High EZH2 Protein Expression Is Associated with Poor Overall Survival in Patients with Luminal A Breast Cancer
- Affiliations
-
- 1Department of Pathology, Soonchunhyang University Cheonan Hospital, Soonchunhyang University College of Medicine, Cheonan, Korea. c84103@schmc.ac.kr
- 2Department of Surgery, Soonchunhyang University Cheonan Hospital, Soonchunhyang University College of Medicine, Cheonan, Korea.
- 3Division of Hemato-Oncology, Department of Internal Medicine, Soonchunhyang University Cheonan Hospital, Soonchunhyang University College of Medicine, Cheonan, Korea.
- KMID: 2176321
- DOI: http://doi.org/10.4048/jbc.2016.19.1.53
Abstract
- PURPOSE
The enhancer of zeste homologue 2 (EZH2) is a catalytic subunit of the polycomb repressive complex 2, a highly conserved histone methyltransferase. EZH2 overexpression has been implicated in various malignancies, including breast cancer, where is associated with poor outcomes. This study aims to clarify nuclear EZH2 expression levels in breast cancers using immunohistochemistry (IHC) and correlate these findings with clinicopathologic variables, including prognostic significance.
METHODS
IHC was performed on tissue microarrays of 432 invasive ductal carcinoma (IDC) tumors. Associations between EZH2 expression, clinicopathologic characteristics, and molecular subtype were retrospectively analyzed. The relationship between EZH2 protein expression in normal breast tissue and ductal carcinoma in situ (DCIS) was also assessed.
RESULTS
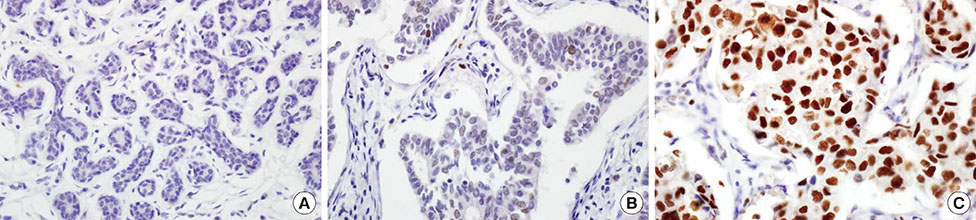
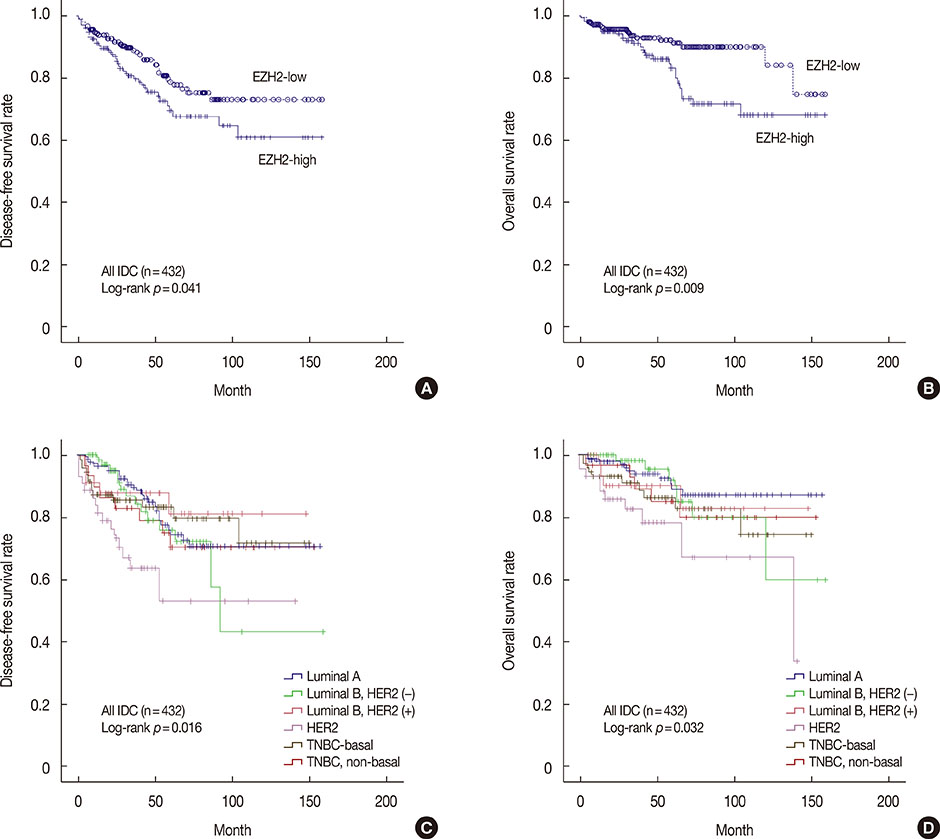
High EZH2 expression was demonstrated in 215 of 432 tumors (49.8%). EZH2 was more frequently expressed in DCIS and IDC than in normal breast tissue (p=0.001). High EZH2 expression significantly correlated with high histologic grade (p<0.001), large tumor size (p=0.014), advanced pathologic stage (p=0.006), negative estrogen receptor status (p<0.001), positive human epidermal growth factor receptor 2 (HER2) status (p<0.001), high Ki-67 staining index (p<0.001), positive cytokeratin 5/6 status (p=0.003), positive epidermal growth factor receptor status (p<0.001), and positive p53 status (p<0.001). Based on molecular subtypes, high EZH2 expression was significantly associated with HER2-negative luminal B, HER2-positive luminal B, and HER2 type and triple-negative basal cancers (p<0.001). In patients with luminal A, there was a significant trend toward shorter overall survival for those with tumors having high EZH2 expression compared to those with tumors having low EZH2 expression (p=0.045).
CONCLUSION
EZH2 is frequently upregulated in breast malignancies, and it may play an important role in cancer development and progression. Furthermore, EZH2 may be a prognostic marker, especially in patients with luminal A cancer.
MeSH Terms
-
Breast Neoplasms*
Breast*
Carcinoma, Ductal
Carcinoma, Intraductal, Noninfiltrating
Catalytic Domain
Estrogens
Histones
Humans
Immunohistochemistry
Keratins
Phenobarbital*
Polycomb Repressive Complex 2
Prognosis
Receptor, Epidermal Growth Factor
Retrospective Studies
Estrogens
Histones
Keratins
Phenobarbital
Polycomb Repressive Complex 2
Receptor, Epidermal Growth Factor
Figure
Cited by 3 articles
-
Expression of Programmed Death Receptor Ligand 1 with High Tumor-Infiltrating Lymphocytes Is Associated with Better Prognosis in Breast Cancer
Sang Byung Bae, Hyun Deuk Cho, Mee-Hye Oh, Ji-Hye Lee, Si-Hyong Jang, Soon Auck Hong, Junhun Cho, Sung Yong Kim, Sun Wook Han, Jong Eun Lee, Han Jo Kim, Hyun Ju Lee
J Breast Cancer. 2016;19(3):242-251. doi: 10.4048/jbc.2016.19.3.242.Clinicopathologic and Prognostic Significance of Transducin-Like Enhancer of Split 1 Protein Expression in Invasive Breast Cancer
Ji-Hye Lee, Sang Byung Bae, Mee-Hye Oh, Hyun Deuk Cho, Si-Hyong Jang, Soon Auck Hong, Junhun Cho, Sung Yong Kim, Sun Wook Han, Jong Eun Lee, Han Jo Kim, Hyun Ju Lee
J Breast Cancer. 2017;20(1):45-53. doi: 10.4048/jbc.2017.20.1.45.T-Cell Immunoglobulin Mucin 3 Expression on Tumor Infiltrating Lymphocytes as a Positive Prognosticator in Triple-Negative Breast Cancer
Kyung Do Byun, Hyo Jun Hwang, Ki Jae Park, Min Chan Kim, Se Heon Cho, Mi Ha Ju, Jin Hwa Lee, Jin Sook Jeong
J Breast Cancer. 2018;21(4):406-414. doi: 10.4048/jbc.2018.21.e61.
Reference
-
1. Margueron R, Reinberg D. The Polycomb complex PRC2 and its mark in life. Nature. 2011; 469:343–349.
Article2. Tan JZ, Yan Y, Wang XX, Jiang Y, Xu HE. EZH2: biology, disease, and structure-based drug discovery. Acta Pharmacol Sin. 2014; 35:161–174.
Article3. Yamaguchi H, Hung MC. Regulation and role of EZH2 in cancer. Cancer Res Treat. 2014; 46:209–222.
Article4. Ding L, Kleer CG. Enhancer of zeste 2 as a marker of preneoplastic progression in the breast. Cancer Res. 2006; 66:9352–9355.
Article5. Bohrer LR, Chen S, Hallstrom TC, Huang H. Androgens suppress EZH2 expression via retinoblastoma (RB) and p130-dependent pathways: a potential mechanism of androgen-refractory progression of prostate cancer. Endocrinology. 2010; 151:5136–5145.
Article6. Bachmann IM, Halvorsen OJ, Collett K, Stefansson IM, Straume O, Haukaas SA, et al. EZH2 expression is associated with high proliferation rate and aggressive tumor subgroups in cutaneous melanoma and cancers of the endometrium, prostate, and breast. J Clin Oncol. 2006; 24:268–273.
Article7. Weikert S, Christoph F, Köllermann J, Müller M, Schrader M, Miller K, et al. Expression levels of the EZH2 polycomb transcriptional repressor correlate with aggressiveness and invasive potential of bladder carcinomas. Int J Mol Med. 2005; 16:349–353.
Article8. Kim KH, Kim L, Choi SJ, Han JY, Kim JM, Chu YC, et al. The clinicopathological significance of epithelial mesenchymal transition associated protein expression in head and neck squamous cell carcinoma. Korean J Pathol. 2014; 48:263–269.
Article9. Ernst T, Chase AJ, Score J, Hidalgo-Curtis CE, Bryant C, Jones AV, et al. Inactivating mutations of the histone methyltransferase gene EZH2 in myeloid disorders. Nat Genet. 2010; 42:722–726.
Article10. Dubois S, Mareschal S, Picquenot JM, Viailly PJ, Bohers E, Cornic M, et al. Immunohistochemical and genomic profiles of diffuse large B-cell lymphomas: implications for targeted EZH2 inhibitor therapy? Oncotarget. 2015; 6:16712–16724.
Article11. Kleer CG, Cao Q, Varambally S, Shen R, Ota I, Tomlins SA, et al. EZH2 is a marker of aggressive breast cancer and promotes neoplastic transformation of breast epithelial cells. Proc Natl Acad Sci U S A. 2003; 100:11606–11611.
Article12. Knudsen ES, Dervishaj O, Kleer CG, Pajak T, Schwartz GF, Witkiewicz AK. EZH2 and ALDH1 expression in ductal carcinoma in situ: complex association with recurrence and progression to invasive breast cancer. Cell Cycle. 2013; 12:2042–2050.
Article13. Alford SH, Toy K, Merajver SD, Kleer CG. Increased risk for distant metastasis in patients with familial early-stage breast cancer and high EZH2 expression. Breast Cancer Res Treat. 2012; 132:429–437.
Article14. Lakhani SR, Ellis IO, Schnitt SJ, Tan PH, van de Vijver MJ. WHO Classification of Tumours of the Breast. 4th ed. Lyon: International Agency for Research on Cancer;2012.15. Allred DC, Harvey JM, Berardo M, Clark GM. Prognostic and predictive factors in breast cancer by immunohistochemical analysis. Mod Pathol. 1998; 11:155–168.16. Ding L, Erdmann C, Chinnaiyan AM, Merajver SD, Kleer CG. Identification of EZH2 as a molecular marker for a precancerous state in morphologically normal breast tissues. Cancer Res. 2006; 66:4095–4099.
Article17. Raaphorst FM, Meijer CJ, Fieret E, Blokzijl T, Mommers E, Buerger H, et al. Poorly differentiated breast carcinoma is associated with increased expression of the human polycomb group EZH2 gene. Neoplasia. 2003; 5:481–488.
Article18. Hussein YR, Sood AK, Bandyopadhyay S, Albashiti B, Semaan A, Nahleh Z, et al. Clinical and biological relevance of enhancer of zeste homolog 2 in triple-negative breast cancer. Hum Pathol. 2012; 43:1638–1644.
Article19. Bae WK, Yoo KH, Lee JS, Kim Y, Chung IJ, Park MH, et al. The methyltransferase EZH2 is not required for mammary cancer development, although high EZH2 and low H3K27me3 correlate with poor prognosis of ER-positive breast cancers. Mol Carcinog. 2015; 54:1172–1180.
Article20. Granit RZ, Gabai Y, Hadar T, Karamansha Y, Liberman L, Waldhorn I, et al. EZH2 promotes a bi-lineage identity in basal-like breast cancer cells. Oncogene. 2013; 32:3886–3895.
Article21. Athanassiadou AM, Tsipis A, Patsouris E, Gonidi M, Nicolopoulou-Stamati P, Chelidonis G, et al. Enhancer of zeste homologue 2 expression in breast carcinoma smears in relationship with p53, Ki-67 and other prognostic parameters. Acta Cytol. 2011; 55:180–186.
Article22. Tang X, Milyavsky M, Shats I, Erez N, Goldfinger N, Rotter V. Activated p53 suppresses the histone methyltransferase EZH2 gene. Oncogene. 2004; 23:5759–5769.
Article23. Bitler BG, Aird KM, Garipov A, Li H, Amatangelo M, Kossenkov AV, et al. Synthetic lethality by targeting EZH2 methyltransferase activity in ARID1A-mutated cancers. Nat Med. 2015; 21:231–238.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Expression of ERCC1, RRM1, and BRCA1 in Breast Cancer According to the Immunohistochemical Phenotypes
- Loss of Tumor Suppressor ARID1A Protein Expression Correlates with Poor Prognosis in Patients with Primary Breast Cancer
- The Clinical Value of PELP1 for Breast Cancer: A Comparison with Multiple Cancers and Analysis in Breast Cancer Subtypes
- Progesterone Receptor Expression as a Prognostic Factor in Luminal B Breast Cancer
- The different prognostic impact of age according to individual molecular subtypes in breast cancer