Infect Chemother.
2015 Dec;47(4):239-246. 10.3947/ic.2015.47.4.239.
The Incidence and Clinical Characteristics of Acute Serum Creatinine Elevation more than 1.5 mg/dL among the Patients Treated with Tenofovir/Emtricitabine-containing HAART Regimens
- Affiliations
-
- 1Department of Internal Medicine, Kyungpook National University School of Medicine, Daegu, Korea. changhha@knu.ac.kr
- KMID: 2170501
- DOI: http://doi.org/10.3947/ic.2015.47.4.239
Abstract
- BACKGROUND
The combination of tenofovir disoproxil fumarate (TDF) and emtricitabine (FTC) has been the first choice nucleoside reverse transcriptase inhibitor (NRTI) according to many reliable antiretroviral treatment (ART) guidelines because of its high efficacy. However, TDF-related renal toxicity reported in Western countries is a challenging issue regarding clinical use. We conducted this study to evaluate the incidence and characteristics of an acute increase in serum creatinine (Cr) level > 1.5 mg/dL among TDF/FTC-based highly active antiretroviral treatment (HAART)-treated patients.
MATERIALS AND METHODS
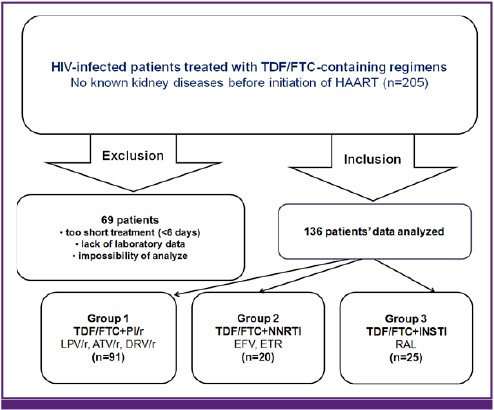
We retrospectively reviewed the medical records of 205 HIV-infected patients treated with TDF/FTC-containing regimens between 1 February 2010 and 30 April 2014. Three groups of TDF/FTC + ritonavir-boosted protease inhibitor (PI/r), TDF/FTC + non-nucleoside reverse transcriptase inhibitor (NNRTI), and TDF/FTC + integrase strand transfer inhibitor (INSTI), and three PI/r subgroups of TDF/FTC + lopinavir (LPV)/r, TDF/FTC + atazanavir (ATV)/r, TDF/FTC + darunavir (DRV)/r were evaluated.
RESULTS
A total 136 patients (91 in the TDF/FTC + PI/r group, 20 in the TDF/FTC + NNRTI group and 25 in the TDF/FTC + INSTI group) were included in the statistical analysis. Four cases (4.9%; all in the TDF/FTC + PI/r group) among 136 patients showed an acute increase in serum Cr more than 1.5 mg/dL, so the overall incidence was 2.8 cases per 100 patient-years. One case was a patient treated with TDF/FTC + LPV/r, and the others were treated with TDF/FTC + ATV/r. No case of an acute increase in serum Cr was observed in the TDF/FTC + DRV/r group. The incidence of serum Cr increase more than 1.5 mg/dL in TDF/FTC + PI/r group was 4.0 cases per 100 patient-years.
CONCLUSION
Although only a small number of patients were evaluated retrospectively from a single center, the TDF/FTC + PI/r regimen may have been related with relatively higher tendency of increment of serum Cr level. These findings reinforce the importance of close follow-ups of HIV-infected patients treated with the TDF/FTC + PI/r regimens.
Keyword
MeSH Terms
-
Anti-Retroviral Agents
Antiretroviral Therapy, Highly Active*
Atazanavir Sulfate
Creatinine*
Darunavir
Emtricitabine
Follow-Up Studies
HIV
Humans
Incidence*
Integrases
Lopinavir
Medical Records
Protease Inhibitors
Retrospective Studies
RNA-Directed DNA Polymerase
Tenofovir
Anti-Retroviral Agents
Creatinine
Integrases
Lopinavir
Protease Inhibitors
RNA-Directed DNA Polymerase
Figure
Reference
-
1. US Food and Drug Administration (FDA). Drug approval package: VIREAD (tenofovir disoproxil fumarate) tablets. Accessed 1 August 2015. Available at: http://www.accessdata.fda.gov/drugsatfda_docs/nda/2001/21-356_Viread.cfm.2. AIDSinfo. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. Accessed 28 December 201. Available at:http://aidsinfo.nih.gov/contentfiles/lvguidelines/adultandadolescentgl.pdf.3. Gnthard HF, Aberg JA, Eron JJ, Hoy JF, Telenti A, Benson CA, Burger DM, Cahn P, Gallant JE, Glesby MJ, Reiss P, Saag MS, Thomas DL, Jacobsen DM, Volberding PA. International Antiviral Society-USA Panel. Antiretroviral treatment of adult HIV infection: 2014 recommendations of the International Antiviral Society-USA Panel. JAMA. 2014; 312:410–425.4. Cooper RD, Wiebe N, Smith N, Keiser P, Naicker S, Tonelli M. Systematic review and meta-analysis: renal safety of tenofovir disoproxil fumarate in HIV-infected ptients. Clin Infect Dis. 2010; 51:496–505.
Article5. Rling J, Schmid H, Fischereder M, Draenert R, Goebel FD. HIV-associated renal diseases and highly active antiretroviral therapy-induced nephropathy. Clin Infect Dis. 2006; 42:1488–1495.
Article6. Choi A, Scherzer R, Bacchetti P, Tien PC, Saag MS, Gibert CL, Szczech LA, Grunfeld C, Shlipak MG. Cystatin C, albuminuria, and 5-year all-cause mortality in HIV-infected persons. Am J Kidney Dis. 2010; 56:872–882.
Article7. Choi AI, Li Y, Deeks SG, Grunfeld C, Volberding PA, Shlipak MG. Association between kidney function and albuminuria with cardiovascular events in HIV-infected persons. Circulation. 2010; 121:651–658.
Article8. Quinn KJ, Emerson CR, Dinsmore WW, Donnelly CM. Incidence of proximal renal tubular dysfunction in patients on tenofovir disoproxil fumarate. Int J STD AIDS. 2010; 21:150–151.
Article9. Nelson MR, Katlama C, Montaner JS, Cooper DA, Gazzard B, Clotet B, Lazzarin A, Schewe K, Lange J, Wyatt C, Curtis S, Chen SS, Smith S, Bischofberger N, Rooney JF. The safety of tenofovir disoproxil fumarate for the treatment of HIV infection in adults: the first 4 years. AIDS. 2007; 21:1273–1281.
Article10. Sax PE, Gallant JE, Klotman PE. Renal safety of tenofovir disoproxil fumarate. AIDS Read. 2007; 17:90–92. 99–104. c311. Créput C, Gonzalez-Canali G, Hill G, Piketty C, Kazatchkine M, Nochy D. Renal lesions in HIV-1-positive patient treated with tenofovir. AIDS. 2003; 17:935–937.
Article12. Rollot F, Nazal EM, Chauvelot-Moachon L, Kélaïdi C, Daniel N, Saba M, Abad S, Blanche P. Tenofovir-related Fanconi syndrome with nephrogenic diabetes insipidus in a patient with acquired immunodeficiency syndrome: the role of lopinavir-ritonavir-didanosine. Clin Infect Dis. 2003; 37:e174–e176.
Article13. Wood SM, Shah SS, Steenhoff AP, Meyers KE, Kaplan BS, Rutstein RM. Tenofovir-associated nephrotoxicity in two HIV-infected adolescent males. AIDS Patient Care STDS. 2009; 23:1–4.
Article14. Goicoechea M, Liu S, Best B, Sun S, Jain S, Kemper C, Witt M, Diamond C, Haubrich R, Louie S;. Greater tenofovir-associated renal function decline with protease inhibitor-based versus nonnucleoside reverse-transcriptase inhibitor-based therapy. J Infect Dis. 2008; 197:102–108.
Article15. Young J, Schafer J, Fux CA, Furrer H, Bernasconi E, Vernazza P, Calmy A, Cavassini M, Weber R, Battegay M, Bucher HC. Swiss HIV Cohort Study. Renal function in patients with HIV starting therapy with tenofovir and either efavirenz, lopinavir or atazanavir. AIDS. 2012; 26:567–575.
Article16. Horberg M, Tang B, Towner W, Silverberg M, Bersoff-Matcha S, Hurley L, Chang J, Blank J, Quesenberry C Jr, Klein D. Impact of tenofovir on renal function in HIV-infected, antiretroviral-naive patients. J Acquir Immune Defic Syndr. 2010; 53:62–69.
Article17. Scherzer R, Estrella M, Li Y, Choi AI, Deeks SG, Grunfeld C, Shlipak MG. Association of tenofovir exposure with kidney disease risk in HIV infection. AIDS. 2012; 26:867–875.
Article18. Calza L, Trapani F, Tedeschi S, Piergentili B, Manfredi R, Colangeli V, Viale P. Tenofovir-induced renal toxicity in 324 HIV-infected, antiretroviral-naive patients. Scand J Infect Dis. 2011; 43:656–660.
Article19. Kohler JJ, Hosseini SH, Hoying-Brandt A, Green E, Johnson DM, Russ R, Tran D, Raper CM, Santoianni R, Lewis W. Tenofovir renal toxicity targets mitochondria of renal proximal tubules. Lab Invest. 2009; 89:513–519.
Article20. Puls RL, Srasuebkul P, Petoumenos K, Boesecke C, Duncombe C, Belloso WH, Molina JM, Li L, Avihingsanon A, Gazzard B, Cooper DA, Emery S. Altair Study Group. Efavirenz versus boosted atazanavir or zidovudine and abacavir in antiretroviral treatment-naive, HIV-infected subjects: week 48 data from the Altair study. Clin Infect Dis. 2010; 51:855–864.
Article21. Ryom L, Mocroft A, Kirk O, Worm SW, Kamara DA, Reiss P, Ross M, Fux CA, Morlat P, Moranne O, Smith C, Lundgren JD. D:A:D Study Group. Association between antiretroviral exposure and renal impairment among HIV-positive persons with normal baseline renal function: the D:A:D study. J Infect Dis. 2013; 207:1359–1369.
Article22. Sax PE, Tierney C, Collier AC, Daar ES, Mollan K, Budhathoki C, Godfrey C, Jahed NC, Myers L, Katzenstein D, Farajallah A, Rooney JF, Ha B, Woodward WC, Feinberg J, Tashima K, Murphy RL, Fischl MA. AIDS Clinical Trials Group Study A5202 Team. Abacavir/lamivudine versus tenofovir DF/emtricitabine as part of combination regimens for initial treatment of HIV: final results. J Infect Dis. 2011; 204:1191–1201.
Article23. Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. Accessed 1 August 2015. Available from: https://aidsinfo.nih.gov/contentfiles/lvguidelines/adultandadolescentgl.pdf.24. Hall AM, Hendry BM, Nitsch D, Connolly JO. Tenofovir-associated kidney toxicity in HIV-infected patients: a review of the evidence. Am J Kidney Dis. 2011; 57:773–780.
Article25. Gallant JE, Moore RD. Renal function with use of a tenofovir-containing initial antiretroviral regimen. AIDS. 2009; 23:1971–1975.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Serial Changes of Serum Creatinine and Resistive Index to Predict Acute Rejection after Renal Transplantation
- The Effect of Withdrawal of Angiotensin II Blockers on Serum Creatinine and Potassium in Patients with Chronic Kidney Diseases
- Changes of Renal Function and Treatment after CABG in Patients with Elevated Serum Creatinine
- Cisplatin nephropathy in patients with lung cancer
- Effect of Long-term Fenofibrate Therapy on Serum Creatinine and Its Reversibility in Hypertriglyceridemic Patients with Hypertension