Anat Cell Biol.
2010 Jun;43(2):118-124. 10.5115/acb.2010.43.2.118.
Assessment of experimental saccular aneurysm using selective angiography in common carotid artery of rabbits
- Affiliations
-
- 1Department of Veterinary Radiology, College of Veterinary Medicine, Chungbuk National University, Cheongju, Korea.
- 2Medical Research Center, Yonsei University College of Medicine, Seoul, Korea. Snumouse@snu.ac.kr
- 3Department of Veterinary Anatomy and Cell Biology, College of Veterinary Medicine, Seoul National University, Seoul, Korea.
- 4Department of Veterinary Internal Medicine, College of Veterinary Medicine, Jeju National University, Jeju, Korea.
- 5National Cancer Center, Koyang, Korea.
- 6Pediatric Oncology Branch, Centre for Cancer Research, National Cancer Institute, National Institutes of Health, Bethesda, USA.
- 7Department of Veterinary Radiology, College of Veterinary Medicine, Chonbuk National University, Jeonju, Korea.
- 8Department of Veterinary Radiology, College of Veterinary Medicine, Chungnam National University, Daejeon, Korea.
- 9Department of Veterinary Surgery, College of Veterinary Medicine, Seoul National University, Seoul, Korea.
- 10Veterinary Radiology, College of Veterinary Medicine, Seoul National University, Seoul, Korea.
- 11Zoo and Wild Animal Medicine, College of Veterinary Medicine, Seoul National University, Seoul, Korea.
- 12Department of Medical Genetics, College of Medicine, Hallym University, Chuncheon, Korea.
- KMID: 2168884
- DOI: http://doi.org/10.5115/acb.2010.43.2.118
Abstract
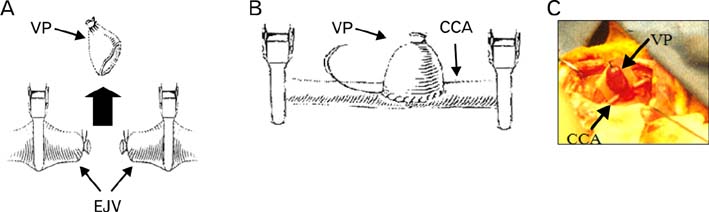
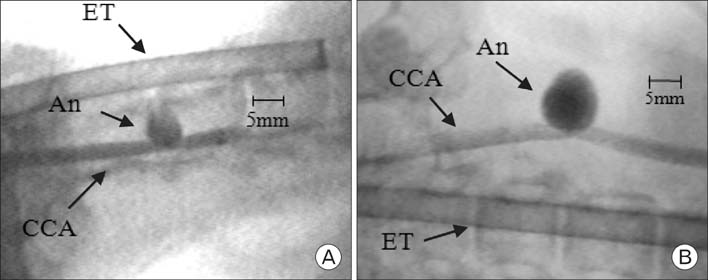
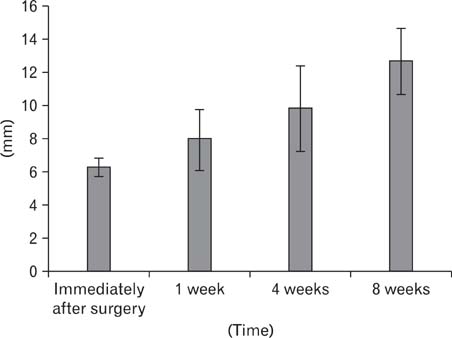
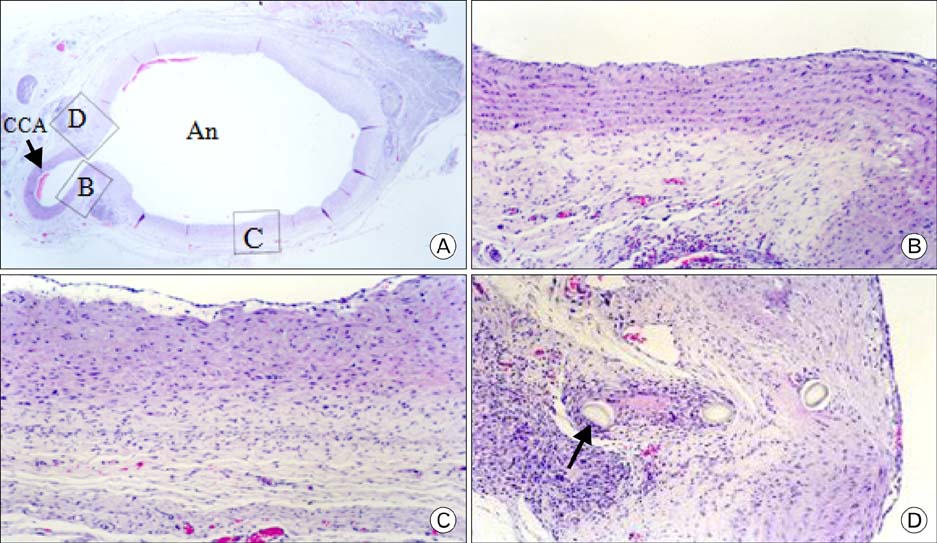
- In order to study the treatment of aneurysms, the technique of making experimental aneurysms in laboratory animals must be established. In our study, to examine the feasibility of making experimental aneurysm and selective angiography on the common carotid artery in rabbits and to determine the size of experimental aneurysm after surgery, saccular aneurysms were fashioned on the right common carotid artery in 17 rabbits using a vein pouch technique. Selective angiography of the common carotid artery was performed immediately after surgery, and at 1 week, 4 weeks, and 8 weeks after surgery. Also, histological changes in the aneurysms were observed. In 16 rabbits with established successful experimental aneurysm, no differences were found in diet intake and behavior before and after surgery. The patency of the carotid artery was confirmed by selective angiography. The average size of the aneurysm immediately after surgery was similar to that of 1 week postoperatively in selective angiography, however it increased with time at 4weeks and 8 weeks. Histologically, infiltration of inflammatory cells and hemorrhage were found at the junction of the carotid artery and the vein pouch at 1 week, which disappeared at 4 weeks and 8 weeks. This study suggests experimental saccular aneurysm using the vein pouch technique might form aneurysms similar to that of the human in its properties such as increment of size, and selective angiography might be suitable for assessment of experimental aneurysm. Therefore, this animal model may be suitable for investigating new treatment methodologies for human aneurysms.
Keyword
MeSH Terms
Figure
Reference
-
1. Allcock J, Canham P. Angiographic study of the growth of intracranial aneurysms. J Neurosurg. 1976. 45:617–621.2. Bookstein JJ, Chlosta EM, Foley D, Walter JF. Transcatheter hemostasis of gastrointestinal bleeding using modified autogenous clot. Radiology. 1974. 113:277–285.3. Byrne JV, Sohn MJ, Molyneux AJ, Chir B. Five-year experience in using coil embolization for ruptured intracranial aneurysms: outcomes and incidence of late rebleeding. J Neurosurg. 1999. 90:656–663.4. Drake CG, Friedman AH, Peerless SJ. Failed aneurysm surgery: reoperation in 115 cases. J Neurosurg. 1984. 61:848–856.5. Forrest MD, O'Reilly GV. Production of experimental aneurysms at a surgically created arterial bifurcation. AJNR Am J Neuroradiol. 1989. 10:400–402.6. German WJ, Black SP. Experimental production of carotid aneurysms. N Engl J Med. 1954. 250:104–106.7. Gertz SD, Kurgan A, Eisenberg D. Aneurysm of the rabbit common carotid artery induced by periarterial application of calcium chloride in vivo. J Clin Invest. 1988. 81:649–656.8. Guglielmi G, Ji C, Massoud TF, Kurata A, Lownie SP, Viñuela F, Robert J. Experimental saccular aneurysms. Neuroradiology. 1994. 36:547–550.9. Hashimoto N, Kim C, Kikuchi H, Kojima M, Kang Y, Hazama F. Experimental induction of cerebral aneurysms in monkeys. J Neurosurg. 1987. 67:903–905.10. Heilman CB, Kwan ES, Wu JK. Aneurysm recurrence following endovascular balloon occlusion. J Neurosurg. 1992. 77:260–264.11. Kirse DJ, Flock S, Teo C, Rahman S, Mrak R. Construction of a vein-pouch aneurysm at a surgically created carotid bifurcation in the rat. Microsurgery. 1996. 17:681–689.12. Krings T, Hans FJ, Möller-Hartmann W, et al. Time-of-flight-, phase contrast and contrast enhanced magnetic resonance angiography for pre-interventional determination of aneurysm size, configuration, and neck morphology in an aneurysm model in rabbits. Neurosci Lett. 2002. 326:46–50.13. Massoud TF, JI C, Guglielmi G, Viñuela F, Robert J. Experimental models of bifurcation and terminal aneurysms: construction techniques in swine. AJNR Am J Neuroradiol. 1994. 15:938–944.14. Osterman FA, Bell WR, Montali RJ, Novak GR, White RI. Natural history of autologous blood clot embolization in swine. Invest Radiol. 1976. 11:267–276.15. Quigley MR, Tuite GF, Cozzens JW. Histology and angiography in a bifurcation aneurysm model. Surg Neurol. 1988. 30:445–451.16. Stehbens WE. Chronic changes in experimental saccular and fusiform aneurysms in rabbits. Arch Pathol Lab Med. 1981a. 105:603–607.17. Stehbens WE. Chronic vascular changes in the walls of experimental berry aneurysms of the aortic bifurcation in rabbits. Stroke. 1981b. 12:643–647.18. Szikora I, Wakhloo AK, Guterman LR, et al. Initial experience with collagen-filled Guglielmi detachable coils for endovascular treatment of experimental aneurysms. AJNR Am J Neuroradiol. 1997. 18:667–672.19. White JC, Sayre GP, Whisnant JP. Experimental destruction of the media for the production of intracranial arterial aneurysms. J Neurosurg. 1961. 18:741–745.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Strategy & Pitfalls of Internal Carotid Artery Aneurysm Surgery
- Renal Artery Aneurysm: Report of Two Cases
- Development of 'De novo' Aneurysm after Therapeutic Carotid Occlusion
- Magnetization Transfer Contrast Angiography for Organized Thrombosed Intracranial Aneurysm in TOF MR Angiography: a Case Report
- Imaging Diagnosis of Pulsatile Bulging on the Neck