Korean J Physiol Pharmacol.
2014 Feb;18(1):47-53. 10.4196/kjpp.2014.18.1.47.
Characteristics of Diprophylline-Induced Bidirectional Modulation on Rat Jejunal Contractility
- Affiliations
-
- 1Department of Pharmacology, Dalian Medical University, Dalian 116044, China. linyuandmu2008@qq.com
- KMID: 2285490
- DOI: http://doi.org/10.4196/kjpp.2014.18.1.47
Abstract
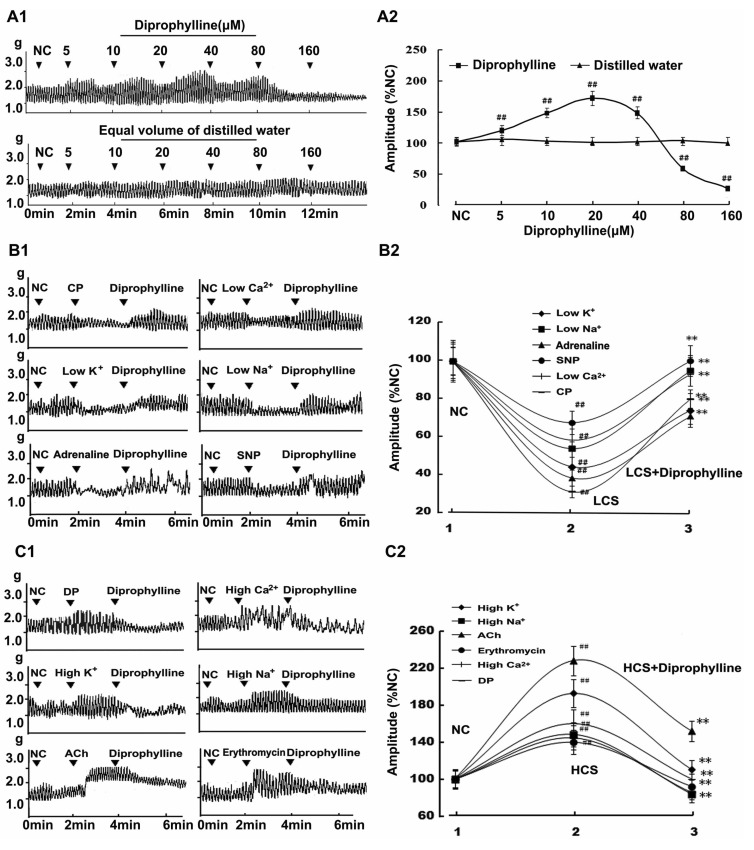
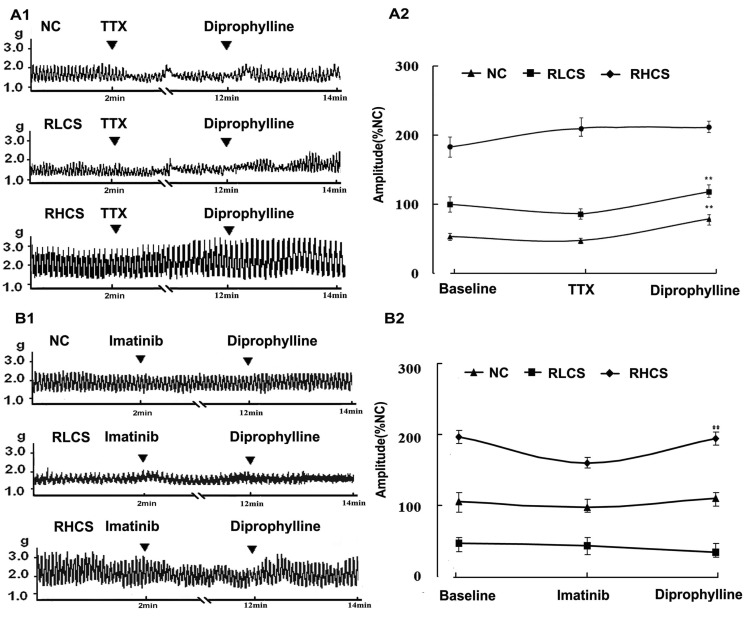
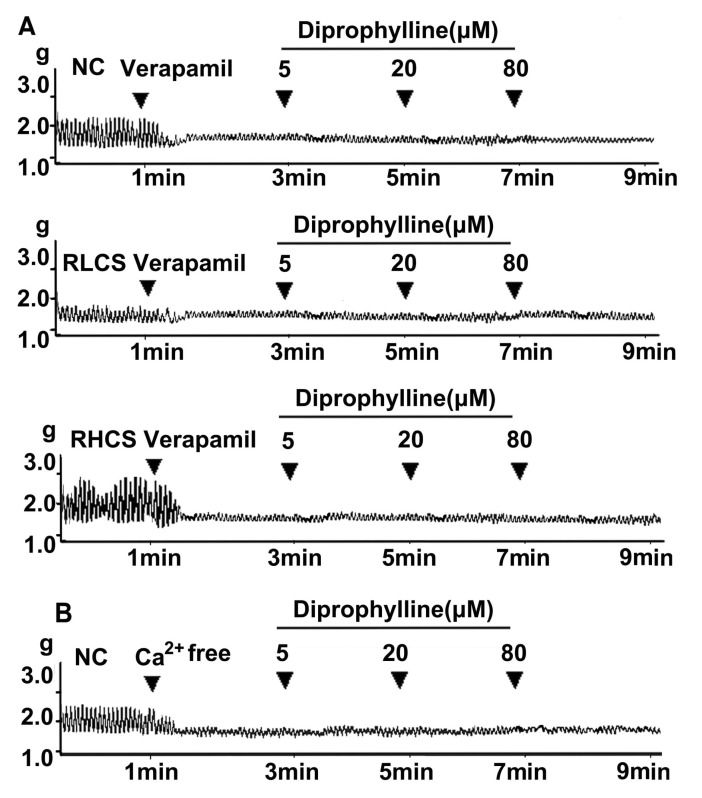
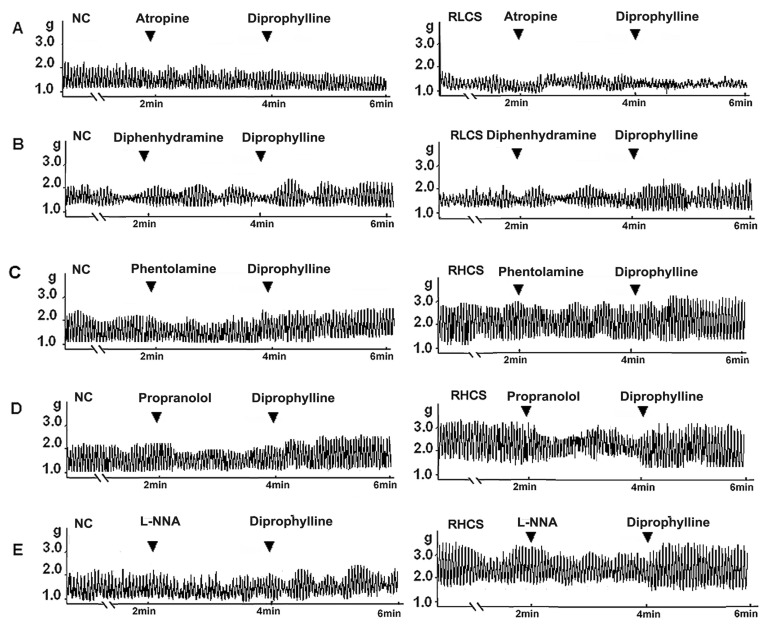
- In this study, we propose that diprophylline exerts bidirectional modulation (BM) on the isolated rat jejunal segment depending on its contractile state. The results supported the hypothesis. Diprophylline (20 microM) exerted stimulatory effects on the contractility of jejunal segment in six low contractile states while inhibitory effects in six high contractile states, showing the characteristics of BM. Diprophylline-induced stimulatory effect was significantly blocked by atropine, indicating the correlation with cholinergic activation. Diprophylline-induced inhibitory effect was partially blocked by phentolamine, propranolol, and L-N-Nitro-Arginine respectively, indicating their correlation with sympathetic activation and nitric oxide-mediated relaxing mechanisms. Diprophylline-induced BM was abolished by tetrodotoxin or in a Ca2+ free condition or pretreated with tyrosine kinase inhibitor imatinib, suggesting that diprophylline-induced BM is Ca2+ dependent, and that it requires the presence of enteric nervous system as well as pacemaker activity of interstitial cells of Cajal. Diprophylline significantly increased the reduced MLCK expression and myosin extent in constipation-prominent rats and significantly decreased the increased MLCK expression and myosin extent in diarrhea-prominent rats, suggesting that the change of MLCK expression may also be involved in diprophylline-induced BM on rat jejunal contractility. In summary, diprophylline-exerted BM depends on the contractile states of the jejunal segments, requires the presence of Ca2+, enteric nervous system, pacemaker activity of interstitial cells of Cajal, and MLCK-correlated myosin phosphorylation. The results suggest the potential implication of diprophylline in relieving alternative hypo/hyper intestinal motility.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Ameliorative effects of atractylodin on intestinal inflammation and co-occurring dysmotility in both constipation and diarrhea prominent rats
Changchun Yu, Yongjian Xiong, Dapeng Chen, Yanli Li, Bin Xu, Yuan Lin, Zeyao Tang, Chunling Jiang, Li Wang
Korean J Physiol Pharmacol. 2017;21(1):1-9. doi: 10.4196/kjpp.2017.21.1.1.
Reference
-
1. Xia Z, Ni Y, Kokot S. Simultaneous determination of caffeine, theophylline and theobromine in food samples by a kinetic spectrophotometric method. Food Chem. 2013; 141:4087–4093. PMID: 23993589.
Article2. Nadai M, Apichartpichean R, Hasegawa T, Nabeshima T. Pharmacokinetics and the effect of probenecid on the renal excretion mechanism of diprophylline. J Pharm Sci. 1992; 81:1024–1027. PMID: 1432614.
Article3. Huang WS, Lin SJ, Wu HL, Chen SH. Simultaneous determination of theophylline and dyphylline by micellar electrokinetic chromatography and application in drug formulations. J Chromatogr B Analyt Technol Biomed Life Sci. 2003; 795:329–335.
Article4. Paterson N. High-performance liquid chromatographic method for the determination of diprophylline in human serum. J Chromatogr. 1982; 232:450–455. PMID: 7153295.
Article5. Gordjani N, Burghard R, Leititis JU, Brandis M, Püschel CH, Gundert-Remy U. Acute intoxication with theophylline, proxyphylline and diprophylline in a 3-month-old infant after rectal application: pharmacokinetic data under hemoperfusion. Acta Paediatr Scand. 1990; 79:112–114. PMID: 2316353.
Article6. Agbaba D, Zivanov-Stakić D, Vukićević N. Dissolution assay of theophylline, diprophylline and proxyphylline from a sustained release dosage form by high performance thin layer chromatography. Biomed Chromatogr. 1992; 6:141–142. PMID: 1525489.
Article7. Kang J. Effects of diprophylline on pulmonary hypertension in patients with chronic pulmonary disease. Zhonghua Jie He He Hu Xi Za Zhi. 1992; 15:153–154. 190PMID: 1473187.8. Zuidema J, Merkus FWHM. Pharmacokinetics and pharmacodynamics of diprophylline. Pharm Weekbl. 1981; 3:1320–1325.
Article9. McEvoy GK. American Society of Health-System Pharmacists. AHFS Drug information 2001. Bethesda: American Society of Health-System Pharmacists;c2001. p. 2296. p. 3486.10. Schemann M. Control of gastrointestinal motility by the "gut brain"--the enteric nervous system. J Pediatr Gastroenterol Nutr. 2005; 41(Suppl 1):S4–S6. PMID: 16131964.11. La JH, Kim TW, Sung TS, Kang JW, Kim HJ, Yang IS. Visceral hypersensitivity and altered colonic motility after subsidence of inflammation in a rat model of colitis. World J Gastroenterol. 2003; 9:2791–2795. PMID: 14669335.
Article12. Zou BC, Dong L, Wang Y, Wang SH, Cao MB. Expression and role of 5-HT7 receptor in brain and intestine in rats with irritable bowel syndrome. Chin Med J (Engl). 2007; 120:2069–2074. PMID: 18167178.
Article13. Tanović A, Fernández E, Jiménez M. Alterations in intestinal contractility during inflammation are caused by both smooth muscle damage and specific receptor-mediated mechanisms. Croat Med J. 2006; 47:318–326. PMID: 16625700.14. Frings M, Haschke G, Heinke B, Schäfer KH, Diener M. Spontaneous contractions of intestinal smooth muscle re-aggregates from the new-born rat triggered by thromboxane A2. J Vet Med A Physiol Pathol Clin Med. 2000; 47:469–475. PMID: 11075538.
Article15. Chen D, Xiong Y, Wang L, Lv B, Lin Y. Characteristics of emodin on modulating the contractility of jejunal smooth muscle. Can J Physiol Pharmacol. 2012; 90:455–462. PMID: 22452333.
Article16. Kobayashi K, Murata T, Hori M, Ozaki H. Prostaglandin E2-prostanoid EP3 signal induces vascular contraction via nPKC and ROCK activation in rat mesenteric artery. Eur J Pharmacol. 2011; 660:375–380. PMID: 21463619.
Article17. Iwabu A, Smith K, Allen FD, Lauffenburger DA, Wells A. Epidermal growth factor induces fibroblast contractility and motility via a protein kinase C delta-dependent pathway. J Biol Chem. 2004; 279:14551–14560. PMID: 14747473.18. Chitano P, Voynow JA, Pozzato V, Cantillana V, Burch LH, Wang L, Murphy TM. Ontogenesis of myosin light chain kinase mRNA and protein content in guinea pig tracheal smooth muscle. Pediatr Pulmonol. 2004; 38:456–464. PMID: 15376333.
Article19. Ruiz IL, Urbano-Cuadrado M, Gómez-Nieto MÁ. Improving the development of QSAR prediction models with the use of approximate similarity approach. Engineering Letters. 2008; 16:36–43.20. Neunlist M, Toumi F, Oreschkova T, Denis M, Leborgne J, Laboisse CL, Galmiche JP, Jarry A. Human ENS regulates the intestinal epithelial barrier permeability and a tight junction-associated protein ZO-1 via VIPergic pathways. Am J Physiol Gastrointest Liver Physiol. 2003; 285:G1028–G1036. PMID: 12881224.
Article21. Seiler R, Rickenbacher A, Shaw S, Balsiger BM. alpha- and beta-adrenergic receptor mechanisms in spontaneous contractile activity of rat ileal longitudinal smooth muscle. J Gastrointest Surg. 2005; 9:227–235. PMID: 15694819.22. Han JH, Chang IH, Myung SC, Lee MY, Kim WY, Lee SY, Lee SY, Lee SW, Kim KD. A novel pathway underlying the inhibitory effects of melatonin on isolated rat urinary bladder contraction. Korean J Physiol Pharmacol. 2012; 16:37–42. PMID: 22416218.
Article23. Sanders KM, Ordög T, Koh SD, Torihashi S, Ward SM. Development and plasticity of interstitial cells of Cajal. Neurogastroenterol Motil. 1999; 11:311–338. PMID: 10520164.
Article24. Kim BJ, Kwon YK, Kim E, So I. Effects of histamine on cultured interstitial cells of cajal in murine small intestine. Korean J Physiol Pharmacol. 2013; 17:149–156. PMID: 23626477.
Article25. Li M, Johnson CP, Adams MB, Sarna SK. Cholinergic and nitrergic regulation of in vivo giant migrating contractions in rat colon. Am J Physiol Gastrointest Liver Physiol. 2002; 283:G544–G552. PMID: 12181166.26. Konturek SJ, Siebers R. Role of histamine H1- and H2-receptors in myoelectric activity of small bowel in the dog. Am J Physiol. 1980; 238:G50–G56. PMID: 6444500.
Article27. Gaddum JH, Holzbauer M. Adrenaline and noradrenaline. Vitam Horm. 1957; 15:151–203. PMID: 13530704.
Article28. Kim YC, Choi W, Yun HY, Sung R, Yoo RY, Park SM, Yun SJ, Kim MJ, Song YJ, Xu WX, Lee SJ. Nitric Oxide-mediated Relaxation by High K in Human Gastric Longitudinal Smooth Muscle. Korean J Physiol Pharmacol. 2011; 15:405–413. PMID: 22359479.29. Winquist RJ. Modulators of intracellular calcium. Drug Develop Res. 1984; 4:241–256.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Ameliorative effects of atractylodin on intestinal inflammation and co-occurring dysmotility in both constipation and diarrhea prominent rats
- Effects of Pancuronium on Isolated Rat Jejunal Preparations
- The Effects of Diazepam on the Carbachol Induced Contraction of the Isolated Rat Ileum
- The Effects of Nitroglycerin on the Contraction of Rat Uterine Smooth Muscle
- Effect of Morphine , Meperidine , Diazepam and Ketamine on Pregnant Rat Uteri , in Vitro