Yonsei Med J.
2015 Sep;56(5):1251-1257. 10.3349/ymj.2015.56.5.1251.
In silico Screening of Chemical Libraries to Develop Inhibitors That Hamper the Interaction of PCSK9 with the LDL Receptor
- Affiliations
-
- 1Department of Biochemistry and Molecular Biology, Institute of Genetic Science, Integrated Genomic Research Center for Metabolic Regulation, Yonsei University College of Medicine, Seoul, Korea. swpark64@yuhs.ac
- 2Brain Korea 21 PLUS Project for Medical Science, Yonsei University College of Medicine, Seoul, Korea.
- 3Department of Biochemistry, Catholic Kwandong University College of Medicine, Gangneung, Korea.
- KMID: 2163616
- DOI: http://doi.org/10.3349/ymj.2015.56.5.1251
Abstract
- PURPOSE
Proprotein convertase subtilisin/kexin type 9 (PCSK9) binds to the low density lipoprotein receptor (LDLR) and promotes degradation of the LDLR. Inhibition of PCSK9 either by reducing its expression or by blocking its activity results in the upregulation of the LDLR and subsequently lowers the plasma concentration of LDL-cholesterol. As a modality to inhibit PCSK9 action, we searched the chemical library for small molecules that block the binding of PCSK9 to the LDLR.
MATERIALS AND METHODS
We selected 100 chemicals that bind to PCSK9 where the EGF-AB fragment of the LDLR binds via in silico screening of the ChemBridge chemical library, using the computational GOLD algorithm analysis. Effects of chemicals were evaluated using the PCSK9-LDLR binding assay, immunoblot analysis, and the LDL-cholesterol uptake assay in vitro, as well as the fast performance liquid chromatography assay for plasma lipoproteins in vivo.
RESULTS
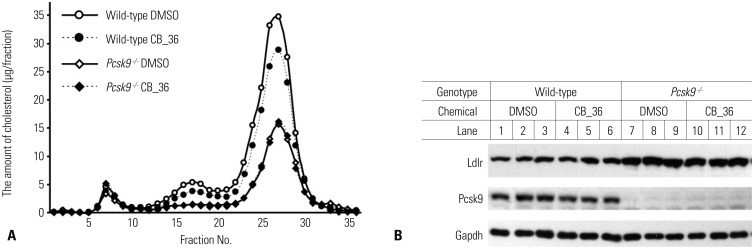
A set of chemicals were found that decreased the binding of PCSK9 to the EGF-AB fragment of the LDLR in a dose-dependent manner. They also increased the amount of the LDLR significantly and subsequently increased the uptake of fluorescence-labeled LDL in HepG2 cells. Additionally, one particular molecule lowered the plasma concentration of total cholesterol and LDL-cholesterol significantly in wild-type mice, while such an effect was not observed in Pcsk9 knockout mice.
CONCLUSION
Our findings strongly suggest that in silico screening of small molecules that inhibit the protein-protein interaction between PCSK9 and the LDLR is a potential modality for developing hypercholesterolemia therapeutics.
Keyword
MeSH Terms
-
Animals
Cholesterol/*blood
Cholesterol, LDL/blood
Hep G2 Cells
Humans
Mice
Mice, Knockout
Proprotein Convertases/*metabolism
Receptors, LDL/*metabolism
Serine Endopeptidases/*metabolism
*Small Molecule Libraries
Cholesterol
Cholesterol, LDL
Proprotein Convertases
Receptors, LDL
Serine Endopeptidases
Small Molecule Libraries
Figure
Reference
-
1. Canto JG, Iskandrian AE. Major risk factors for cardiovascular disease: debunking the "only 50%" myth. JAMA. 2003; 290:947–949. PMID: 12928473.3. Abifadel M, Varret M, Rabès JP, Allard D, Ouguerram K, Devillers M, et al. Mutations in PCSK9 cause autosomal dominant hypercholesterolemia. Nat Genet. 2003; 34:154–156. PMID: 12730697.
Article4. Cohen JC, Boerwinkle E, Mosley TH Jr, Hobbs HH. Sequence variations in PCSK9, low LDL, and protection against coronary heart disease. N Engl J Med. 2006; 354:1264–1272. PMID: 16554528.5. Farnier M. PCSK9: From discovery to therapeutic applications. Arch Cardiovasc Dis. 2014; 107:58–66. PMID: 24373748.
Article6. Stein EA, Mellis S, Yancopoulos GD, Stahl N, Logan D, Smith WB, et al. Effect of a monoclonal antibody to PCSK9 on LDL cholesterol. N Engl J Med. 2012; 366:1108–1118. PMID: 22435370.
Article7. Dias CS, Shaywitz AJ, Wasserman SM, Smith BP, Gao B, Stolman DS, et al. Effects of AMG 145 on low-density lipoprotein cholesterol levels: results from 2 randomized, double-blind, placebocontrolled, ascending-dose phase 1 studies in healthy volunteers and hypercholesterolemic subjects on statins. J Am Coll Cardiol. 2012; 60:1888–1898. PMID: 23083772.8. Koren MJ, Giugliano RP, Raal FJ, Sullivan D, Bolognese M, Langslet G, et al. Efficacy and safety of longer-term administration of evolocumab (AMG 145) in patients with hypercholesterolemia: 52-week results from the Open-Label Study of Long-Term Evaluation Against LDL-C (OSLER) randomized trial. Circulation. 2014; 129:234–243. PMID: 24255061.9. Frank-Kamenetsky M, Grefhorst A, Anderson NN, Racie TS, Bramlage B, Akinc A, et al. Therapeutic RNAi targeting PCSK9 acutely lowers plasma cholesterol in rodents and LDL cholesterol in nonhuman primates. Proc Natl Acad Sci U S A. 2008; 105:11915–11920. PMID: 18695239.
Article10. Rhainds D, Arsenault BJ, Tardif JC. PCSK9 inhibition and LDL cholesterol lowering: the biology of an attractive therapeutic target and critical review of the latest clinical trials. Clin Lipidol. 2012; 7:621–640.
Article11. Mitchell T, Chao G, Sitkoff D, Lo F, Monshizadegan H, Meyers D, et al. Pharmacologic profile of the Adnectin BMS-962476, a small protein biologic alternative to PCSK9 antibodies for low-density lipoprotein lowering. J Pharmacol Exp Ther. 2014; 350:412–424. PMID: 24917546.
Article12. Andricopulo AD, Salum LB, Abraham DJ. Structure-based drug design strategies in medicinal chemistry. Curr Top Med Chem. 2009; 9:771–790. PMID: 19754394.
Article13. Pearlstein RA, Hu QY, Zhou J, Yowe D, Levell J, Dale B, et al. New hypotheses about the structure-function of proprotein convertase subtilisin/kexin type 9: analysis of the epidermal growth factor-like repeat A docking site using WaterMap. Proteins. 2010; 78:2571–2586. PMID: 20589640.
Article14. Bottomley MJ, Cirillo A, Orsatti L, Ruggeri L, Fisher TS, Santoro JC, et al. Structural and biochemical characterization of the wild type PCSK9-EGF(AB) complex and natural familial hypercholesterolemia mutants. J Biol Chem. 2009; 284:1313–1323. PMID: 19001363.
Article15. Kwon HJ, Lagace TA, McNutt MC, Horton JD, Deisenhofer J. Molecular basis for LDL receptor recognition by PCSK9. Proc Natl Acad Sci U S A. 2008; 105:1820–1825. PMID: 18250299.
Article16. Russell DW, Schneider WJ, Yamamoto T, Luskey KL, Brown MS, Goldstein JL. Domain map of the LDL receptor: sequence homology with the epidermal growth factor precursor. Cell. 1984; 37:577–585. PMID: 6327078.
Article17. Jeong HJ, Lee HS, Kim KS, Kim YK, Yoon D, Park SW. Sterol-dependent regulation of proprotein convertase subtilisin/kexin type 9 expression by sterol-regulatory element binding protein-2. J Lipid Res. 2008; 49:399–409. PMID: 17921436.
Article18. Verdonk ML, Cole JC, Hartshorn MJ, Murray CW, Taylor RD. Improved protein-ligand docking using GOLD. Proteins. 2003; 52:609–623. PMID: 12910460.
Article19. Hannah VC, Ou J, Luong A, Goldstein JL, Brown MS. Unsaturated fatty acids down-regulate srebp isoforms 1a and 1c by two mechanisms in HEK-293 cells. J Biol Chem. 2001; 276:4365–4372. PMID: 11085986.
Article20. Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012; 9:671–675. PMID: 22930834.
Article21. Kim KW, McCormick J, Helmering J, Véniant MM, Wang M. An optimized fast-performance liquid chromatography method for analyzing lipoprotein profiles using microliter volumes of serum. Anal Biochem. 2008; 376:268–274. PMID: 18358227.
Article22. Rashid S, Curtis DE, Garuti R, Anderson NN, Bashmakov Y, Ho YK, et al. Decreased plasma cholesterol and hypersensitivity to statins in mice lacking Pcsk9. Proc Natl Acad Sci U S A. 2005; 102:5374–5379. PMID: 15805190.
Article23. Peng H, Huang N, Qi J, Xie P, Xu C, Wang J, et al. Identification of novel inhibitors of BCR-ABL tyrosine kinase via virtual screening. Bioorg Med Chem Lett. 2003; 13:3693–3699. PMID: 14552760.
Article24. Rastelli G, Pacchioni S, Sirawaraporn W, Sirawaraporn R, Parenti MD, Ferrari AM. Docking and database screening reveal new classes of Plasmodium falciparum dihydrofolate reductase inhibitors. J Med Chem. 2003; 46:2834–2845. PMID: 12825927.
Article25. Kamionka M, Rehm T, Beisel HG, Lang K, Engh RA, Holak TA. In silico and NMR identification of inhibitors of the IGF-I and IGFbinding protein-5 interaction. J Med Chem. 2002; 45:5655–5660. PMID: 12477349.
Article26. Kortagere S, Welsh WJ, Morrisey JM, Daly T, Ejigiri I, Sinnis P, et al. Structure-based design of novel small-molecule inhibitors of Plasmodium falciparum. J Chem Inf Model. 2010; 50:840–849. PMID: 20426475.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Role of PCSK9 Inhibitors in Patients with Familial Hypercholesterolemia
- How to Interpret Recent CV Outcome Trials and Future: PCSK9 Inhibitors
- Pharmacological Strategies beyond Statins: Ezetimibe and PCSK9 Inhibitors
- Re-evaluating the PCSK9 guidelines for low-density lipoprotein cholesterol targets: weighing the benefits against the risks
- Evaluation of Advanced Structure-Based Virtual Screening Methods for Computer-Aided Drug Discovery