Cancer Res Treat.
2010 Jun;42(2):95-100.
Role of Loss of O(6)-Methylguanine DNA Methyltransferase (MGMT) Expression in Non-Small Cell Lung Carcinomas (NSCLCs): with Reference to the Relationship with p53 Overexpression
- Affiliations
-
- 1Department of Pathology, Dankook University College of Medicine, Cheonan, Korea. myongnh@dankook.ac.kr
Abstract
- PURPOSE
Functional inactivation of the O(6)-methylguanine-DNA methyltransferase (MGMT) gene has been demonstrated as loss of MGMT protein and suggested that it plays an important role in primary human neoplasia, including lung cancer. It has also been reported to be associated with the G : C-->A : T transition mutation in the p53 gene of lung cancer. The aims of this study were to investigate the role of MGMT expression loss and its prognostic significance in non-small cell lung carcinomas (NSCLCs), and its correlation with p53 overexpression as well as influence on patient survival.
MATERIALS AND METHODS
112 surgically resected NSCLC specimens were reviewed by medical records for their clinicopathologic variables. Their tissue microarray blocks were immunostained with anti-human MGMT and p53 primary antibodies. Correlation between MGMT loss and the clinicopathologic prognostic factors, including p53 overexpression and the single or combined actions of MGMT loss and p53 overexpression on patient survival were statistically analyzed by SPSS15.0.
RESULTS
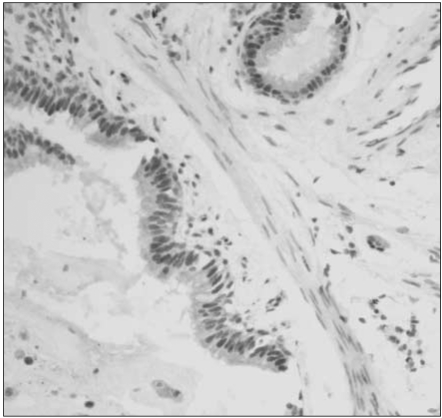
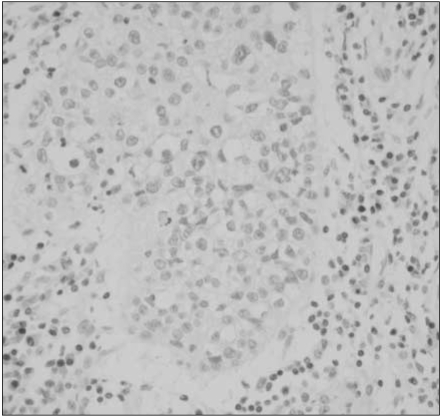
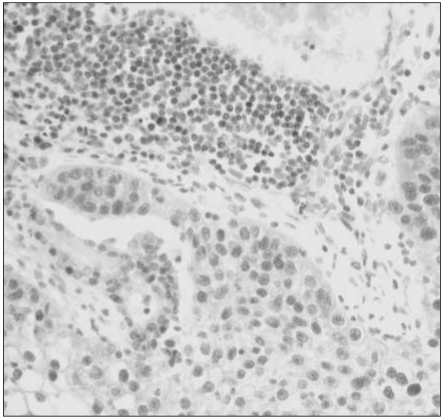
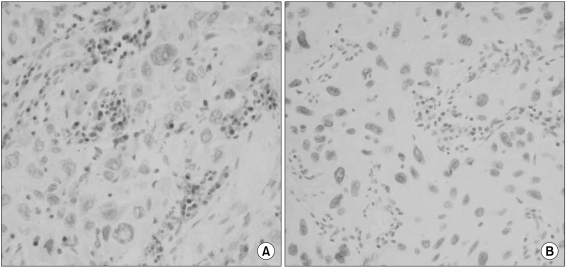
Reduced or absent MGMT expression was found in 48 of 112 NSCLCs (43%), and significantly associated with nodal metastasis and squamous or undifferentiated cell types. Loss of MGMT expression was correlated with p53 overexpression in adenocarcinomas, but not in overall NSCLCs. Its solitary or combined actions with p53 overexpression did not have influence on patient survival.
CONCLUSION
Loss of MGMT expression is a relatively common event in NSCLCs and significantly associated with nodal metastasis and p53 overexpression, suggesting that it may play a major role in pulmonary carcinogenesis, and also in disease progression of NSCLCs.
Keyword
MeSH Terms
Figure
Reference
-
1. Pegg AE. Mammalian O6-alkylguanine-DNA alkyltransferase regulation and importance in response to alkylating carcinogenic and therapeutic agents. Cancer Res. 1990; 50:6119–6129. PMID: 2205376.2. Costello JF, Futscher BW, Tano K, Graunke DM, Pieper RO. Graded methylation in the promoter and in the body of the O6-methylguanine-DNA methyltransferase (MGMT) gene correlates with MGMT expression in human glioma cells. J Biol Chem. 1994; 269:17228–17237. PMID: 8006031.3. Qian XC, Brent TP. Methylation hot spots in the 5'-flanking region denote silencing of the O6-methylguanine-DNA methyltransferase gene. Cancer Res. 1997; 57:3672–3677. PMID: 9288770.4. Watts GS, Pieper RO, Costello JF, Peng YM, Dalton WS, Futscher BW. Methylation of discrete regions of the O6-methylguanine DNA methyltransferase (MGMT) CpG island is associated with heterochromatinization of the MGMT transcription start site and silencing of the gene. Mol Cell Biol. 1997; 17:5612–5619. PMID: 9271436.5. Esteller M, Hamilton SR, Burger PC, Baylin SB, Herman JG. Inactivation of the DNA repair gene O6-methylguanine-DNA methyltransferase by promoter hypermethylation is a common event in primary human neoplasia. Cancer Res. 1999; 59:793–797. PMID: 10029064.6. Feng Q, Hawes SE, Stern JE, Wiens L, Lu H, Dong ZM, et al. DNA methylation in tumor and matched normal tissues from non-small lung cancer patients. Cancer Epidemiol Biomarkers Prev. 2008; 17:645–654. PMID: 18349282.7. de Jong WK, Verpooten GF, Kramer H, Louwagie J, Groen HJ. Promoter methylation primarily occurs in tumor cells of patients with non-small cell lung cancer. Anticancer Res. 2009; 29:363–369. PMID: 19331174.8. Wolf P, Hu YC, Doffek K, Sidransky D, Ahrendt SA. O6-methylguanine-DNA methyltransferase promoter hypermethylation shifts the p53 mutational spectrum in non-small cell lung cancer. Cancer Res. 2001; 61:8113–8117. PMID: 11719438.9. Wu JY, Wang J, Lai JC, Cheng YW, Yeh KT, Wu TC, et al. Association of O6-methylguanine-DNA methyltransferase (MGMT) promoter methylation with p53 mutation occurrence in non-small cell lung cancer with different histology, gender, and smoking status. Ann Surg Oncol. 2008; 15:3272–3277. PMID: 18712569.10. Brabender J, Usadel H, Metzger R, Schneider PM, Park J, Salonga D, et al. Quantitative O6-methylguanine DNA methyltransferase methylation analysis in curatively resected non-small cell lung cancer: Association with clinical outcome. Clin Cancer Res. 2003; 9:223–227. PMID: 12538473.11. Cooper WA, Kohonen-Corish MR, Chan C, Kwun SY, McCaughan B, Kennedy C, et al. Prognostic significance of DNA repair proteins MLH1, MSH2 and MGMT expression in non-small-cell kung cancer and precursor lesions. Histopathology. 2008; 52:613–622. PMID: 18370958.12. Sawhney M, Rohatgi N, Kaur J, Gupta SD, Deo SV, Shukla NK, et al. MGMT expression in oral precancerous and cancerous lesions: Correlation with progression, nodal metastasis and poor prognosis. Oral Oncol. 2007; 43:515–522. PMID: 16996781.
Article13. Pulling LC, Divine KK, Klinge DM, Gilliland FD, Kang T, Schwartz AG, et al. Promoter hypermethylation of the O6-methylguanine-DNA methyltransferase gene: more common in lung adenocarcinomas from never-smokers than smokers and associated with tumor progression. Cancer Res. 2003; 63:4842–4848. PMID: 12941804.14. Laird PW, Jaenisch R. The role of DNA methylation in cancer genetics and epigenetics. Annu Rev Genet. 1996; 30:441–464. PMID: 8982461.
Article15. Baylin SB, Herman JG, Graff JR, Vertino PM, Issa JP. Alterations in DNA methylation: a fundamental aspect of neoplasia. Adv Cancer Res. 1998; 72:141–196. PMID: 9338076.16. Jones PA, Laird PW. Cancer epigenetics comes of age. Nat Genet. 1999; 21:163–167. PMID: 9988266.17. Kitajima Y, Miyazaki K, Matsukura S, Tanaka M, Sekiguchi M. Loss of expression of DNA repair enzymes MGMT, hMLH1 and hMSH2 during tumor progression in gastric cancer. Gastric Cancer. 2003; 2:86–95. PMID: 12861399.
Article18. Kohya N, Miyazaki K, Matsukura S, Yakushiji H, Kitajima Y, Kitahara K, et al. Deficient expression of O6-methylguanine-DNA methyltransferase combined with mismatch-repair proteins hMLH1 and hMSH2 is related to poor prognosis in human biliary tract carcinoma. Ann Surg Oncol. 2002; 9:371–379. PMID: 11986189.19. Lai JC, Cheng YW, Goan YG, Chang JT, Wu TC, Chen CY, et al. Promoter methylation of O6-methylguanine-DNA-methyltransferase in lung cancer is regulated by p53. DNA Repair. 2008; 7:1352–1363. PMID: 18555750.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Immunohistochemical Study on the Expression of p53 and Bcl-2 Proteins in Non-Small Cell Lung Carcinomas
- Tissue Microarray Analysis of Fas and FasL Expressions in Human Non-small Cell Lung Carcinomas; with Reference to the p53 and bcl-2 Overexpressions
- The Expression of Telomerase Reverse Transcriptase Protein is an Independent Prognostic Marker in Early Stage Non-Small Cell Lung Carcinomas
- The Study of p53 Expression and DNA Ploidy in Colorectal Carcinoma
- Expression of Cyclin-dependent Kinase Inhibitor p21(WAF1/CIP1) in Non-small Cell Lung Carcinomas: Relationship with p53 Status and Proliferative Activity