J Korean Med Sci.
2016 Jun;31(6):950-956. 10.3346/jkms.2016.31.6.950.
Impact of IgM Antibodies on Cross-Protection against Pneumococcal Serogroups 6 and 19 after Immunization with 7-Valent Pneumococcal Conjugate Vaccine in Children
- Affiliations
-
- 1Center for Vaccine Evaluation and Study, Medical Research Institute, Ewha Womans University School of Medicine, Seoul, Korea. kaykim@ewha.ac.kr
- 2Department of Pediatrics, Graduate School of Medicine, Gachon University, Incheon, Korea.
- 3Departments of Pathology and Microbiology, University of Alabama at Birmingham, Birmingham, AL, USA.
- 4Department of Pediatrics, Ewha Womans University School of Medicine, Seoul, Korea.
- KMID: 2373705
- DOI: http://doi.org/10.3346/jkms.2016.31.6.950
Abstract
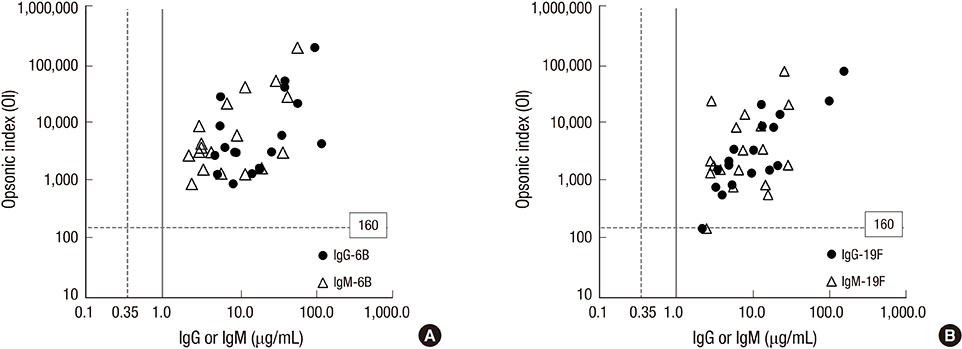
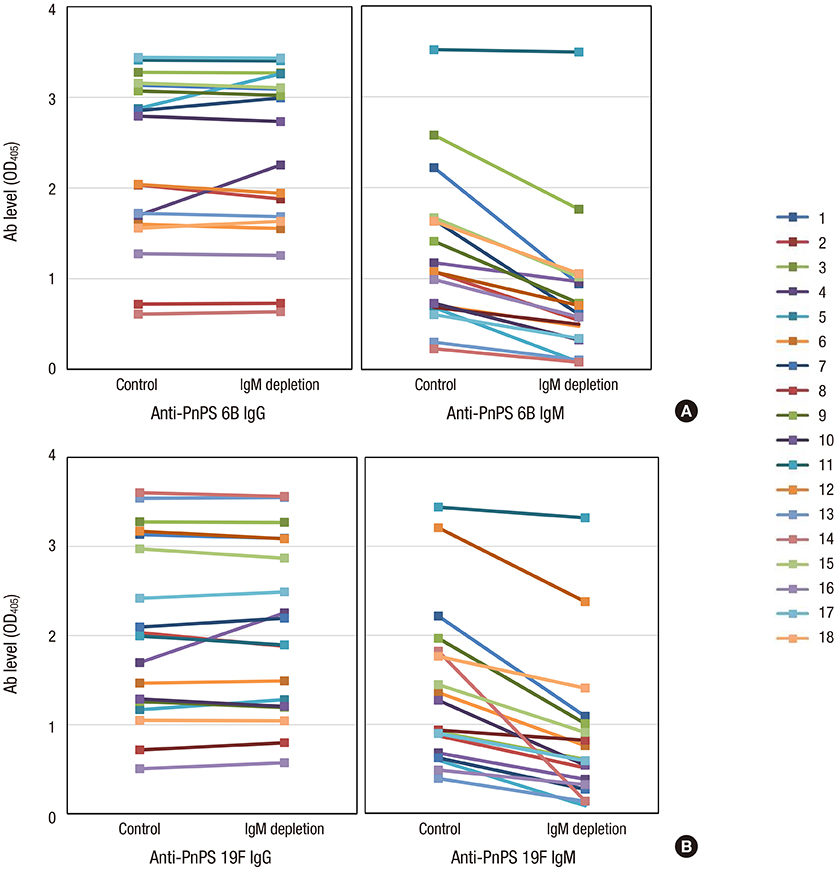
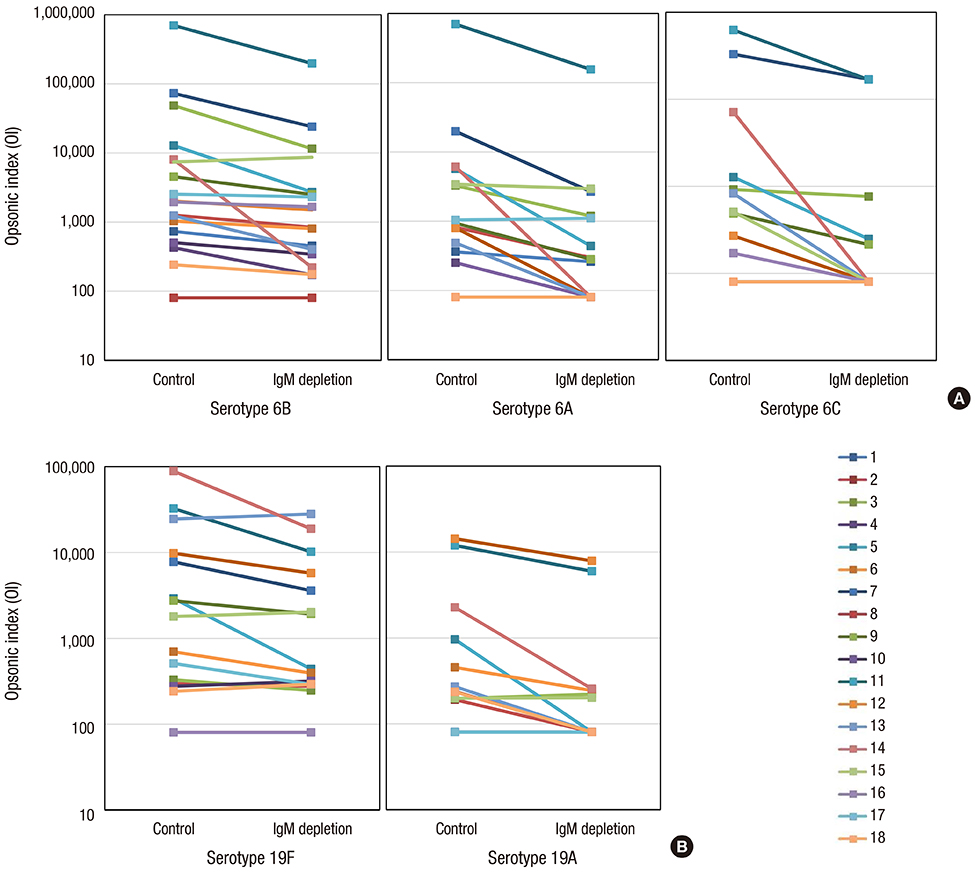
- Although it is well known that pneumococcal conjugate vaccines provide cross-protection against some vaccine-related serotypes, these mechanisms are still unclear. This study was performed to investigate the role of cross-protective IgM antibodies against vaccine-related serotypes 6A, 6C, and 19A induced in children aged 12-23 months after immunization with 7-valent pneumococcal conjugate vaccine (PCV7). We obtained serum samples from 18 Korean children aged 12-23 months after a PCV7 booster immunization. The serum IgG and IgM concentrations of serotypes 6B and 19F were measured by enzyme-linked immunosorbent assay (ELISA) in serum. The opsonic indices (OIs) against vaccine serotypes 6B and 19F and vaccine-related serotypes 6A, 6C, and 19A were determined by an opsonophagocytic killing assay (OPA) in IgM-depleted and control serum. Both IgG and IgM antibodies in ELISA and opsonic indices in OPA against serotypes 6B and 19F were demonstrated in the immune serum. IgM depletion decreased the OIs against vaccine serotypes 6B (geometric means of OIs (GMIs) of 3,009 vs. 1,396, 38% reduction) and 19F (1,117 vs. 750, 36% reduction). In addition, IgM depletion markedly decreased the OIs against vaccine-related serotypes 6A (GMIs of 961 vs. 329, 70% reduction), 6C (432 vs. 185, 72% reduction), and 19A (301 vs. 166, 58% reduction). The booster immunization PCV7 induced protective antibodies in the form of both IgG and IgM isotypes. IgM antibodies contributed to eliciting cross-protection against vaccine-related serotypes as well as against vaccine serotypes.
Keyword
MeSH Terms
-
Antibodies, Bacterial/blood
Antibodies, Neutralizing/blood
Enzyme-Linked Immunosorbent Assay
Heptavalent Pneumococcal Conjugate Vaccine/*immunology
Humans
Immunoglobulin M/*blood
Infant
Pneumococcal Infections/*prevention & control
Pneumococcal Vaccines/*immunology
Serogroup
Streptococcus pneumoniae/immunology
Antibodies, Bacterial
Antibodies, Neutralizing
Heptavalent Pneumococcal Conjugate Vaccine
Immunoglobulin M
Pneumococcal Vaccines
Figure
Cited by 1 articles
-
Evaluation of Antibodies Against Haemophilus influenzae Type b in Korean Adults
Ji Hyen Lee, Han Wool Kim, Soyoung Lee, Kyung-Hyo Kim
Pediatr Infect Vaccine. 2017;24(3):125-133. doi: 10.14776/piv.2017.24.3.125.
Reference
-
1. Lee JH, Cho HK, Kim KH, Kim CH, Kim DS, Kim KN, Cha SH, Oh SH, Hur JK, Kang JH, et al. Etiology of invasive bacterial infections in immunocompetent children in Korea (1996-2005): a retrospective multicenter study. J Korean Med Sci. 2011; 26:174–183.2. World Health Organization. Pneumococcal conjugate vaccine for childhood immunization--WHO position paper. Wkly Epidemiol Rec. 2007; 82:93–104.3. Centers for Disease Control and Prevention (CDC). Invasive pneumococcal disease in children 5 years after conjugate vaccine introduction--eight states, 1998-2005. MMWR Morb Mortal Wkly Rep. 2008; 57:144–148.4. Hsu KK, Shea KM, Stevenson AE, Pelton SI; Massachusetts Department of Public Health. Changing serotypes causing childhood invasive pneumococcal disease: Massachusetts, 2001-2007. Pediatr Infect Dis J. 2010; 29:289–293.5. Väkeväinen M, Eklund C, Eskola J, Käyhty H. Cross-reactivity of antibodies to type 6B and 6A polysaccharides of Streptococcus pneumoniae, evoked by pneumococcal conjugate vaccines, in infants. J Infect Dis. 2001; 184:789–793.6. Lee H, Cha JH, Nahm MH, Burton RL, Kim KH. The 7-valent pneumococcal conjugate vaccine elicits cross-functional opsonophagocytic killing responses to Streptococcus pneumoniae serotype 6D in children. BMC Infect Dis. 2013; 13:474.7. Hausdorff WP, Hoet B, Schuerman L. Do pneumococcal conjugate vaccines provide any cross-protection against serotype 19A? BMC Pediatr. 2010; 10:4.8. Lee HJ, Park SE, Kim KH. Immune response to 19A serotype after immunization of 19F containing pneumococcal conjugate vaccine in Korean children aged 12-23 months. Korean J Pediatr. 2011; 54:163–168.9. Lee H, Nahm MH, Burton R, Kim KH. Immune response in infants to the heptavalent pneumococcal conjugate vaccine against vaccine-related serotypes 6A and 19A. Clin Vaccine Immunol. 2009; 16:376–381.10. Janeway CA, Travers P, Walport M, Shlomchik MJ. The humoral immune response. In : Janeway CA, Travers P, Walport M, Shlomchik MJ, editors. Immunobiology: the Immune System in Health and Disease. 6th ed. New York, NY: Garland Science;2005. p. 367–408.11. Pollack M, Koles NL, Preston MJ, Brown BJ, Pier GB. Functional properties of isotype-switched immunoglobulin M (IgM) and IgG monoclonal antibodies to Pseudomonas aeruginosa lipopolysaccharide. Infect Immun. 1995; 63:4481–4488.12. Raff HV, Bradley C, Brady W, Donaldson K, Lipsich L, Maloney G, Shuford W, Walls M, Ward P, Wolff E, et al. Comparison of functional activities between IgG1 and IgM class-switched human monoclonal antibodies reactive with group B streptococci or Escherichia coli K1. J Infect Dis. 1991; 163:346–354.13. Park S, Nahm MH. Older adults have a low capacity to opsonize pneumococci due to low IgM antibody response to pneumococcal vaccinations. Infect Immun. 2011; 79:314–320.14. Simell B, Nurkka A, Ekström N, Givon-Lavi N, Käyhty H, Dagan R. Serum IgM antibodies contribute to high levels of opsonophagocytic activities in toddlers immunized with a single dose of the 9-valent pneumococcal conjugate vaccine. Clin Vaccine Immunol. 2012; 19:1618–1623.15. Burton RL, Nahm MH. Development and validation of a fourfold multiplexed opsonization assay (MOPA4) for pneumococcal antibodies. Clin Vaccine Immunol. 2006; 13:1004–1009.16. Wang D, Burton RL, Nahm MH, Soong SJ. A four-parameter logistic model for estimating titers of functional multiplexed pneumococcal opsonophagocytic killing assay. J Biopharm Stat. 2008; 18:307–325.17. Romero-Steiner S, Frasch CE, Carlone G, Fleck RA, Goldblatt D, Nahm MH. Use of opsonophagocytosis for serological evaluation of pneumococcal vaccines. Clin Vaccine Immunol. 2006; 13:165–169.18. Shyur SD, Raff HV, Bohnsack JF, Kelsey DK, Hill HR. Comparison of the opsonic and complement triggering activity of human monoclonal IgG1 and IgM antibody against group B streptococci. J Immunol. 1992; 148:1879–1884.19. Shi Y, Yamazaki T, Okubo Y, Uehara Y, Sugane K, Agematsu K. Regulation of aged humoral immune defense against pneumococcal bacteria by IgM memory B cell. J Immunol. 2005; 175:3262–3267.20. Notkins AL. Polyreactivity of antibody molecules. Trends Immunol. 2004; 25:174–179.21. Dentinger CM, Hennessy TW, Bulkow LR, Reasonover AL, Romero-Steiner S, Holder PF, de Leon PG, Carlone GM, Parks DJ, Parkinson AJ, et al. Immunogenicity and reactogenicity to Haemophilus influenzae type B (Hib) conjugate vaccine among rural Alaska adults. Hum Vaccin. 2006; 2:24–28.22. Perez-Shibayama C, Gil-Cruz C, Pastelin-Palacios R, Cervantes-Barragan L, Hisaki E, Chai Q, Onder L, Scandella E, Regen T, Waisman A, et al. IFN-γ-producing CD4+ T cells promote generation of protective germinal center-derived IgM+ B cell memory against Salmonella Typhi . J Immunol. 2014; 192:5192–5200.23. Tsai TF, Borrow R, Gnehm HE, Vaudaux B, Heininger U, Desgrandchamps D, Aebi C, Balmer P, Pedersen RD, Fritzell B, et al. Early appearance of bactericidal antibodies after polysaccharide challenge of toddlers primed with a group C meningococcal conjugate vaccine: what is its role in the maintenance of protection? Clin Vaccine Immunol. 2006; 13:854–861.24. Yu X, Gray B, Chang S, Ward JI, Edwards KM, Nahm MH. Immunity to cross-reactive serotypes induced by pneumococcal conjugate vaccines in infants. J Infect Dis. 1999; 180:1569–1576.25. Kim NH, Lee J, Lee SJ, Lee H, Kim KH, Park SE, Lee HJ. Immunogenicity and safety of pneumococcal 7-valent conjugate vaccine (diphtheria CRM197 protein conjugate; Prevenar™) in Korean infants: differences that are found in Asian children. Vaccine. 2007; 25:7858–7865.26. Centers for Disease Control and Prevention (CDC). Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine for adults with immunocompromising conditions: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep. 2012; 61:816–819.27. Centers for Disease Control and Prevention (CDC). Licensure of a 13-valent pneumococcal conjugate vaccine (PCV13) and recommendations for use among children - Advisory Committee on Immunization Practices (ACIP), 2010. MMWR Morb Mortal Wkly Rep. 2010; 59:258–261.28. Silfverdal SA, Hogh B, Bergsaker MR, Skerlikova H, Lommel P, Borys D, Schuerman L. Immunogenicity of a 2-dose priming and booster vaccination with the 10-valent pneumococcal nontypeable Haemophilus influenzae protein D conjugate vaccine. Pediatr Infect Dis J. 2009; 28:e276–82.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cross-reaction of 6B and 19F Specific Antibodies to Serotypes 6A, 6C, and 19A after Immunization with 7-valent Pneumococcal Conjugate Vaccine in Korean Children Aged 12-23 Months
- Immunogenicity of 7-valent pneumococcal conjugate vaccine related to booster immunization in Korean children
- Efficacy and effectiveness of extended-valency pneumococcal conjugate vaccines
- Indirect Effects of Pneumococcal Conjugate Vaccines in National Immunization Programs for Children on Adult Pneumococcal Disease
- An Update on Pneumococcal Vaccination