangiogenesis, and blood flow in a diabetic mouse model. Proc Natl Acad Sci U S A. 2006. 103:4946–4951.14. Kobori H, Ozawa Y, Suzaki YY, Nishiyama A. Enhanced intrarenal angiotensinogen contributes to early renal injury in spontaneously hypertensive rats. J Am Soc Nephrol. 2005. 16:2073–2080.15. Du WM, Miao CY, Liu JG, Shen FM, Yang XQ, Su DF. Effects of long-term treatment with ketanserin on blood pressure variability and end-organ damage in spontaneously hypertensive rats. J Cardiovasc Pharmacol. 2003. 41:233–239.16. Noon JP, Walker BR, Webb DJ, et al. Impaired microvascular dilatation and capillary rarefaction in young adults with a predisposition to high blood pressure. J Clin Invest. 1997. 99:1873–1879.17. Antonios TF, Singer DR, Markandu ND, Mortimer PS, MacGregor GA. Rarefaction of skin capillaries in borderline essential hypertension suggests an early structural abnormality. Hypertension. 1999. 34:655–658.18. Antonios TF, Rattray FM, Singer DR, Markandu ND, Mortimer PS, MacGregor GA. Rarefaction of skin capillaries in normotensive offspring of individuals with essential hypertension. Heart. 2003. 89:175–178.19. Jung AD, Kim W, Park SH, et al. The effect of telmisartan on endothelial function and arterial stiffness in patients with essential hypertension. Korean Circ J. 2009. 39:180–184.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Aminoguanidine on Norepinephrine-Induced Vascular Contraction in Renovascular Hypertensive Rats
- Altered renal expression of nitric oxide synthase isozymes in spontaneously hypertensive rats
- Distribution of Nitric Oxide Synthase Isoforms in Perioral Exocrine Glands in Rats
- Involvement of Fibronectin in the Migration of Macrophage and Expression of Nitric Oxide Synthase in the BCG induced Inflammatory Sites in Rat Bladder
- Increased expression of nitric oxide synthase coincides with reversal of renovascular hypertension