Korean Circ J.
2014 Jul;44(4):233-242. 10.4070/kcj.2014.44.4.233.
Histological and Biochemical Comparisons between Right Atrium and Left Atrium in Patients with Mitral Valvular Atrial Fibrillation
- Affiliations
-
- 1Division of Cardiology, Yonsei University Health System, Seoul, Korea. hnpak@yuhs.ac ygko@yuhs.ac
- KMID: 2223884
- DOI: http://doi.org/10.4070/kcj.2014.44.4.233
Abstract
- BACKGROUND AND OBJECTIVES
It has been known that the dominant driver of atrial fibrillation (AF) exists in the left atrium (LA) and the incidence of systemic thromboembolism is higher than that of pulmonary thromboembolism in patients with AF. Therefore, we hypothesized that histological and biochemical characteristics of the LA and the right atrium (RA) are different in patients with mitral valvular AF.
SUBJECTS AND METHODS
We analyzed the histology and messenger ribonucleic acid (mRNA) or protein expression associated with endothelial function and thrombogenesis in 33 human atrial appendage tissues (20 LA tissues, 13 RA tissues) taken from 25 patients {57.7+/-11.3 years old, 44% males, AF: sinus rhythm (SR)=17:8} with mitral valve disease. We also performed whole mRNA quantification in 8 tissues (both LA and RA tissues from 4 patients) by using next generation sequencing (NGS).
RESULTS
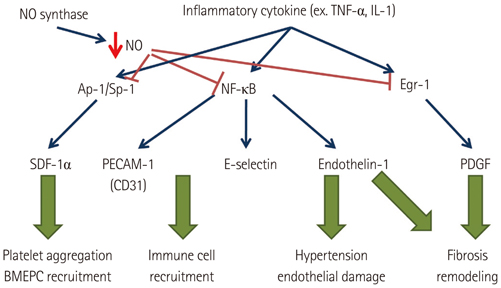
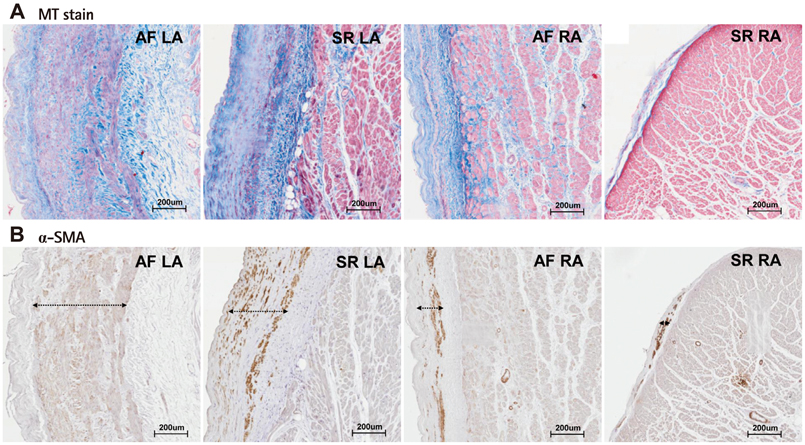
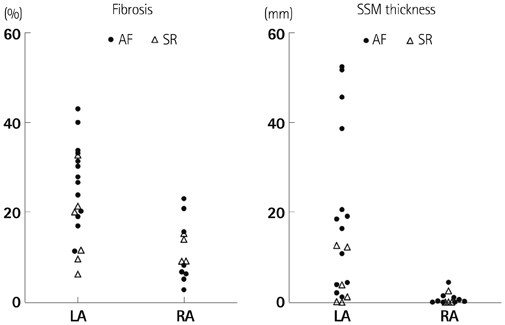
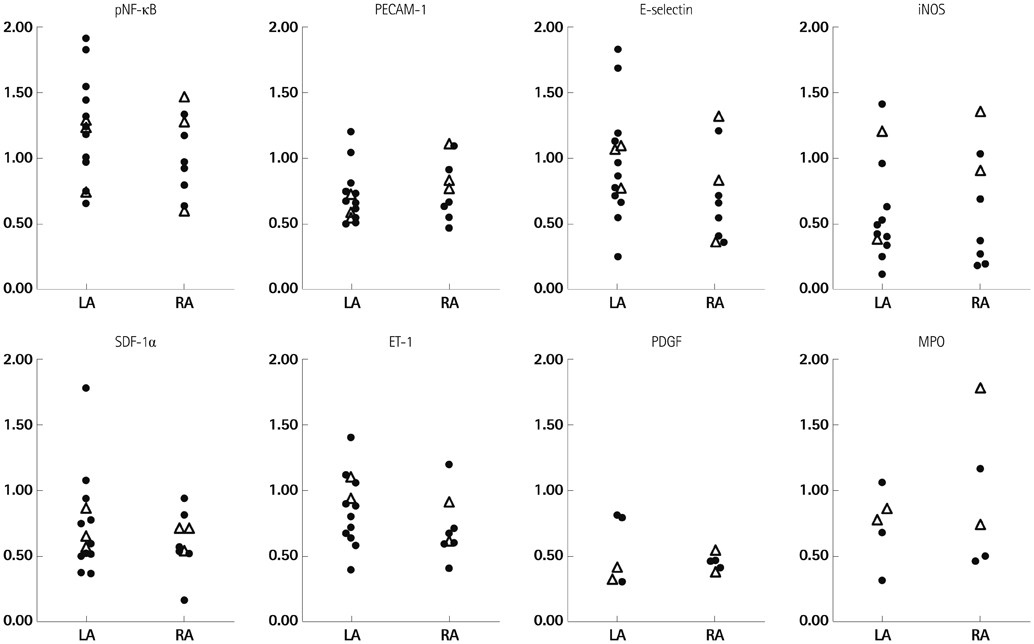
1) The degree of fibrosis (p=0.001) and subendocardial smooth muscle thickness (p=0.004) were significantly greater in the LA than in the RA. 2) More advanced matrix fibrosis was found in the LA of patients with AF than in the LA of patients with SR (p=0.046), but not in the RA. 3) There was no LA-RA difference in protein (Western blot) and mRNA {quantitative real-time polymerase chain reaction (qRT-PCR)} expressions of NF-kappaB, 3-NT, CD31, E-selectin, inducible NO synthase, stromal cell-derived factor-1alpha, Endothelin-1, platelet-derived growth factor, myeloperoxidase, or NCX, except for higher mRNA expression of HCN4 in the RA (qRT-PCR, p=0.026) and that of KCNN1 in the LA (NGS, p=0.016).
CONCLUSION
More advanced matrix and subendocardial remodeling were noticed in the LA than in the RA in patients with mitral valvular AF. However, the expressions of tissue factors associated with thrombogenesis were not significantly different between the RA and the LA.
MeSH Terms
-
Atrial Appendage
Atrial Fibrillation*
Chemokine CXCL12
E-Selectin
Endothelin-1
Fibrosis
Heart Atria*
Humans
Incidence
Male
Mitral Valve
Muscle, Smooth
NF-kappa B
Nitric Oxide Synthase
Peroxidase
Platelet-Derived Growth Factor
Pulmonary Embolism
Real-Time Polymerase Chain Reaction
RNA
RNA, Messenger
Thromboembolism
Thromboplastin
Chemokine CXCL12
E-Selectin
Endothelin-1
NF-kappa B
Nitric Oxide Synthase
Peroxidase
Platelet-Derived Growth Factor
RNA
RNA, Messenger
Thromboplastin
Figure
Reference
-
1. Sohara H, Amitani S, Kurose M, Miyahara K. Atrial fibrillation activates platelets and coagulation in a time-dependent manner: a study in patients with paroxysmal atrial fibrillation. J Am Coll Cardiol. 1997; 29:106–112.2. Li-Saw-Hee FL, Blann AD, Lip GY. Effect of degree of blood pressure on the hypercoagulable state in chronic atrial fibrillation. Am J Cardiol. 2000; 86:795–797. A93. Freestone B, Lip GY. The endothelium and atrial fibrillation. The prothrombotic state revisited. Hamostaseologie. 2008; 28:207–212.4. Cai H, Li Z, Goette A, et al. Downregulation of endocardial nitric oxide synthase expression and nitric oxide production in atrial fibrillation: potential mechanisms for atrial thrombosis and stroke. Circulation. 2002; 106:2854–2858.5. Dudley SC Jr, Hoch NE, McCann LA, et al. Atrial fibrillation increases production of superoxide by the left atrium and left atrial appendage: role of the NADPH and xanthine oxidases. Circulation. 2005; 112:1266–1273.6. Sung ML, Wu CC, Chang HI, et al. Shear stress inhibits homocysteine-induced stromal cell-derived factor-1 expression in endothelial cells. Circ Res. 2009; 105:755–763.7. Malmström RE, Settergren M, Pernow J. Endothelin attenuates endothelium-dependent platelet inhibition in man. Acta Physiol (Oxf). 2010; 198:441–448.8. Abi-Younes S, Sauty A, Mach F, Sukhova GK, Libby P, Luster AD. The stromal cell-derived factor-1 chemokine is a potent platelet agonist highly expressed in atherosclerotic plaques. Circ Res. 2000; 86:131–138.9. Chen CN, Chang SF, Lee PL, et al. Neutrophils, lymphocytes, and monocytes exhibit diverse behaviors in transendothelial and subendothelial migrations under coculture with smooth muscle cells in disturbed flow. Blood. 2006; 107:1933–1942.10. Urbich C, Dimmeler S. Endothelial progenitor cells: characterization and role in vascular biology. Circ Res. 2004; 95:343–353.11. Frenette PS, Moyna C, Hartwell DW, Lowe JB, Hynes RO, Wagner DD. Platelet-endothelial interactions in inflamed mesenteric venules. Blood. 1998; 91:1318–1324.12. Rosenblum WI, Murata S, Nelson GH, Werner PK, Ranken R, Harmon RC. Anti-CD31 delays platelet adhesion/aggregation at sites of endothelial injury in mouse cerebral arterioles. Am J Pathol. 1994; 145:33–36.13. Liao CH, Akazawa H, Tamagawa M, et al. Cardiac mast cells cause atrial fibrillation through PDGF-A-mediated fibrosis in pressure-overloaded mouse hearts. J Clin Invest. 2010; 120:242–253.14. Ohkita M, Takaoka M, Shiota Y, Nojiri R, Matsumura Y. Nitric oxide inhibits endothelin-1 production through the suppression of nuclear factor kappa B. Clin Sci (Lond). 2002; 103:Suppl 48. 68S–71S.15. Rudolph V, Andrié RP, Rudolph TK, et al. Myeloperoxidase acts as a profibrotic mediator of atrial fibrillation. Nat Med. 2010; 16:470–474.16. Park JH, Pak HN, Lee S, Park HK, Seo JW, Chang BC. The clinical significance of the atrial subendocardial smooth muscle layer and cardiac myofibroblasts in human atrial tissue with valvular atrial fibrillation. Cardiovasc Pathol. 2013; 22:58–64.17. Mortazavi A, Williams BA, McCue K, Schaeffer L, Wold B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods. 2008; 5:621–628.18. De Caterina R, Libby P, Peng HB, et al. Nitric oxide decreases cytokine-induced endothelial activation. Nitric oxide selectively reduces endothelial expression of adhesion molecules and proinflammatory cytokines. J Clin Invest. 1995; 96:60–68.19. Turu MM, Slevin M, Matou S, et al. C-reactive protein exerts angiogenic effects on vascular endothelial cells and modulates associated signalling pathways and gene expression. BMC Cell Biol. 2008; 9:47.20. Burstein B, Libby E, Calderone A, Nattel S. Differential behaviors of atrial versus ventricular fibroblasts: a potential role for platelet-derived growth factor in atrial-ventricular remodeling differences. Circulation. 2008; 117:1630–1641.21. Goldsmith I, Kumar P, Carter P, Blann AD, Patel RL, Lip GY. Atrial endocardial changes in mitral valve disease: a scanning electron microscopy study. Am Heart J. 2000; 140:777–784.22. Modrego J, Maroto L, Tamargo J, et al. Comparative expression of proteins in left and right atrial appendages from patients with mitral valve disease at sinus rhythm and atrial fibrillation. J Cardiovasc Electrophysiol. 2010; 21:859–868.23. Fuster V, Rydén LE, Asinger RW, et al. ACC/AHA/ESC guidelines for the management of patients with atrial fibrillation: executive summary. A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the European Society of Cardiology Committee for Practice Guidelines and Policy Conferences (Committee to Develop Guidelines for the Management of Patients With Atrial Fibrillation): developed in Collaboration With the North American Society of Pacing and Electrophysiology. J Am Coll Cardiol. 2001; 38:1231–1266.24. Franco D, Campione M. The role of Pitx2 during cardiac development. Linking left-right signaling and congenital heart diseases. Trends Cardiovasc Med. 2003; 13:157–163.25. King T, Bland Y, Webb S, Barton S, Brown NA. Expression of Peg1 (Mest) in the developing mouse heart: involvement in trabeculation. Dev Dyn. 2002; 225:212–215.26. Corradi D, Maestri R, Macchi E, Callegari S. The atria: from morphology to function. J Cardiovasc Electrophysiol. 2011; 22:223–235.27. Ho SY, McCarthy KP. Anatomy of the left atrium for interventional electrophysiologists. Pacing Clin Electrophysiol. 2010; 33:620–627.28. Subramaniam B, Riley MF, Panzica PJ, Manning WJ. Transesophageal echocardiographic assessment of right atrial appendage anatomy and function: comparison with the left atrial appendage and implications for local thrombus formation. J Am Soc Echocardiogr. 2006; 19:429–433.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Relation of Right Atrial Pathology to Atrial Fibrillation in Mitral Valvular Disease
- Relation between Atrial Fibrillation and Echocardiographic Size of Left Atrium
- Relation between Left Atrial Size and Atrial Fibrillation
- Plain chest PA evaluation of left atrial thrombosis in mitral valvular disease
- Echocardiographic Study on the Mitral Valvular Heart Diseases