J Adv Prosthodont.
2014 Apr;6(2):96-102. 10.4047/jap.2014.6.2.96.
Cell attachment and proliferation of bone marrow-derived osteoblast on zirconia of various surface treatment
- Affiliations
-
- 1Department of Prosthodontics, School of Dentistry, Kyung Hee University, Seoul, Republic of Korea. ahranp@khu.ac.kr
- 2Department of Oral Anatomy, Dental School, Gangneung-Wonju National University, Wonju, Republic of Korea.
- KMID: 2233336
- DOI: http://doi.org/10.4047/jap.2014.6.2.96
Abstract
- PURPOSE
This study was performed to characterize the effects of zirconia coated with calcium phosphate and hydroxyapatite compared to smooth zirconia after bone marrow-derived osteoblast culture.
MATERIALS AND METHODS
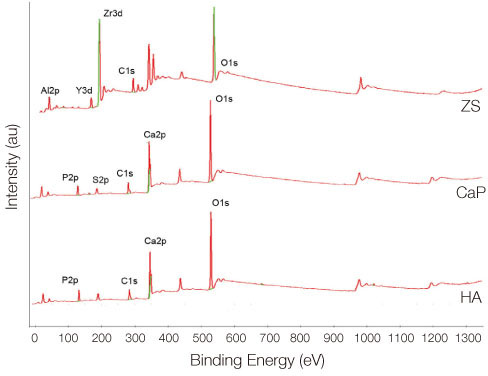
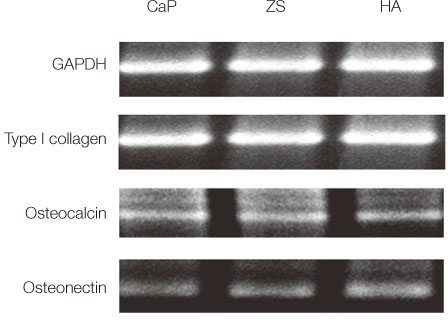
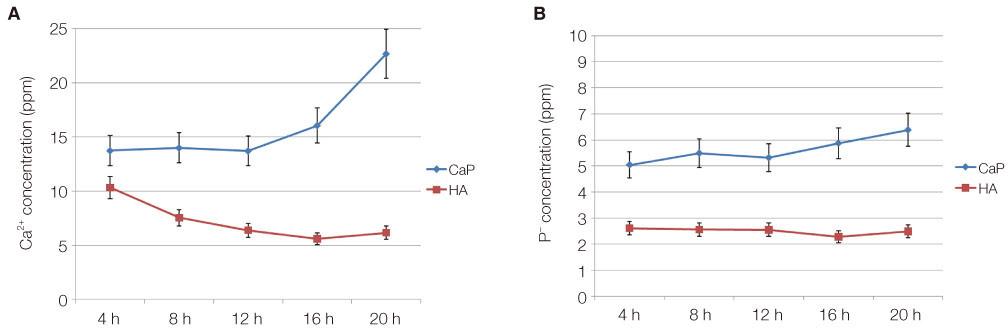
Bone marrow-derived osteoblasts were cultured on (1) smooth zirconia, (2) zirconia coated with calcium phosphate (CaP), and (3) zirconia coated with hydroxyapatite (HA). The tetrazolium-based colorimetric assay (MTT test) was used for cell proliferation evaluation. Scanning electron microscopy (SEM) and alkaline phosphatase (ALP) activity was measured to evaluate the cellular morphology and differentiation rate. X-ray photoelectron spectroscopy (XPS) was employed for the analysis of surface chemistry. The genetic expression of the osteoblasts and dissolution behavior of the coatings were observed. Assessment of the significance level of the differences between the groups was done with analysis of variance (ANOVA).
RESULTS
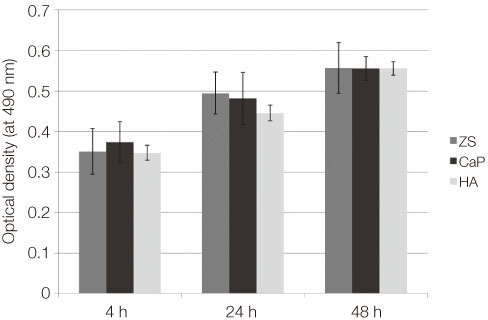
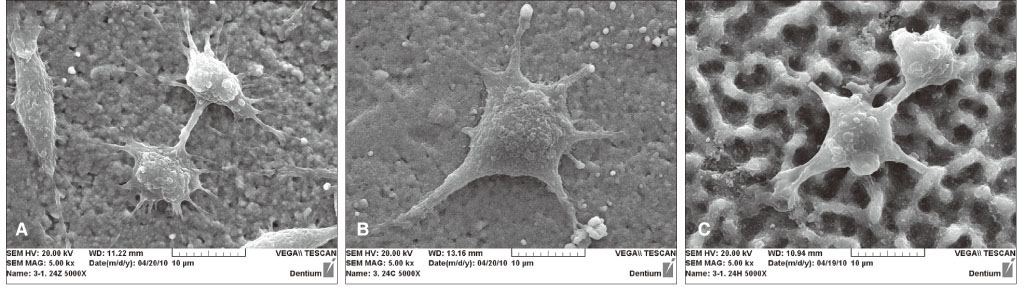
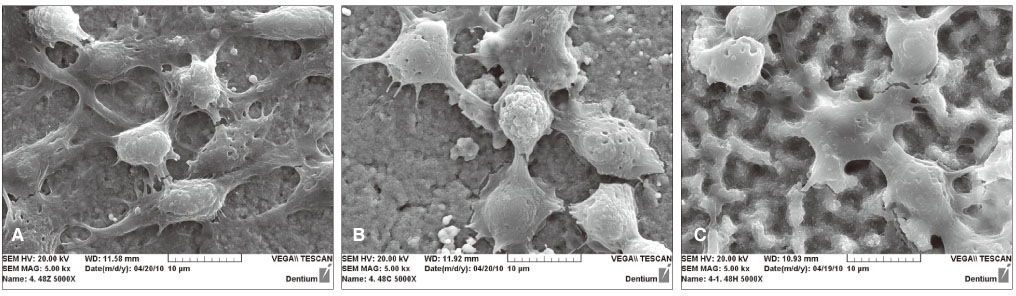
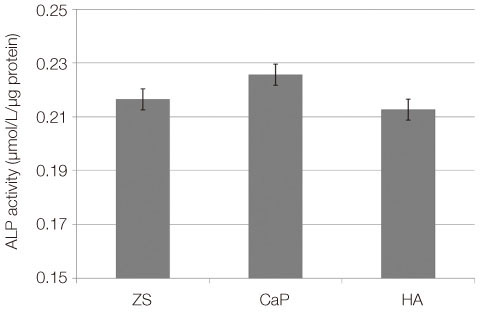
From the MTT assay, no significant difference between smooth and surface coated zirconia was found (P>.05). From the SEM image, cells on all three groups of discs were sporadically triangular or spread out in shape with formation of filopodia. From the ALP activity assay, the optical density of osteoblasts on smooth zirconia discs was higher than that on surface treated zirconia discs (P>.05). Most of the genes related to cell adhesion showed similar expression level between smooth and surface treated zirconia. The dissolution rate was higher with CaP than HA coating.
CONCLUSION
The attachment and growth behavior of bone-marrow-derived osteoblasts cultured on smooth surface coated zirconia showed comparable results. However, the HA coating showed more time-dependent stability compared to the CaP coating.
Keyword
MeSH Terms
Figure
Reference
-
1. Pae A, Lee H, Kim HS, Kwon YD, Woo YH. Attachment and growth behaviour of human gingival fibroblasts on titanium and zirconia ceramic surfaces. Biomed Mater. 2009; 4:025005.2. Mustafa K, Silva Lopez B, Hultenby K, Wennerberg A, Arvidson K. Attachment and proliferation of human oral fibroblasts to titanium surfaces blasted with TiO2 particles. A scanning electron microscopic and histomorphometric analysis. Clin Oral Implants Res. 1998; 9:195–207.3. Pae A, Kim SS, Kim HS, Woo YH. Osteoblast-like cell attachment and proliferation on turned, blasted, and anodized titanium surfaces. Int J Oral Maxillofac Implants. 2011; 26:475–481.4. Orsini G, Assenza B, Scarano A, Piattelli M, Piattelli A. Surface analysis of machined versus sandblasted and acid-etched titanium implants. Int J Oral Maxillofac Implants. 2000; 15:779–784.5. Bächle M, Butz F, Hübner U, Bakalinis E, Kohal RJ. Behavior of CAL72 osteoblast-like cells cultured on zirconia ceramics with different surface topographies. Clin Oral Implants Res. 2007; 18:53–59.6. Gahlert M, Gudehus T, Eichhorn S, Steinhauser E, Kniha H, Erhardt W. Biomechanical and histomorphometric comparison between zirconia implants with varying surface textures and a titanium implant in the maxilla of miniature pigs. Clin Oral Implants Res. 2007; 18:662–668.7. Ichikawa Y, Akagawa Y, Nikai H, Tsuru H. Tissue compatibility and stability of a new zirconia ceramic in vivo. J Prosthet Dent. 1992; 68:322–326.8. Josset Y, Oum'Hamed Z, Zarrinpour A, Lorenzato M, Adnet JJ, Laurent-Maquin D. In vitro reactions of human osteoblasts in culture with zirconia and alumina ceramics. J Biomed Mater Res. 1999; 47:481–493.9. Scarano A, Piattelli M, Caputi S, Favero GA, Piattelli A. Bacterial adhesion on commercially pure titanium and zirconium oxide disks: an in vivo human study. J Periodontol. 2004; 75:292–296.10. Rimondini L, Cerroni L, Carrassi A, Torricelli P. Bacterial colonization of zirconia ceramic surfaces: an in vitro and in vivo study. Int J Oral Maxillofac Implants. 2002; 17:793–798.11. Degidi M, Artese L, Scarano A, Perrotti V, Gehrke P, Piattelli A. Inflammatory infiltrate, microvessel density, nitric oxide synthase expression, vascular endothelial growth factor expression, and proliferative activity in peri-implant soft tissues around titanium and zirconium oxide healing caps. J Periodontol. 2006; 77:73–80.12. Ismail FS, Rohanizadeh R, Atwa S, Mason RS, Ruys AJ, Martin PJ, Bendavid A. The influence of surface chemistry and topography on the contact guidance of MG63 osteoblast cells. J Mater Sci Mater Med. 2007; 18:705–714.13. Mustafa K, Wennerberg A, Arvidson K, Messelt EB, Haag P, Karlsson S. Influence of modifying and veneering the surface of ceramic abutments on cellular attachment and proliferation. Clin Oral Implants Res. 2008; 19:1178–1187.14. Hulbert SF, Young FA, Mathews RS, Klawitter JJ, Talbert CD, Stelling FH. Potential of ceramic materials as permanently implantable skeletal prostheses. J Biomed Mater Res. 1970; 4:433–456.15. Maniatopoulos C, Sodek J, Melcher AH. Bone formation in vitro by stromal cells obtained from bone marrow of young adult rats. Cell Tissue Res. 1988; 254:317–330.16. Kim HW, Kong YM, Bae CJ, Noh YJ, Kim HE. Sol-gel derived fluor-hydroxyapatite biocoatings on zirconia substrate. Biomaterials. 2004; 25:2919–2926.17. Takemoto M, Fujibayashi S, Neo M, Suzuki J, Kokubo T, Nakamura T. Bone-bonding ability of a hydroxyapatite coated zirconia-alumina nanocomposite with a microporous surface. J Biomed Mater Res A. 2006; 78:693–701.18. Boyan BD, Batzer R, Kieswetter K, Liu Y, Cochran DL, Szmuckler-Moncler S, Dean DD, Schwartz Z. Titanium surface roughness alters responsiveness of MG63 osteoblast-like cells to 1 alpha,25-(OH)2D3. J Biomed Mater Res. 1998; 39:77–85.19. Jarcho M. Calcium phosphate ceramics as hard tissue prosthetics. Clin Orthop Relat Res. 1981; 157:259–278.20. Sennerby L, Dasmah A, Larsson B, Iverhed M. Bone tissue responses to surface-modified zirconia implants: A histomorphometric and removal torque study in the rabbit. Clin Implant Dent Relat Res. 2005; 7:S13–S20.21. Wang G, Meng F, Ding C, Chu PK, Liu X. Microstructure, bioactivity and osteoblast behavior of monoclinic zirconia coating with nanostructured surface. Acta Biomater. 2010; 6:990–1000.22. Sun L, Berndt CC, Gross KA, Kucuk A. Material fundamentals and clinical performance of plasma-sprayed hydroxyapatite coatings: a review. J Biomed Mater Res. 2001; 58:570–592.23. Zeng H, Lacefield WR. The study of surface transformation of pulsed laser deposited hydroxyapatite coatings. J Biomed Mater Res. 2000; 50:239–247.24. de Groot K, de Putter C, Smitt P, Driessen A. Mechanical failure of artificial teeth made of dense calcium hydroxyapatite. Sci Ceram. 1981; 11:433–437.25. Cui FZ, Luo ZS, Feng QL. Highly adhesive hydroxyapatite coatings on titanium alloy formed by ion beam assisted deposition. J Mater Sci Mater Med. 1997; 8:403–405.26. Benzaid R, Chevalier J, Saâdaoui M, Fantozzi G, Nawa M, Diaz LA, Torrecillas R. Fracture toughness, strength and slow crack growth in a ceria stabilized zirconia-alumina nanocomposite for medical applications. Biomaterials. 2008; 29:3636–3641.27. Piattelli A, Degidi M, Paolantonio M, Mangano C, Scarano A. Residual aluminum oxide on the surface of titanium implants has no effect on osseointegration. Biomaterials. 2003; 24:4081–4089.28. Zinelis S, Thomas A, Syres K, Silikas N, Eliades G. Surface characterization of zirconia dental implants. Dent Mater. 2010; 26:295–305.29. Jha LJ, Best SM, Knowles JC, Rehman I, Santos JD, Bonfield W. Preparation and characterization of fluoride-substituted apatites. J Mater Sci Mater Med. 1997; 8:185–191.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Acid etching of glass-infiltrated zirconia and its biological response
- Cytotoxicity and biocompatibility of high mol% yttria containing zirconia
- Response of osteoblast-like cells on titanium surface treatment
- Clonal Heterogeneity of Human Bone Marrow-Derived Stromal Cells
- On the effects of the characteristics of the titanium oxide to the osteoblast cell culture