Hanyang Med Rev.
2014 Nov;34(4):211-216. 10.7599/hmr.2014.34.4.211.
Current Issues in the Treatment of Chronic Antibody-Mediated Rejection in Kidney Transplantation
- Affiliations
-
- 1Transplant Research Center, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 2Division of Nephrology, Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea. yangch@catholic.ac.kr
- KMID: 2168354
- DOI: http://doi.org/10.7599/hmr.2014.34.4.211
Abstract
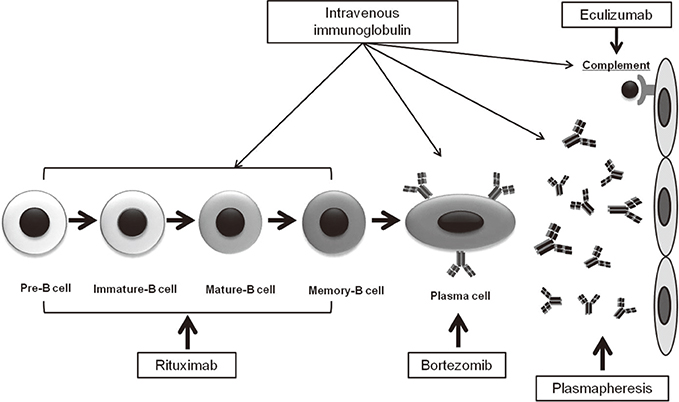
- Circulating alloantibodies are found in a substantial number of renal allograft recipients, and can induce chronic allograft injury, which is represented microscopically as transplant glomerulopathy and diffuse C4d deposition in peritubular capillaries (PTCs). Development of these injuries is significantly correlated with late allograft loss, and in this regard, it was included as a new disease entity named chronic antibody-mediated rejection (cAMR) in the updated Banff 05 classification. Usually, the prognosis of cAMR is poor and conventional immunosuppressants mainly targeting T cell-mediated immunity cannot prevent or reverse it. Therefore, some researchers have suggested that therapies directed at the humoral response may be required for the treatment of cAMR. Recently, some reports have suggested that the combined use of rituximab and intravenous immunoglobulin (IVIg) therapy may be useful for the treatment of cAMR. Our previous study also showed that rituximab and IVIg combination therapy effectively delayed the progression of cAMR. We administered rituximab and IVIg combination therapy to 18 biopsy-proven cAMR patients and found that it significantly slowed the decline of the estimated glomerular filtration rate. However, this effect was limited in patients with heavy proteinuria, and dissipated in all patients by 1 year post-treatment. Recently, new drugs targeting the humoral immune system, such as bortezomib and eculizumab, have been tested for the treatment of cAMR. However, the studies still lack definitive data in terms of successful treatment of cAMR. We speculate that those therapies will compensate for the limitation of previous anti-humoral therapies for cAMR.
Keyword
MeSH Terms
-
Allografts
Capillaries
Classification
Glomerular Filtration Rate
Humans
Immune System
Immunity, Cellular
Immunoglobulins
Immunoglobulins, Intravenous
Immunosuppressive Agents
Isoantibodies
Kidney Transplantation*
Pathology
Prognosis
Proteinuria
Bortezomib
Rituximab
Immunoglobulins
Immunoglobulins, Intravenous
Immunosuppressive Agents
Isoantibodies
Figure
Cited by 2 articles
-
Bortezomib Treatment for Refractory Antibody-Mediated Rejection Superimposed with BK Virus-Associated Nephropathy during the Progression of Recurrent C3 Glomerulonephritis
Wonseok Do, Jong-Hak Lee, Kyung Joo Kim, Man-Hoon Han, Hee-Yeon Jung, Ji-Young Choi, Sun-Hee Park, Yong-Lim Kim, Chan-Duck Kim, Jang-Hee Cho, Youngae Yang, Minjung Kim, Inryang Hwang, Kyu Yeun Kim, Taehoon Yim, Yong-Jin Kim
J Korean Soc Transplant. 2018;32(3):57-62. doi: 10.4285/jkstn.2018.32.3.57.Cutting Edge Technologies in Organ Transplantation
Dongho Choi
Hanyang Med Rev. 2014;34(4):143-144. doi: 10.7599/hmr.2014.34.4.143.
Reference
-
1. Sellares J, de Freitas DG, Mengel M, Reeve J, Einecke G, Sis B, et al. Understanding the causes of kidney transplant failure: the dominant role of antibody-mediated rejection and nonadherence. Am J Transplant. 2012; 12:388–399.
Article2. Mauiyyedi S, Pelle PD, Saidman S, Collins AB, Pascual M, Tolkoff-Rubin NE, et al. Chronic humoral rejection: identification of antibody-mediated chronic renal allograft rejection by C4d deposits in peritubular capillaries. J Am Soc Nephrol. 2001; 12:574–582.
Article3. Regele H, Bohmig GA, Habicht A, Gollowitzer D, Schillinger M, Rockenschaub S, et al. Capillary deposition of complement split product C4d in renal allografts is associated with basement membrane injury in peritubular and glomerular capillaries: a contribution of humoral immunity to chronic allograft rejection. J Am Soc Nephrol. 2002; 13:2371–2380.
Article4. Nankivell BJ, Borrows RJ, Fung CL, O'Connell PJ, Allen RD, Chapman JR. The natural history of chronic allograft nephropathy. N Engl J Med. 2003; 349:2326–2333.
Article5. Vongwiwatana A, Gourishankar S, Campbell PM, Solez K, Halloran PF. Peritubular capillary changes and C4d deposits are associated with transplant glomerulopathy but not IgA nephropathy. Am J Transplant. 2004; 4:124–129.
Article6. Ishii Y, Sawada T, Kubota K, Fuchinoue S, Teraoka S, Shimizu A. Injury and progressive loss of peritubular capillaries in the development of chronic allograft nephropathy. Kidney Int. 2005; 67:321–332.
Article7. Solez K, Colvin RB, Racusen LC, Sis B, Halloran PF, Birk PE, et al. Banff '05 Meeting Report: differential diagnosis of chronic allograft injury and elimination of chronic allograft nephropathy ('CAN'). Am J Transplant. 2007; 7:518–526.
Article8. Aita K, Yamaguchi Y, Shimizu T, Horita S, Furusawa M, Tanabe K, et al. Histological analysis of late renal allografts of antidonor antibody positive patients with C4d deposits in peritubular capillaries. Clin Transplant. 2004; 18:Suppl 11. 7–12.
Article9. Cosio FG, Gloor JM, Sethi S, Stegall MD. Transplant glomerulopathy. Am J Transplant. 2008; 8:492–496.
Article10. Pascual J, Perez-Saez MJ, Mir M, Crespo M. Chronic renal allograft injury: early detection, accurate diagnosis and management. Transplant Rev (Orlando). 2012; 26:280–290.
Article11. Einecke G, Sis B, Reeve J, Mengel M, Campbell PM, Hidalgo LG, et al. Antibody-mediated microcirculation injury is the major cause of late kidney transplant failure. Am J Transplant. 2009; 9:2520–2531.
Article12. Stegall MD, Chedid MF, Cornell LD. The role of complement in antibody-mediated rejection in kidney transplantation. Nat Rev Nephrol. 2012; 8:670–678.
Article13. Lee PC, Terasaki PI, Takemoto SK, Lee PH, Hung CJ, Chen YL, et al. All chronic rejection failures of kidney transplants were preceded by the development of HLA antibodies. Transplantation. 2002; 74:1192–1194.
Article14. Everly MJ, Rebellato LM, Haisch CE, Ozawa M, Parker K, Briley KP, et al. Incidence and impact of de novo donor-specific alloantibody in primary renal allografts. Transplantation. 2013; 95:410–417.
Article15. Wiebe C, Gibson IW, Blydt-Hansen TD, Karpinski M, Ho J, Storsley LJ, et al. Evolution and clinical pathologic correlations of de novo donor-specific HLA antibody post kidney transplant. Am J Transplant. 2012; 12:1157–1167.
Article16. Husain S, Sis B. Advances in the understanding of transplant glomerulopathy. Am J Kidney Dis. 2013; 62:352–363.
Article17. Haas M, Sis B, Racusen LC, Solez K, Glotz D, Colvin RB, et al. Banff 2013 meeting report: inclusion of c4d-negative antibody-mediated rejection and antibody-associated arterial lesions. Am J Transplant. 2014; 14:272–283.
Article18. Solez K, Colvin RB, Racusen LC, Haas M, Sis B, Mengel M, et al. Banff 07 classification of renal allograft pathology: updates and future directions. Am J Transplant. 2008; 8:753–760.
Article19. Gloor JM, Cosio FG, Rea DJ, Wadei HM, Winters JL, Moore SB, et al. Histologic findings one year after positive crossmatch or ABO blood group incompatible living donor kidney transplantation. Am J Transplant. 2006; 6:1841–1847.
Article20. Ishida H, Furusawa M, Shimizu T, Nozaki T, Tanabe K. Influence of preoperative anti-HLA antibodies on short- and long-term graft survival in recipients with or without rituximab treatment. Transpl Int. 2014; 27:371–382.
Article21. Hirai T, Kohei N, Omoto K, Ishida H, Tanabe K. Significance of low-level DSA detected by solid-phase assay in association with acute and chronic antibody-mediated rejection. Transpl Int. 2012; 25:925–934.
Article22. Pascual J, Perez-Saez MJ, Mir M, Crespo M. Chronic renal allograft injury: early detection, accurate diagnosis and management. Transplant Rev (Orlando). 2012; 26:280–290.
Article23. Colvin RB. Antibody-mediated renal allograft rejection: diagnosis and pathogenesis. J Am Soc Nephrol. 2007; 18:1046–1056.
Article24. Hong YA, Kim HG, Choi SR, Sun IO, Park HS, Chung BH, et al. Effectiveness of rituximab and intravenous immunoglobulin therapy in renal transplant recipients with chronic active antibody-mediated rejection. Transplant Proc. 2012; 44:182–184.
Article25. Terasaki PI, Ozawa M. Predictive value of HLA antibodies and serum creatinine in chronic rejection: results of a 2-year prospective trial. Transplantation. 2005; 80:1194–1197.
Article26. Vo AA, Lukovsky M, Toyoda M, Wang J, Reinsmoen NL, Lai CH, et al. Rituximab and intravenous immune globulin for desensitization during renal transplantation. N Engl J Med. 2008; 359:242–251.
Article27. Salama AD, Pusey CD. Drug insight: rituximab in renal disease and transplantation. Nat Clin Pract Nephrol. 2006; 2:221–230.
Article28. Billing H, Rieger S, Ovens J, Susal C, Melk A, Waldherr R, et al. Successful treatment of chronic antibody-mediated rejection with IVIG and rituximab in pediatric renal transplant recipients. Transplantation. 2008; 86:1214–1221.
Article29. Kohei N, Hirai T, Omoto K, Ishida H, Tanabe K. Chronic antibody-mediated rejection is reduced by targeting B-cell immunity during an introductory period. Am J Transplant. 2012; 12:469–476.
Article30. Smith RN, Malik F, Goes N, Farris AB, Zorn E, Saidman S, et al. Partial therapeutic response to Rituximab for the treatment of chronic alloantibody mediated rejection of kidney allografts. Transpl Immunol. 2012; 27:107–113.
Article31. Kazatchkine MD, Kaveri SV. Immunomodulation of autoimmune and inflammatory diseases with intravenous immune globulin. N Engl J Med. 2001; 345:747–755.
Article32. Jordan SC, Vo AA, Peng A, Toyoda M, Tyan D. Intravenous gammaglobulin (IVIG): a novel approach to improve transplant rates and outcomes in highly HLA-sensitized patients. Am J Transplant. 2006; 6:459–466.
Article33. Jordan SC, Quartel AW, Czer LS, Admon D, Chen G, Fishbein MC, et al. Posttransplant therapy using high-dose human immunoglobulin (intravenous gammaglobulin) to control acute humoral rejection in renal and cardiac allograft recipients and potential mechanism of action. Transplantation. 1998; 66:800–805.
Article34. Jordan SC, Vo A, Bunnapradist S, Toyoda M, Peng A, Puliyanda D, et al. Intravenous immune globulin treatment inhibits crossmatch positivity and allows for successful transplantation of incompatible organs in living-donor and cadaver recipients. Transplantation. 2003; 76:631–636.
Article35. Billing H, Rieger S, Susal C, Waldherr R, Opelz G, Wuhl E, et al. IVIG and rituximab for treatment of chronic antibody-mediated rejection: a prospective study in paediatric renal transplantation with a 2-year follow-up. Transpl Int. 2012; 25:1165–1173.
Article36. Fehr T, Rusi B, Fischer A, Hopfer H, Wuthrich RP, Gaspert A. Rituximab and intravenous immunoglobulin treatment of chronic antibody-mediated kidney allograft rejection. Transplantation. 2009; 87:1837–1841.
Article37. An GH, Yun J, Hong YA, Khvan M, Chung BH, Choi BS, et al. The Effect of Combination Therapy with Rituximab and Intravenous Immunoglobulin on the Progression of Chronic Antibody Mediated Rejection in Renal Transplant Recipients. J Immunol Res. 2014; 2014:828732.
Article38. Clatworthy MR. Targeting B cells and antibody in transplantation. Am J Transplant. 2011; 11:1359–1367.
Article39. Everly MJ. A summary of bortezomib use in transplantation across 29 centers. Clin Transpl. 2009; 323–337.40. Schwaiger E, Regele H, Wahrmann M, Werzowa J, Haidbauer B, Schmidt A, et al. Bortezomib for the treatment of chronic antibody-mediated kidney allograft rejection: a case report. Clin Transpl. 2010; 391–396.41. Perry DK, Burns JM, Pollinger HS, Amiot BP, Gloor JM, Gores GJ, et al. Proteasome inhibition causes apoptosis of normal human plasma cells preventing alloantibody production. Am J Transplant. 2009; 9:201–209.
Article42. Everly MJ. An update on antibody reduction and rejection reversal following bortezomib use: a report of 52 cases across 10 centers. Clin Transpl. 2010; 353–362.43. Yang KS, Jeon H, Park Y, Jo IH, Kim JI, Moon IS, et al. Use of bortezomib as anti-humoral therapy in kidney transplantation. J Korean Med Sci. 2014; 29:648–651.
Article44. Vogelbacher R, Meister S, Guckel E, Starke C, Wittmann S, Stief A, et al. Bortezomib and sirolimus inhibit the chronic active antibody-mediated rejection in experimental renal transplantation in the rat. Nephrol Dial Transplant. 2010; 25:3764–3773.
Article45. Kim MG, Kim YJ, Kwon HY, Park HC, Koo TY, Jeong JC, et al. Outcomes of combination therapy for chronic antibody-mediated rejection in renal transplantation. Nephrology (Carlton). 2013; 18:820–826.
Article46. Larrea CF, Cofan F, Oppenheimer F, Campistol JM, Escolar G, Lozano M. Efficacy of eculizumab in the treatment of recurrent atypical hemolytic-uremic syndrome after renal transplantation. Transplantation. 2010; 89:903–904.
Article47. Eculizumab Therapy for Chronic Complement-Mediated Injury in Kidney Transplantation. cited 2014 Jul 10. Available from: http://clinicaltrialsgov/show/NCT01327573.48. Picinich SC, Mishra PJ, Glod J, Banerjee D. The therapeutic potential of mesenchymal stem cells. Cell- & tissue-based therapy. Expert Opin Biol Ther. 2007; 7:965–973.49. Hematti P. Role of mesenchymal stromal cells in solid organ transplantation. Transplant Rev (Orlando). 2008; 22:262–273.
Article50. Togel F, Weiss K, Yang Y, Hu Z, Zhang P, Westenfelder C. Vasculotropic, paracrine actions of infused mesenchymal stem cells are important to the recovery from acute kidney injury. Am J Physiol Renal Physiol. 2007; 292:F1626–F1635.51. Bi B, Schmitt R, Israilova M, Nishio H, Cantley LG. Stromal cells protect against acute tubular injury via an endocrine effect. J Am Soc Nephrol. 2007; 18:2486–2496.
Article52. Tan J, Wu W, Xu X, Liao L, Zheng F, Messinger S, et al. Induction therapy with autologous mesenchymal stem cells in living-related kidney transplants: a randomized controlled trial. JAMA. 2012; 307:1169–1177.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pathologic Updates on Antibody Mediated Rejection in Renal Transplantation
- Effect of High Dose Intravenous Immunoglobulins on the Treatment of Antibody Mediated Humoral Rejection and BK Virus Infection in Renal Transplant Recipients
- New treatment for antibody-mediated rejection: interleukin-6 inhibitors
- Clinical course of graft failure after kidney transplantation investigated focusing on immune rejection
- Current status of pediatric kidney transplantation