Korean J Pediatr Infect Dis.
2013 Aug;20(2):53-62.
Cross-reaction of 6B and 19F Specific Antibodies to Serotypes 6A, 6C, and 19A after Immunization with 7-valent Pneumococcal Conjugate Vaccine in Korean Children Aged 12-23 Months
- Affiliations
-
- 1Department of Pediatrics, School of Medicine, Ewha Womans University, Seoul, Republic of Korea. kaykim@ewha.ac.kr
- 2Center for Vaccine Evaluation and Study, School of Medicine, Ewha Womans University, Seoul, Republic of Korea.
Abstract
- PURPOSE
The cross-protection of 7-valent pneumococcal conjugate vaccine (PCV7) against vaccine-related serotypes has been controversial. We investigated the serological properties of cross-protective antibodies against vaccine-related serotypes 6A, 6C, and 19A induced in young children aged 12-23 months after booster immunization of PCV7.
METHODS
IgG and IgM antibody concentrations and opsonic index (OI) against vaccine serotypes 6B and 19F and vaccine-related serotypes 6A, 6C, and 19A were measured by ELISA and opsonophagocytic killing assay (OPA) in 4 selected immunesera. The serological properties and antigenic specificity of protective antibodies were determined by IgM depletion of immunesera, OPA, and competitive OPA against serogroup 6 and 19 pneumococci.
RESULTS
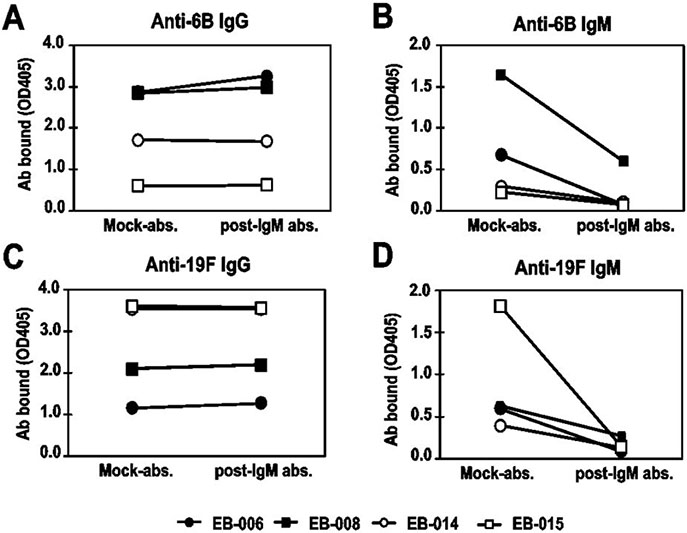
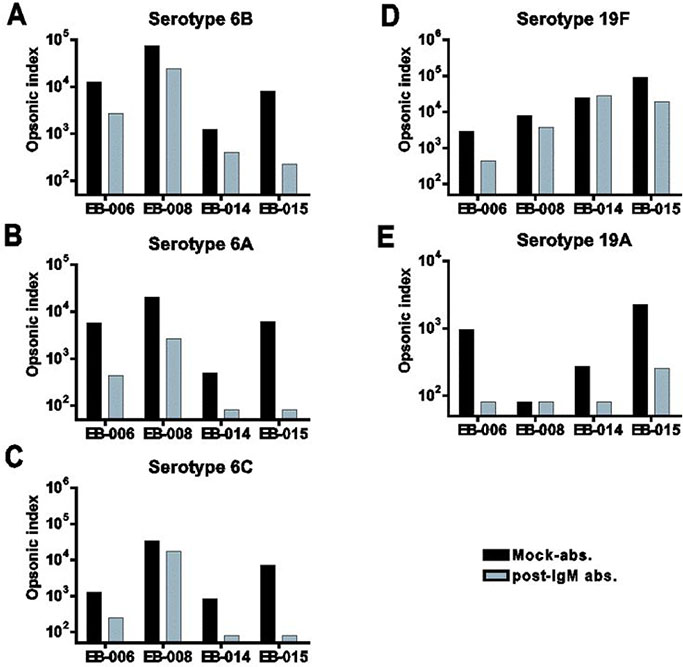
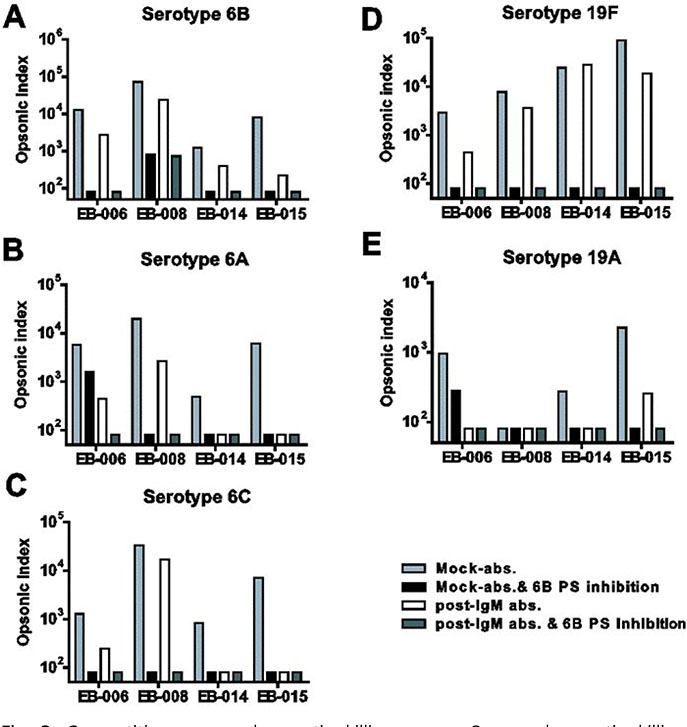
Compared to pre-IgM depleted immunesera, OI of IgM-depleted immunesera against 6B and 19F decreased and OI against 6A, 6C, and 19A decreased, too. In competition OPA, free 6B and 19F polysaccharide completely inhibited the immune protection against vaccine-related serotypes 6A, 6C, and 19A as well as vaccine types 6B and 19F.
CONCLUSIONS
The booster immunization of PCV7 certainly induced cross-protective antibodies against vaccine-related serotypes 6A, 6C, and 19A with both IgG and IgM isotypes. Furthermore, IgM antibodies are more highly contributed to opsonophagocytic activity against vaccine-related serotypes as well as most of vaccine types than do IgG antibodies. Further studies are needed for the more immunized sera in the children as well as adults.
Keyword
MeSH Terms
-
Adult
Aged
Antibodies
Child
Cross Protection
Enzyme-Linked Immunosorbent Assay
Epitopes
Homicide
Humans
Immunization
Immunization, Secondary
Immunoglobulin G
Immunoglobulin M
Pneumococcal Vaccines
Streptococcus pneumoniae
Heptavalent Pneumococcal Conjugate Vaccine
Antibodies
Epitopes
Immunoglobulin G
Immunoglobulin M
Pneumococcal Vaccines
Figure
Reference
-
1. The Korean Pediatric Society. Pneumococcal vaccine. In : Lee HJ, editor. Immunization Guideline. 7th ed. Seoul: The Korean Pediatric Society;2012. p. 160–174.2. Lagergård T, Branefors P. Nature of cross-reactivity between Haemophilus influenzae types A and B and Streptococcus pneumoniae types 6A and 6B. Acta Pathol Microbiol Immunol Scand C. 1983; 91:371–376.3. Jakobsen H, Sigurdsson VD, Sigurdardottir S, Schulz D, Jonsdottir I. Pneumococcal serotype 19F conjugate vaccine induces cross-protective immunity to serotype 19A in a murine pneumococcal pneumonia model. Infect Immun. 2003; 71:2956–2959.
Article4. Lee CJ, Lee LH, Frasch CE. Protective immunity of pneumococcal glycoconjugates. Crit Rev Microbiol. 2003; 29:333–349.
Article5. Saeland E, Jakobsen H, Ingolfsdottir G, Sigurdardottir ST, Jonsdottir I. Serum samples from infants vaccinated with a pneumococcal conjugate vaccine, PncT, protect mice against invasive infection caused by Streptococcus pneumoniae serotypes 6A and 6B. J Infect Dis. 2001; 183:253–260.
Article6. Richter SS, Heilmann KP, Dohrn CL, Riahi F, Beekmann SE, Doern GV. Changing epidemiology of antimicrobial-resistant Streptococcus pneumoniae in the United States, 2004-2005. Clin Infect Dis. 2009; 48:e23–e33.7. Cohen R. The need for prudent use of antibiotics and routine use of vaccines. Clin Microbiol Infect. 2009; 15:Suppl 3. 21–23.
Article8. Choi EH, Kim KH, Kim YJ, Kim JH, Park SE, Lee HJ, et al. Recommendation for use of the newly introduced pneumococcal protein conjugate vaccines in Korea. Korean J Pediatr. 2011; 54:146–151.
Article9. Kim KH, Hong JY, Lee H, Kwak GY, Nam CH, Lee YS, et al. Nasopharyngeal pneumococcal carriage of children attending day care centers in Korea: comparison between children immunized with 7-valent pneumococcal conjugate vaccine and non-immunized. J Korean Med Sci. 2011; 26:184–190.
Article10. Pelton SI, Huot H, Finkelstein JA, Bishop CJ, Hsu KK, Kellenber J, et al. Emergence of 19A as virulent and multidrug resistant Pneumococcus in Massachusetts following universal immunization of infants with pneumococcal conjugate vaccine. Pediatr Infect Dis J. 2007; 26:468–472.
Article11. Park IH, Pritchard DG, Cartee R, Brandao A, Brandileone MC, Nahm MH. Discovery of a new capsular serotype (6C) within serogroup 6 of Streptococcus pneumoniae. J Clin Microbiol. 2007; 45:1225–1233.
Article12. Park IH, Moore MR, Treanor JJ, Pelton SI, Pilishvili T, Beall B, et al. Differential effects of pneumococcal vaccines against serotypes 6A and 6C. J Infect Dis. 2008; 198:1818–1822.
Article13. Lee H, Nahm MH, Burton R, Kim KH. Immune response in infants to the heptavalent pneumococcal conjugate vaccine against vaccine-related serotypes 6A and 19A. Clin Vaccine Immunol. 2009; 16:376–381.
Article14. Bryant KA, Block SL, Baker SA, Gruber WC, Scott DA. Safety and immunogenicity of a 13-valent pneumococcal conjugate vaccine. Pediatrics. 2010; 125:866–875.
Article15. Nuorti JP, Whitney CG. Prevention of pneumococcal disease among infants and children - use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine - recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2010; 59:1–18.16. Lee HJ, Park SE, Kim KH. Immune response to 19A serotype after immunization of 19F containing pneumococcal conjugate vaccine in Korean children aged 12-23 months. Korean J Pediatr. 2011; 54:163–168.
Article17. Cha JH. Immune responses against serogroup 6 (6A, 6B, 6C, and 6D) among subjects vaccinated with the 7-valent pneumococcal conjugate vaccine. Seoul: School of Medicine, Ewha Womans University;2012. Ph.D. dissertation.18. Simell B, Nurkka A, Ekstrom N, Givon-Lavi N, Kayhty H, Dagan R. Serum IgM antibodies contribute to high levels of opsonophagocytic activities in toddlers immunized with a single dose of the 9-valent pneumococcal conjugate vaccine. Clin Vaccine Immunol. 2012; 19:1618–1623.
Article19. Burton RL, Nahm MH. Development and validation of a fourfold multiplexed opsonization assay (MOPA4) for pneumococcal antibodies. Clin Vaccine Immunol. 2006; 13:1004–1009.
Article20. Mond JJ, Lees A, Snapper CM. T cell-independent antigens type 2. Annu Rev Immunol. 1995; 13:655–692.
Article21. de Vinuesa CG, Cook MC, Ball J, Sunners Y, Cascalho M, Wabl M, et al. Germinal centers without T cells. J Exp Med. 2000; 191:485–494.
Article22. Lentz VM, Manser T. Cutting edge: germinal centers can be induced in the absence of T cells. J Immunol. 2001; 167:15–20.
Article23. Toellner KM, Jenkinson WE, Taylor DR, Khan M, Sze DMY, Sansom DM, et al. Low-level hypermutation in T cell-independent germinal centers compared with high mutation rates associated with T cell-dependent germinal centers. J Exp Med. 2002; 195:383–389.
Article24. Obukhanych TV, Nussenzweig MC. T-independent type II immune responses generate memory B cells. J Exp Med. 2006; 203:305–310.
Article25. Shi Y, Yamazaki T, Okubo Y, Uehara Y, Sugane K, Agematsu K. Regulation of aged humoral immune defense against pneumococcal bacteria by IgM memory B cell. J Immunol. 2005; 175:3262–3267.
Article26. Maxwell KF, Powell MS, Hulett MD, Barton PA, Mc-Kenzie IFC, Garrett TPJ, et al. Crystal structure of the human leukocyte Fc receptor, FcγRIIa. Nat Struct Biol. 1999; 6:437–442.27. Yee AM, Phan HM, Zuniga R, Salmon JE, Musher DM. Association between FcγRIIa-R131 allotype and bacteremic pneumococcal pneumonia. Clin Infect Dis. 2000; 30:25–28.
Article28. Yuan FF, Wong M, Pererva N, Keating J, Davis AR, Bryant JA, et al. FcγRIIA polymorphisms in Streptococcus pneumoniae infection. Immunol Cell Biol. 2003; 81:192–195.
Article29. Stray-Pedersen A, Aaberge IS, Fruh A, Abrahamsen TG. Pneumococcal conjugate vaccine followed by pneumococcal polysaccharide vaccine; immunogenicity in patients with ataxia-telangiectasia. Clin Exp Immunol. 2005; 140:507–516.
Article30. Mackenzie GA, Carapetis JR, Leach AJ, Morris PS. Pneumococcal vaccination and otitis media in Australian Aboriginal infants: comparison of two birth cohorts before and after introduction of vaccination. BMC pediatr. 2009; 9:14.
Article31. Baxendale HE, Johnson M, Stephens RC, Yuste J, Klein N, Brown JS, et al. Natural human antibodies to pneumococcus have distinctive molecular characteristics and protect against pneumococcal disease. Clin Exp Immunol. 2008; 151:51–60.
Article32. Raff HV, Bradley C, Brady W, Donaldson K, Lipsich L, Maloney G, et al. Comparison of functional activities between IgG1 and IgM class-switched human monoclonal antibodies reactive with group B streptococci or Escherichia coli K1. J Infect Dis. 1991; 163:346–354.
Article33. Shyur SD, Raff HV, Bohnsack JF, Kelsey DK, Hill HR. Comparison of the opsonic and complement triggering activity of human monoclonal IgG1 and IgM antibody against group B streptococci. J Immunol. 1992; 148:1879–1884.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Impact of IgM Antibodies on Cross-Protection against Pneumococcal Serogroups 6 and 19 after Immunization with 7-Valent Pneumococcal Conjugate Vaccine in Children
- Immune response to 19A serotype after immunization of 19F containing pneumococcal conjugate vaccine in Korean children aged 12-23 months
- Immunogenicity of 7-valent pneumococcal conjugate vaccine related to booster immunization in Korean children
- Functional Immunity to Cross-Reactive Serotype 6A Induced by Serotype 6B in Pneumococcal Polysaccharide Vaccine
- Functional Antibody Responses to Seven Serotypes in Pneumococcal Polysaccharide Vaccine in Children