Korean J Physiol Pharmacol.
2013 Aug;17(4):359-365. 10.4196/kjpp.2013.17.4.359.
Mechanism of Relaxation Via TASK-2 Channels in Uterine Circular Muscle of Mouse
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Chungbuk National University College of Medicine, Cheongju 361-763, Korea. hakskim@chungbuk.ac.kr
- 2Department of Pathology, Chungbuk National University College of Medicine, Cheongju 361-763, Korea.
- 3Department of Physiology, Chungbuk National University College of Medicine, Cheongju 361-763, Korea. physiokyc@chungbuk.ac.kr
- 4Department of Physiology, Nagoya City University Medical School, Nagoya 467-8601, Japan.
- 5Department of Pharmacology, Chungbuk National University College of Medicine, Cheongju 361-763, Korea.
- 6Department of Urology, College of Medicine, Chung-Ang University, Seoul 156-756, Korea.
- 7Department of Physiology, College of Medicine, Chung-Ang University, Seoul 156-756, Korea.
- 8Department of Physiology, Sungkyunkwan University School of Medicine, Suwon 440-746, Korea.
- 9Department of Anesthesiology and Pain Medicine, Chungbuk National University College of Medicine, Cheongju 361-763, Korea.
- 10Department of Surgery, Chungbuk National University College of Medicine, Cheongju 361-763, Korea.
- 11Department of Physiology, Shanghai Jiaotong University School of Medicine, Shanghai 200240, China.
- KMID: 2285471
- DOI: http://doi.org/10.4196/kjpp.2013.17.4.359
Abstract
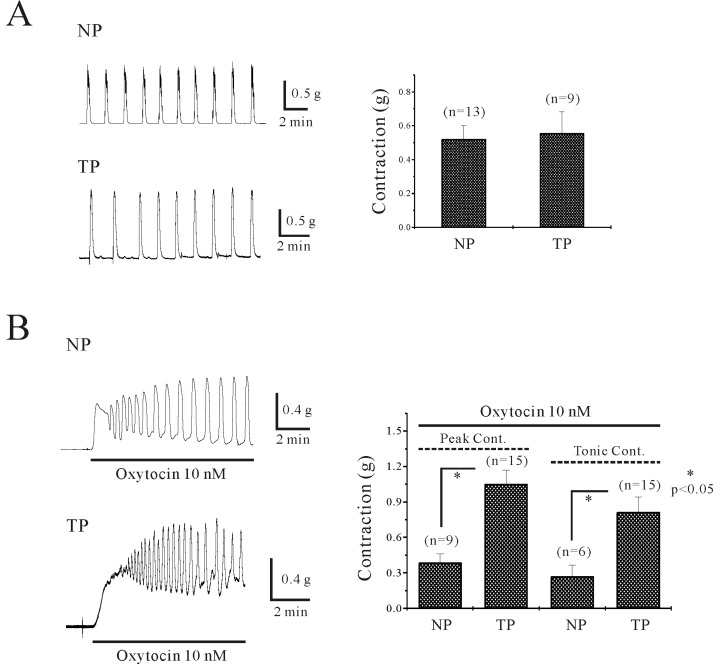
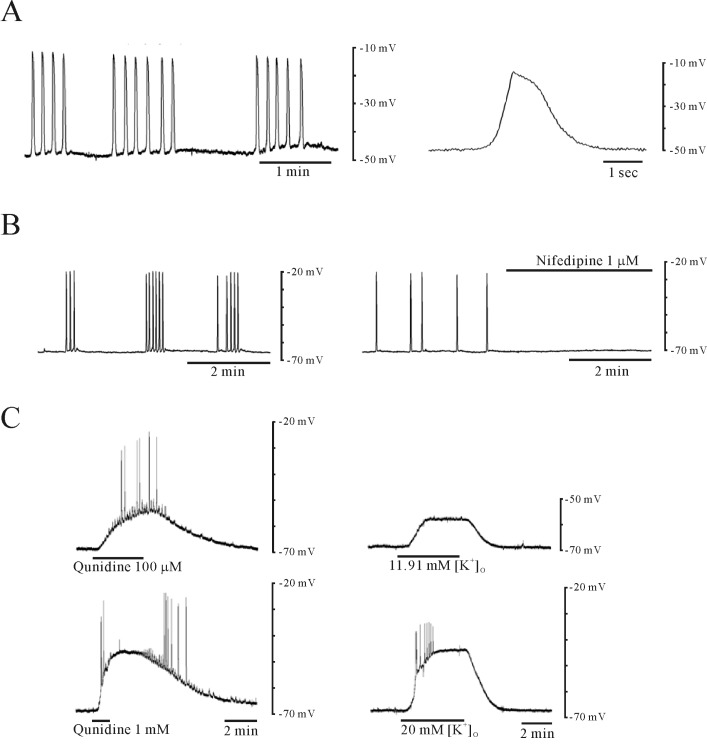
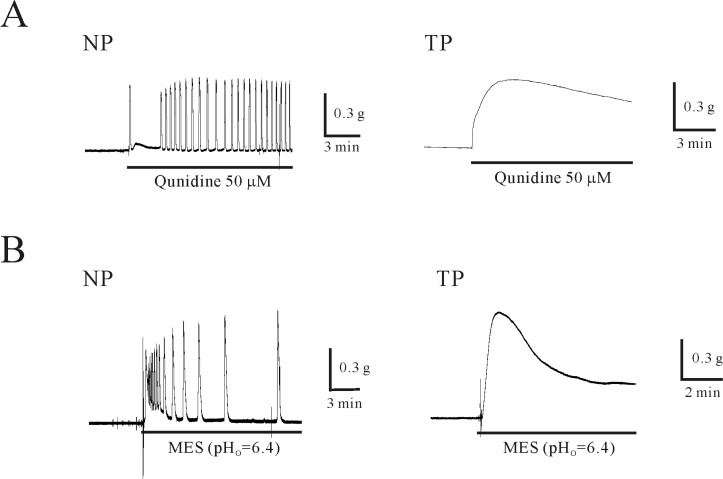
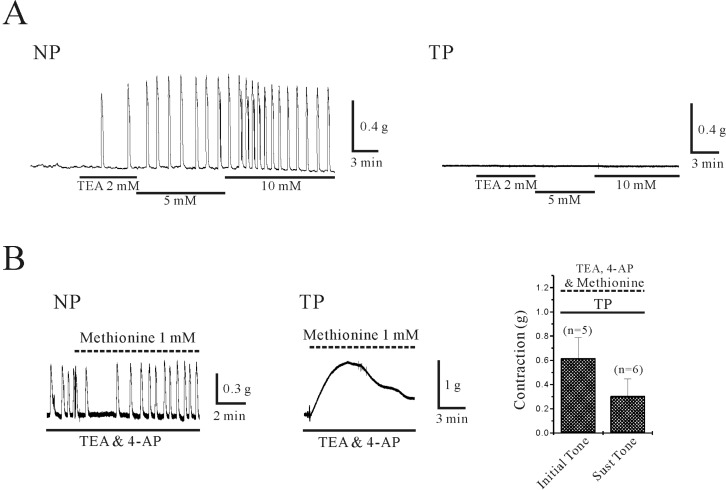
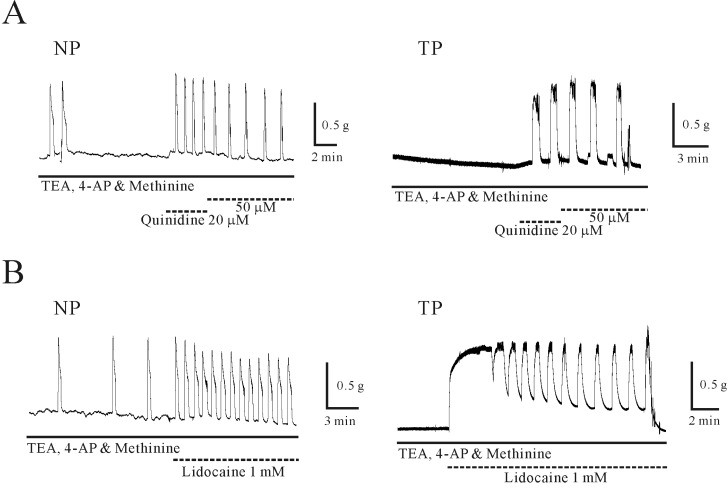
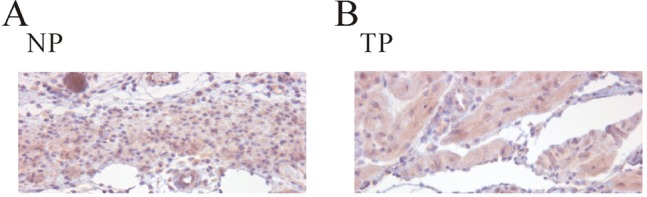
- Plasma pH can be altered during pregnancy and at labor. Membrane excitability of smooth muscle including uterine muscle is suppressed by the activation of K+ channels. Because contractility of uterine muscle is regulated by extracellular pH and humoral factors, K+ conductance could be connected to factors regulating uterine contractility during pregnancy. Here, we showed that TASK-2 inhibitors such as quinidine, lidocaine, and extracellular acidosis produced contraction in uterine circular muscle of mouse. Furthermore, contractility was significantly increased in pregnant uterine circular muscle than that of non-pregnant muscle. These patterns were not changed even in the presence of tetraetylammonium (TEA) and 4-aminopyridine (4-AP). Finally, TASK-2 inhibitors induced strong myometrial contraction even in the presence of L-methionine, a known inhibitor of stretchactivated channels in myometrium. When compared to non-pregnant myometrium, pregnant myometrium showed increased immunohistochemical expression of TASK-2. Therefore, TASK-2, seems to play a key role during regulation of myometrial contractility in the pregnancy and provides new insight into preventing preterm delivery.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Myometrial relaxation of mice via expression of two pore domain acid sensitive K+ (TASK-2) channels
Kyu-Sang Kyeong, Seung Hwa Hong, Young Chul Kim, Woong Cho, Sun Chul Myung, Moo Yeol Lee, Ra Young You, Chan Hyung Kim, So Yeon Kwon, Hikaru Suzuki, Yeon Jin Park, Eun-Hwan Jeong, Hak Soon Kim, Heon Kim, Seung Woon Lim, Wen-Xie Xu, Sang Jin Lee, Il Woon Ji
Korean J Physiol Pharmacol. 2016;20(5):547-556. doi: 10.4196/kjpp.2016.20.5.547.
Reference
-
1. Schwarz MK, Page P. Preterm labour: an overview of current and emerging therapeutics. Curr Med Chem. 2003; 10:1441–1468. PMID: 12871140.
Article2. Creasy RK, Resnik R, Iams JD. Creasy and Resnik's maternal-fetal medicine: principles and practice. 6th ed. Philadelphia, PA: Saunders/Elsevier;2009. p. 243–521.3. Wray S, Noble K. Sex hormones and excitation-contraction coupling in the uterus: the effects of oestrous and hormones. J Neuroendocrinol. 2008; 20:451–461. PMID: 18266942.
Article4. Rooth G, Sjostedt S. The placental transfer of gases and fixed acids. Arch Dis Child. 1962; 37:366–370. PMID: 14493734.
Article5. Cerri V, Tarantini M, Zuliani G, Schena V, Redaelli C, Nicolini U. Intravenous glucose infusion in labor does not affect maternal and fetal acid-base balance. J Matern Fetal Med. 2000; 9:204–208. PMID: 11048829.
Article6. Sjostedt S. Acid-base balance of arterial blood during pregnancy, at delivery, and in the puerperium. Am J Obstet Gynecol. 1962; 84:775–779. PMID: 13913556.7. Harrison N, Larcombe-McDouall JB, Earley L, Wray S. An in vivo study of the effects of ischaemia on uterine contraction, intracellular pH and metabolites in the rat. J Physiol. 1994; 476:349–354. PMID: 8046648.
Article8. Brinkman CR. Circulation in the pregnant uterus. In : Carsten ME, Miller JD, editors. Uterine function: molecular and cellular aspects. New York: Plenum Press;1990. p. 519–537.10. Pinto RM, Lerner U, Pontelli H. The effect of progesterone on oxytocin-induced contraction of the three separate layers of human gestational myometrium in the uterine body and lower segment. Am J Obstet Gynecol. 1967; 98:547–554. PMID: 6025148.
Article11. Tulchinsky D, Hobel CJ, Yeager E, Marshall JR. Plasma estrone, estradiol, estriol, progesterone, and 17-hydroxyprogesterone in human pregnancy. I. Normal pregnancy. Am J Obstet Gynecol. 1972; 112:1095–1100. PMID: 5025870.12. Monaghan K, Baker SA, Dwyer L, Hatton WC, Sik Park K, Sanders KM, Koh SD. The stretch-dependent potassium channel TREK-1 and its function in murine myometrium. J Physiol. 2011; 589:1221–1233. PMID: 21224218.
Article13. Pierce SJ, Kupittayanant S, Shmygol T, Wray S. The effects of pH change on Ca2+ signaling and force in pregnant human myometrium. Am J Obstet Gynecol. 2003; 188:1031–1038. PMID: 12712106.14. Taggart MJ, Sheader EA, Walker SD, Naderali EK, Moore S, Wray S. External alkalinization decreases intracellular Ca2+ and spontaneous contractions in pregnant rat myometrium. Am J Obstet Gynecol. 1997; 177:959–963. PMID: 9369852.15. Sanborn BM. Ion channels and the control of myometrial electrical activity. Semin Perinatol. 1995; 19:31–40. PMID: 7538700.
Article16. Anwer K, Oberti C, Perez GJ, Perez-Reyes N, McDougall JK, Monga M, Sanborn BM, Stefani E, Toro L. Calcium-activated K+ channels as modulators of human myometrial contractile activity. Am J Physiol. 1993; 265:C976–C985. PMID: 8238323.17. Brown A, Cornwell T, Korniyenko I, Solodushko V, Bond CT, Adelman JP, Taylor MS. Myometrial expression of small conductance Ca2+-activated K+ channels depresses phasic uterine contraction. Am J Physiol Cell Physiol. 2007; 292:C832–C840. PMID: 17296820.18. Khan RN, Smith SK, Morrison JJ, Ashford ML. Ca2+ dependence and pharmacology of large-conductance K+ channels in nonlabor and labor human uterine myocytes. Am J Physiol. 1997; 273:C1721–C1731. PMID: 9374660.19. Smith RC, McClure MC, Smith MA, Abel PW, Bradley ME. The role of voltage-gated potassium channels in the regulation of mouse uterine contractility. Reprod Biol Endocrinol. 2007; 5:41. PMID: 17980032.
Article20. Ketchum KA, Joiner WJ, Sellers AJ, Kaczmarek LK, Goldstein SA. A new family of outwardly rectifying potassium channel proteins with two pore domains in tandem. Nature. 1995; 376:690–695. PMID: 7651518.
Article21. O'Connell AD, Morton MJ, Hunter M. Two-pore domain K+ channels-molecular sensors. Biochim Biophys Acta. 2002; 1566:152–161. PMID: 12421546.22. Duprat F, Lesage F, Fink M, Reyes R, Heurteaux C, Lazdunski M. TASK, a human background K+ channel to sense external pH variations near physiological pH. EMBO J. 1997; 16:5464–5471. PMID: 9312005.23. Buxton IL, Singer CA, Tichenor JN. Expression of stretchactivated two-pore potassium channels in human myometrium in pregnancy and labor. PLoS One. 2010; 5:e12372. PMID: 20811500.
Article24. Kim YC, Choi W, Yun HY, Sung R, Yoo RY, Park SM, Yun SJ, Kim MJ, Song YJ, Xu WX, Lee SJ. Nitric oxide-mediated relaxation by high K+ in human gastric longitudinal smooth muscle. Korean J Physiol Pharmacol. 2011; 15:405–413. PMID: 22359479.25. Sung R, Kim YC, Yun HY, Choi W, Kim HS, Kim H, Lee KJ, You RY, Park SM, Youn SJ, Kim MJ, Kim WS, Song YJ, Kim SY, Xu WX, Lee SJ. Interstitial cells of Cajal (ICC)-like-c-Kit positive cells are involved in gastritis and carcinogenesis in human stomach. Oncol Rep. 2011; 26:33–42. PMID: 21573494.
Article26. Yun HY, Sung R, Kim YC, Choi W, Kim HS, Kim H, Lee GJ, You RY, Park SM, Yun SJ, Kim MJ, Kim WS, Song YJ, Xu WX, Lee SJ. Regional distribution of interstitial cells of cajal (ICC) in human stomach. Korean J Physiol Pharmacol. 2010; 14:317–324. PMID: 21165331.
Article27. Reyes R, Duprat F, Lesage F, Fink M, Salinas M, Farman N, Lazdunski M. Cloning and expression of a novel pH-sensitive two pore domain K+ channel from human kidney. J Biol Chem. 1998; 273:30863–30869. PMID: 9812978.28. Brayden JE, Nelson MT. Regulation of arterial tone by activation of calcium-dependent potassium channels. Science. 1992; 256:532–535. PMID: 1373909.
Article29. Park JK, Kim YC, Sim JH, Choi MY, Choi W, Hwang KK, Cho MC, Kim KW, Lim SW, Lee SJ. Regulation of membrane excitability by intracellular pH (pHi) changers through Ca2+-activated K+ current (BK channel) in single smooth muscle cells from rabbit basilar artery. Pflugers Arch. 2007; 454:307–319. PMID: 17285302.30. Miller P, Kemp PJ, Lewis A, Chapman CG, Meadows HJ, Peers C. Acute hypoxia occludes hTREK-1 modulation: re-evaluation of the potential role of tandem P domain K+ channels in central neuroprotection. J Physiol. 2003; 548:31–37. PMID: 12611912.31. Parkington HC, Coleman HA. Excitability in uterine smooth muscle. Front Horm Res. 2001; 27:179–200. PMID: 11450426.
Article32. Khan RN, Matharoo-Ball B, Arulkumaran S, Ashford ML. Potassium channels in the human myometrium. Exp Physiol. 2001; 86:255–264. PMID: 11429642.
Article34. Knock GA, Tribe RM, Hassoni AA, Aaronson PI. Modulation of potassium current characteristics in human myometrial smooth muscle by 17beta-estradiol and progesterone. Biol Reprod. 2001; 64:1526–1534. PMID: 11319161.35. Toro L, Stefani E, Erulkar S. Hormonal regulation of potassium currents in single myometrial cells. Proc Natl Acad Sci U S A. 1990; 87:2892–2895. PMID: 2326254.
Article36. Taggart M, Wray S. Simultaneous measurement of intracellular pH and contraction in uterine smooth muscle. Pflugers Arch. 1993; 423:527–529. PMID: 8351202.
Article37. Parratt JR, Taggart MJ, Wray S. Functional effects of intracellular pH alteration in the human uterus: simultaneous measurements of pH and force. J Reprod Fertil. 1995; 105:71–75. PMID: 7490717.
Article38. Naderali EK, Wray S. Modulation of force induced by pH in the guinea-pig uterus examined at two stages of the oestrous cycle. J Reprod Fertil. 1999; 117:153–157. PMID: 10645256.
Article39. Bai X, Bugg GJ, Greenwood SL, Glazier JD, Sibley CP, Baker PN, Taggart MJ, Fyfe GK. Expression of TASK and TREK, two-pore domain K+ channels, in human myometrium. Reproduction. 2005; 129:525–530. PMID: 15798028.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Myometrial relaxation of mice via expression of two pore domain acid sensitive K⺠(TASK-2) channels
- Longitudinal Muscle Dysfunction in Achalasia Esophagus and Its Relevance
- The effects of estrogen and progesterone on vascular reactivity of endothelium-denuded human uterine artery
- Physiological Roles and Properties of Ion Channels in Corporal Smooth Muscle
- The relaxant effect of nicardipine on the isolated uterine smooth muscle of the pregnant rat