Korean J Physiol Pharmacol.
2014 Dec;18(6):509-516. 10.4196/kjpp.2014.18.6.509.
Distinct Cellular Calcium Metabolism in Radiation-sensitive RKO Human Colorectal Cancer Cells
- Affiliations
-
- 1Department of Physiology and Biophysics, Inha University College of Medicine, Incheon 401-751, Korea. cksuh@inha.ac.kr
- 2Research Group of Food Functionality, Korea Food Research Institute, Seongnam 463-746, Division of Food Biotechnology, Korea University of Science and Technology, Daejeon 305-350, Korea.
- 3Department of Radiation Oncology, Hanyang University College of Medicine, Seoul 133-791, Korea.
- KMID: 2285554
- DOI: http://doi.org/10.4196/kjpp.2014.18.6.509
Abstract
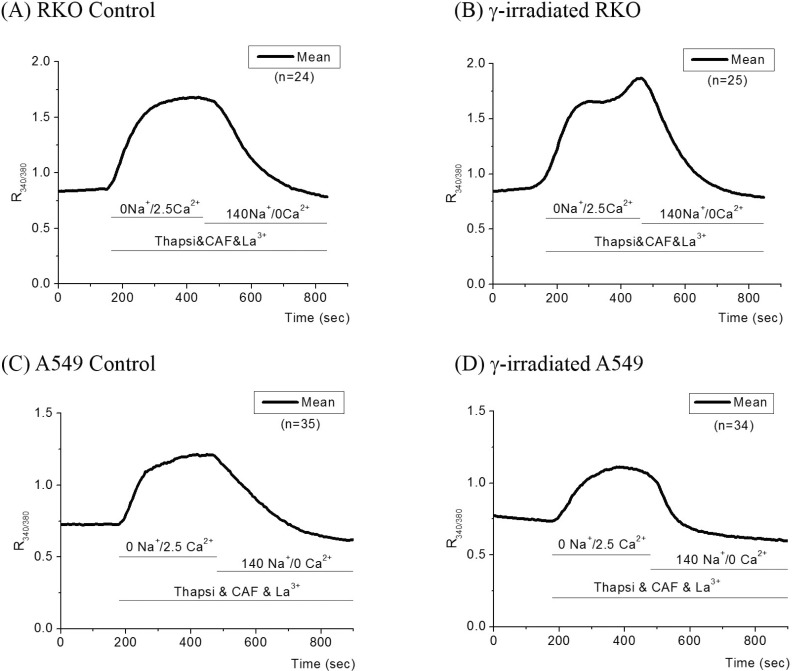
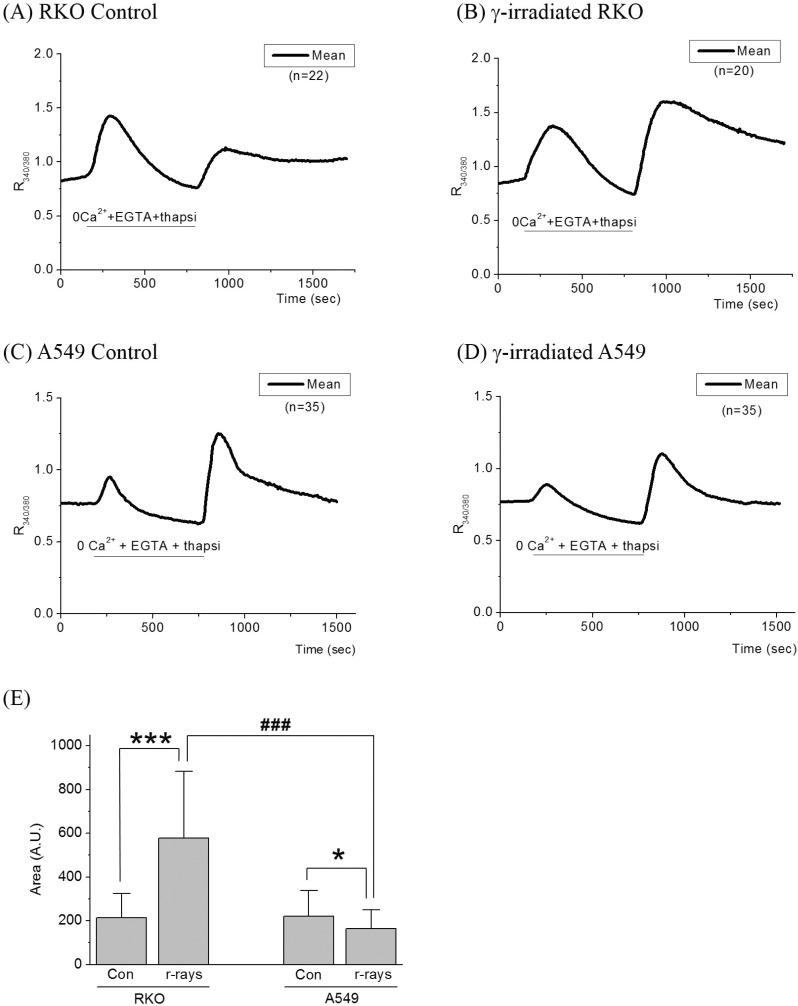
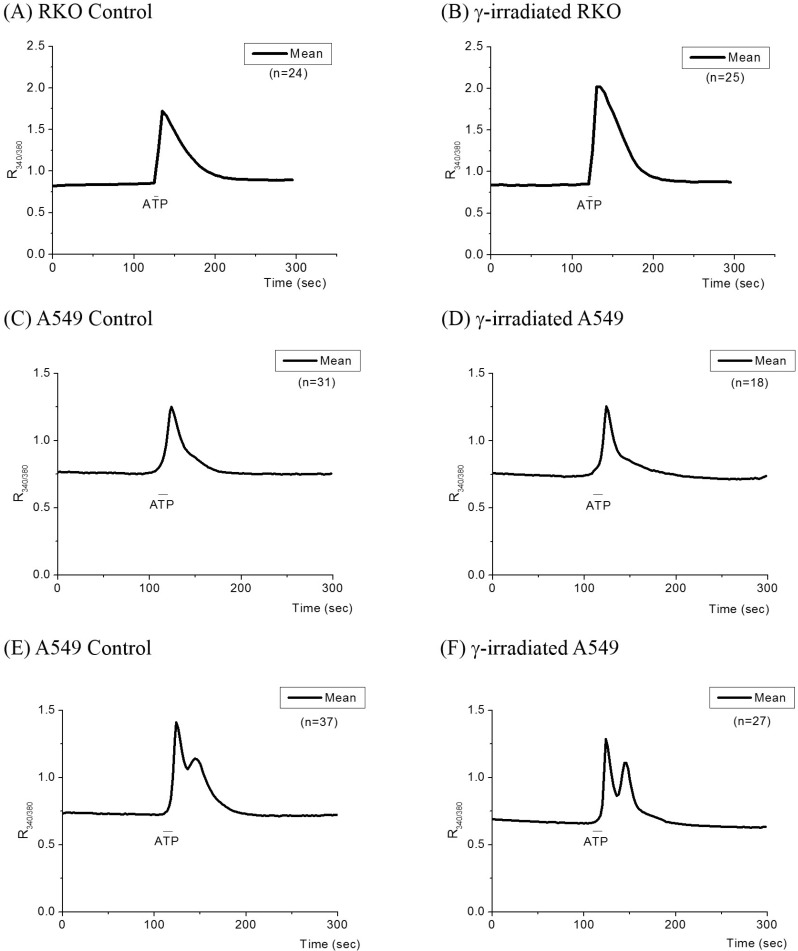
- Radiation therapy for variety of human solid tumors utilizes mechanism of cell death after DNA damage caused by radiation. In response to DNA damage, cytochrome c was released from mitochondria by activation of pro-apoptotic Bcl-2 family proteins, and then elicits massive Ca2+ release from the ER that lead to cell death. It was also suggested that irradiation may cause the deregulation of Ca2+ homeostasis and trigger programmed cell death and regulate death specific enzymes. Thus, in this study, we investigated how cellular Ca2+ metabolism in RKO cells, in comparison to radiation-resistant A549 cells, was altered by gamma (gamma)-irradiation. In irradiated RKO cells, Ca2+ influx via activation of NCX reverse mode was enhanced and a decline of [Ca2+]i via forward mode was accelerated. The amount of Ca2+ released from the ER in RKO cells by the activation of IP3 receptor was also enhanced by irradiation. An increase in [Ca2+]i via SOCI was enhanced in irradiated RKO cells, while that in A549 cells was depressed. These results suggest that gamma-irradiation elicits enhancement of cellular Ca2+ metabolism in radiation-sensitive RKO cells yielding programmed cell death.
Keyword
MeSH Terms
Figure
Reference
-
1. Miyashita T, Reed JC. Tumor suppressor p53 is a direct transcriptional activator of the human bax gene. Cell. 1995; 80:293–299. PMID: 7834749.
Article2. Oda E, Ohki R, Murasawa H, Nemoto J, Shibue T, Yamashita T, Tokino T, Taniguchi T, Tanaka N. Noxa, a BH3-only member of the Bcl-2 family and candidate mediator of p53-induced apoptosis. Science. 2000; 288:1053–1058. PMID: 10807576.
Article3. Nakano K, Vousden KH. PUMA, a novel proapoptotic gene, is induced by p53. Mol Cell. 2001; 7:683–694. PMID: 11463392.
Article4. Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES, Wang X. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell. 1997; 91:479–489. PMID: 9390557.5. Adams JM, Cory S. The Bcl-2 protein family: arbiters of cell survival. Science. 1998; 281:1322–1326. PMID: 9735050.
Article7. Hajnóczky G, Csordás G, Madesh M, Pacher P. Control of apoptosis by IP(3) and ryanodine receptor driven calcium signals. Cell Calcium. 2000; 28:349–363. PMID: 11115374.8. Pinton P, Ferrari D, Rapizzi E, Di Virgilio F, Pozzan T, Rizzuto R. A role for calcium in Bcl-2 action? Biochimie. 2002; 84:195–201. PMID: 12022950.
Article9. Green DR, Kroemer G. The pathophysiology of mitochondrial cell death. Science. 2004; 305:626–629. PMID: 15286356.
Article10. Pinton P, Rizzuto R. Bcl-2 and Ca2+ homeostasis in the endoplasmic reticulum. Cell Death Differ. 2006; 13:1409–1418. PMID: 16729032.11. Baffy G, Miyashita T, Williamson JR, Reed JC. Apoptosis induced by withdrawal of interleukin-3 (IL-3) from an IL-3-dependent hematopoietic cell line is associated with repartitioning of intracellular calcium and is blocked by enforced Bcl-2 oncoprotein production. J Biol Chem. 1993; 268:6511–6519. PMID: 8454620.
Article12. Pinton P, Ferrari D, Magalhães P, Schulze-Osthoff K, Di Virgilio F, Pozzan T, Rizzuto R. Reduced loading of intracellular Ca2+ stores and downregulation of capacitative Ca2+ influx in Bcl-2-overexpressing cells. J Cell Biol. 2000; 148:857–862. PMID: 10704437.13. Foyouzi-Youssefi R, Arnaudeau S, Borner C, Kelley WL, Tschopp J, Lew DP, Demaurex N, Krause KH. Bcl-2 decreases the free Ca2+ concentration within the endoplasmic reticulum. Proc Natl Acad Sci U S A. 2000; 97:5723–5728. PMID: 10823933.14. Berridge MJ, Lipp P, Bootman MD. The versatility and universality of calcium signalling. Nat Rev Mol Cell Biol. 2000; 1:11–21. PMID: 11413485.
Article15. Carafoli E, Santella L, Branca D, Brini M. Generation, control, and processing of cellular calcium signals. Crit Rev Biochem Mol Biol. 2001; 36:107–260. PMID: 11370791.
Article16. Trebak M, Bird GS, McKay RR, Putney JW Jr. Comparison of human TRPC3 channels in receptor-activated and store-operated modes. Differential sensitivity to channel blockers suggests fundamental differences in channel composition. J Biol Chem. 2002; 277:21617–21623. PMID: 11943785.17. Berridge MJ, Bootman MD, Roderick HL. Calcium signalling: dynamics, homeostasis and remodelling. Nat Rev Mol Cell Biol. 2003; 4:517–529. PMID: 12838335.
Article18. Kirichok Y, Krapivinsky G, Clapham DE. The mitochondrial calcium uniporter is a highly selective ion channel. Nature. 2004; 427:360–364. PMID: 14737170.
Article19. Rizzuto R, Pozzan T. Microdomains of intracellular Ca2+: molecular determinants and functional consequences. Physiol Rev. 2006; 86:369–408. PMID: 16371601.20. Roderick HL, Cook SJ. Ca2+ signalling checkpoints in cancer: remodelling Ca2+ for cancer cell proliferation and survival. Nat Rev Cancer. 2008; 8:361–375. PMID: 18432251.21. Skryma R, Mariot P, Bourhis XL, Coppenolle FV, Shuba Y, Vanden Abeele F, Legrand G, Humez S, Boilly B, Prevarskaya N. Store depletion and store-operated Ca2+ current in human prostate cancer LNCaP cells: involvement in apoptosis. J Physiol. 2000; 527(Pt 1):71–83. PMID: 10944171.22. Chen R, Valencia I, Zhong F, McColl KS, Roderick HL, Bootman MD, Berridge MJ, Conway SJ, Holmes AB, Mignery GA, Velez P, Distelhorst CW. Bcl-2 functionally interacts with inositol 1,4,5-trisphosphate receptors to regulate calcium release from the ER in response to inositol 1,4,5-trisphosphate. J Cell Biol. 2004; 166:193–203. PMID: 15263017.
Article23. Zhong F, Davis MC, McColl KS, Distelhorst CW. Bcl-2 differentially regulates Ca2+ signals according to the strength of T cell receptor activation. J Cell Biol. 2006; 172:127–137. PMID: 16391001.24. Rizzuto R, Pinton P, Ferrari D, Chami M, Szabadkai G, Magalhães PJ, Di Virgilio F, Pozzan T. Calcium and apoptosis: facts and hypotheses. Oncogene. 2003; 22:8619–8627. PMID: 14634623.
Article25. Huber SM, Butz L, Stegen B, Klumpp D, Braun N, Ruth P, Eckert F. Ionizing radiation, ion transports, and radioresistance of cancer cells. Front Physiol. 2013; 4:212. PMID: 23966948.
Article26. Park SI, Park EJ, Kim NH, Baek WK, Lee YT, Lee CJ, Suh CK. Hypoxia delays the intracellular Ca2+ clearance by Na+-Ca2+ exchanger in human adult cardiac myocytes. Yonsei Med J. 2001; 42:333–337. PMID: 11456400.27. Kim YT, Park YJ, Jung SY, Seo WS, Suh CK. Effects of Na+-Ca2+ exchanger activity on the alpha-amino-3-hydroxy-5-methyl-4-isoxazolone-propionate-induced Ca2+ influx in cerebellar Purkinje neurons. Neuroscience. 2005; 131:589–599. PMID: 15730865.28. Song M, Chen D, Yu SP. The TRPC channel blocker SKF 96365 inhibits glioblastoma cell growth by enhancing reverse mode of the Na+/Ca2+ exchanger and increasing intracellular Ca2+. Br J Pharmacol. 2014; 171:3432–3447. PMID: 24641279.29. Park HJ, Lyons JC, Ohtsubo T, Song CW. Cell cycle progression and apoptosis after irradiation in an acidic environment. Cell Death Differ. 2000; 7:729–738. PMID: 10918447.
Article30. Berridge MJ. The endoplasmic reticulum: a multifunctional signaling organelle. Cell Calcium. 2002; 32:235–249. PMID: 12543086.
Article31. Rao RV, Ellerby HM, Bredesen DE. Coupling endoplasmic reticulum stress to the cell death program. Cell Death Differ. 2004; 11:372–380. PMID: 14765132.
Article32. Elliott AC. Recent developments in non-excitable cell calcium entry. Cell Calcium. 2001; 30:73–93. PMID: 11440466.
Article33. Putney JW Jr, Broad LM, Braun FJ, Lievremont JP, Bird GS. Mechanisms of capacitative calcium entry. J Cell Sci. 2001; 114:2223–2229. PMID: 11493662.
Article34. Scorrano L, Oakes SA, Opferman JT, Cheng EH, Sorcinelli MD, Pozzan T, Korsmeyer SJ. BAX and BAK regulation of endoplasmic reticulum Ca2+: a control point for apoptosis. Science. 2003; 300:135–139. PMID: 12624178.35. Urashima T, Wang K, Adelstein SJ, Kassis AI. Activation of diverse pathways to apoptosis by (125)IdUrd and gamma-photon exposure. Int J Radiat Biol. 2004; 80:867–874. PMID: 15764395.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Synergistic Effect of Ionizing Radiation and beta-lapachone against RKO Human Colon Adenocarcinoma Cells
- Stimulatory Anticancer Effect of Resveratrol Mediated by G Protein-Coupled Estrogen Receptor in Colorectal Cancer
- Calcium, Vitamin D, and Colorectal Cancer
- Combination Effect of Nimotuzumab with Radiation in Colorectal Cancer Cells
- Metabolic Adaptation and Cellular Stress Response As Targets for Cancer Therapy