Immune Netw.
2013 Dec;13(6):264-274. 10.4110/in.2013.13.6.264.
Detection of Foreign Antigen-specific CD4+Foxp3+ Regulatory T Cells by MHC Class II Tetramer and Intracellular CD154 Staining
- Affiliations
-
- 1College of Veterinary Medicine and Bio-Safety Research Institute, Chonbuk National University, Jeonju 561-756, Korea. vetvirus@chonbuk.ac.kr
- KMID: 2150787
- DOI: http://doi.org/10.4110/in.2013.13.6.264
Abstract
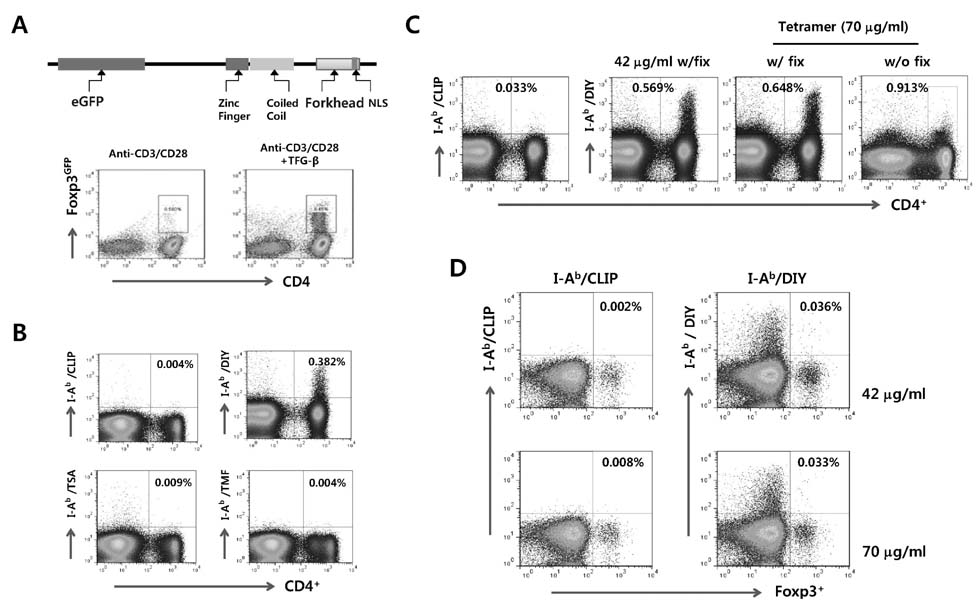
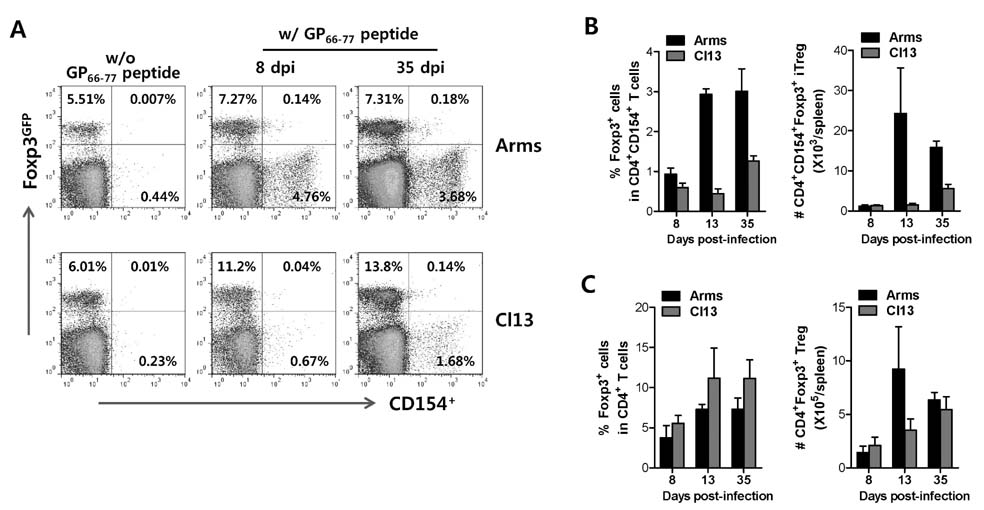
- The unrestricted population of CD4+Foxp3+ regulatory T (Treg) cells, which have been known to control the expression of autoimmune diseases and protective immunity to inflammatory reactions, has led to greater appreciation of functional plasticity. Detecting and/or isolating Ag-specific CD4+Foxp3+ Tregs at the single cell level are required to study their function and plasticity. In this study, we established and compared both MHC class II tetramer and intracellular CD154 staining, in order to detect CD4+Foxp3+ Treg specific for foreign Ag in acute and chronic infections with lymphocytic choriomeningitis virus (LCMV). Our results revealed that MHC class II tetramer staining showed a lower detection rate of LCMV GP66-77-specific CD4+ T cells because most of MHC class II tetramers were unbound and unstable when combined staining was performed with intracellular cytokines. In contrast, intracellular CD154 staining was revealed to be easier and simple for detecting LCMV GP66-77-specific CD4+ T cells, compared to MHC class II tetramer staining. Subsequently, we employed intracellular CD154 staining to detect LCMV GP66-77-specific CD4+Foxp3+ Tregs using Foxp3GFP knock-in mouse, and found that LCMV GP66-77-specific CD4+Foxp3+ Tregs and polyclonal CD4+Foxp3+ Tregs showed differential expansion in mice infected with LCMV Arms or Cl13 at acute (8 and 13 days pi) and chronic phases (35 days pi). Therefore, our results provide insight into the valuable use of intracellular CD154 staining to detect and characterize foreign Ag-specific CD4+Foxp3+ Treg in various models.
Keyword
MeSH Terms
Figure
Reference
-
1. Valencia X, Lipsky PE. CD4+CD25+FoxP3+ regulatory T cells in autoimmune diseases. Nat Clin Pract Rheumatol. 2007; 3:619–626.2. Huan J, Culbertson N, Spencer L, Bartholomew R, Burrows GG, Chou YK, Bourdette D, Ziegler SF, Offner H, Vandenbark AA. Decreased Foxp3 levels in multiple sclerosis patients. J Neurosci Res. 2005; 81:45–52.
Article3. Xu W, Lan Q, Chen M, Chen H, Zhu N, Zhou X, Wang J, Fan H, Yan CS, Kuang JL, Warburton D, Togbe D, Ryffel B, Zheng SG, Shi W. Adoptive transfer of induced-Treg cells effectively attenuates murine airway allergic inflammation. PLoS One. 2012; 7:e40314.
Article4. Adeegbe D, Levy RB, Malek TR. Allogeneic T regulatory cell-mediated transplantation tolerance in adoptive therapy depends on dominant peripheral suppression and central tolerance. Blood. 2010; 115:1932–1940.
Article5. Li P, Gan Y, Sun BL, Zhang F, Lu B, Gao Y, Liang W, Thomson AW, Chen J, Hu X. Adoptive regulatory T-cell therapy protects against cerebral ischemia. Ann Neurol. 2012; in press: doi: 10.1002/ana.23815.
Article6. Cao Y, Xu W, Xiong S. Adoptive Transfer of Regulatory T Cells Protects against Coxsackievirus B3-Induced Cardiac Fibrosis. PLoS One. 2013; 8:e74955.
Article7. Vasconcelos JF, Souza BS, Lins TF, Garcia LM, Kaneto CM, Sampaio GP, de Alcantara AC, Meira CS, Macambira SG, Ribeiro-Dos-Santos R, Soares MB. Administration of granulocyte colony-stimulating factor induces immunomodulation, recruitment of T regulatory cells, reduction of myocarditis and decrease of parasite load in a mouse model of chronic Chagas disease cardiomyopathy. FASEB J. 2013; in press: doi: 10.1096/fj.13-229351.
Article8. Kleinschnitz C, Kraft P, Dreykluft A, Hagedorn I, Gobel K, Schuhmann MK, Langhauser F, Helluy X, Schwarz T, Bittner S, Mayer CT, Brede M, Varallyay C, Pham M, Bendszus M, Jakob P, Magnus T, Meuth SG, Iwakura Y, Zernecke A, Sparwasser T, Nieswandt B, Stoll G, Wiendl H. Regulatory T cells are strong promoters of acute ischemic stroke in mice by inducing dysfunction of the cerebral microvasculature. Blood. 2013; 121:679–691.
Article9. Yadav M, Stephan S, Bluestone JA. Peripherally induced tregs - role in immune homeostasis and autoimmunity. Front Immunol. 2013; 4:232.
Article10. Sakaguchi S, Ono M, Setoguchi R, Yagi H, Hori S, Fehervari Z, Shimizu J, Takahashi T, Nomura T. Foxp3+ CD25+ CD4+ natural regulatory T cells in dominant self-tolerance and autoimmune disease. Immunol Rev. 2006; 212:8–27.
Article11. Komatsu N, Mariotti-Ferrandiz ME, Wang Y, Malissen B, Waldmann H, Hori S. Heterogeneity of natural Foxp3+ T cells: a committed regulatory T-cell lineage and an uncommitted minor population retaining plasticity. Proc Natl Acad Sci USA. 2009; 106:1903–1908.
Article12. Pacholczyk R, Kern J. The T-cell receptor repertoire of regulatory T cells. Immunology. 2008; 125:450–458.
Article13. Song KD, Hwang S, Yun CH. T cell receptor signaling that regulates the development of intrathymic natural regulatory T cells. Immune Netw. 2011; 11:336–341.
Article14. Curotto de Lafaille MA, Lafaille JJ. Natural and adaptive Foxp3+ regulatory T cells: more of the same or a division of labor? Immunity. 2009; 30:626–635.
Article15. Altman JD, Moss PA, Goulder PJ, Barouch DH, McHeyzer-Williams MG, Bell JI, McMichael AJ, Davis MM. Phenotypic analysis of antigen-specific T lymphocytes. Science. 1996; 274:94–96.
Article16. Krueger LA, Nugent CT, Hampl J. Identification of human antigen-specific T cells using MHC class I and class II tetramers. Curr Protoc Cytom. 2004; Chapter 6:Unit 6.18.17. Cecconi V, Moro M, Del Mare S, Dellabona P, Casorati G. Use of MHC class II tetramers to investigate CD4+ T cell responses: problems and solutions. Cytometry A. 2008; 73:1010–1018.18. Guillaume P, Dojcinovic D, Luescher IF. Soluble MHC-peptide complexes: tools for the monitoring of T cell responses in clinical trials and basic research. Cancer Immun. 2009; 9:7–12.19. Chattopadhyay PK, Yu J, Roederer M. A live-cell assay to detect antigen-specific CD4+ T cells with diverse cytokine profiles. Nat Med. 2005; 11:1113–1117.
Article20. Frentsch M, Arbach O, Kirchhoff D, Moewes B, Worm M, Rothe M, Scheffold A, Thiel A. Direct access to CD4+ T cells specific for defined antigens according to CD154 expression. Nat Med. 2005; 11:1118–1124.
Article21. Chattopadhyay PK, Yu J, Roederer M. Live-cell assay to detect antigen-specific CD4+ T-cell responses by CD154 expression. Nat Protoc. 2006; 1:1–6.
Article22. Holst PJ, Christensen JP, Thomsen AR. Vaccination against lymphocytic choriomeningitis virus infection in MHC class II-deficient mice. J Immunol. 2011; 186:3997–4007.
Article23. McDermott DS, Varga SM. Quantifying antigen-specific CD4 T cells during a viral infection: CD4 T cell responses are larger than we think. J Immunol. 2011; 187:5568–5576.
Article24. Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH, Freeman GJ, Ahmed R. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature. 2006; 439:682–687.
Article25. Shin H, Wherry EJ. CD8 T cell dysfunction during chronic viral infection. Curr Opin Immunol. 2007; 19:408–415.
Article26. Zinselmeyer BH, Heydari S, Sacristan C, Nayak D, Cammer M, Herz J, Cheng X, Davis SJ, Dustin ML, McGavern DB. PD-1 promotes immune exhaustion by inducing antiviral T cell motility paralysis. J Exp Med. 2013; 210:757–774.
Article27. Roy M, Waldschmidt T, Aruffo A, Ledbetter JA, Noelle RJ. The regulation of the expression of gp39, the CD40 ligand, on normal and cloned CD4+ T cells. J Immunol. 1993; 151:2497–2510.28. Yellin MJ, Sippel K, Inghirami G, Covey LR, Lee JJ, Sinning J, Clark EA, Chess L, Lederman S. CD40 molecules induce down-modulation and endocytosis of T cell surface T cell-B cell activating molecule/CD40-L. Potential role in regulating helper effector function. J Immunol. 1994; 152:598–608.29. Graf D, Muller S, Korthauer U, van Kooten C, Weise C, Kroczek RA. A soluble form of TRAP (CD40 ligand) is rapidly released after T cell activation. Eur J Immunol. 1995; 25:1749–1754.
Article30. Hartigan-O'Connor DJ, Poon C, Sinclair E, McCune JM. Human CD4+ regulatory T cells express lower levels of the IL-7 receptor alpha chain (CD127), allowing consistent identification and sorting of live cells. J Immunol Methods. 2007; 319:41–52.31. Miyara M, Yoshioka Y, Kitoh A, Shima T, Wing K, Niwa A, Parizot C, Taflin C, Heike T, Valeyre D, Mathian A, Nakahata T, Yamaguchi T, Nomura T, Ono M, Amoura Z, Gorochov G, Sakaguchi S. Functional delineation and differentiation dynamics of human CD4+ T cells expressing the FoxP3 transcription factor. Immunity. 2009; 30:899–911.
Article32. Hoffmann P, Boeld TJ, Eder R, Huehn J, Floess S, Wieczorek G, Olek S, Dietmaier W, Andreesen R, Edinger M. Loss of FOXP3 expression in natural human CD4+CD25+ regulatory T cells upon repetitive in vitro stimulation. Eur J Immunol. 2009; 39:1088–1097.
Article33. Vukmanovic-Stejic M, Agius E, Booth N, Dunne PJ, Lacy KE, Reed JR, Sobande TO, Kissane S, Salmon M, Rustin MH, Akbar AN. The kinetics of CD4+Foxp3+ T cell accumulation during a human cutaneous antigen-specific memory response in vivo. J Clin Invest. 2008; 118:3639–3650.
Article34. Litjens NH, Boer K, Betjes MG. Identification of circulating human antigen-reactive CD4+ FOXP3+ natural regulatory T cells. J Immunol. 2012; 188:1083–1090.
Article35. Pacholczyk R, Kern J, Singh N, Iwashima M, Kraj P, Ignatowicz L. Nonself-antigens are the cognate specificities of Foxp3+ regulatory T cells. Immunity. 2007; 27:493–504.
Article36. Kasow KA, Chen X, Knowles J, Wichlan D, Handgretinger R, Riberdy JM. Human CD4+CD25+ regulatory T cells share equally complex and comparable repertoires with CD4+CD25- counterparts. J Immunol. 2004; 172:6123–6128.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Transcription of class II MHC gene by interferon-gamma in FRTL-5 cells
- Peripheral Generation of CD4+ CD25+ Foxp3+ Regulatory T Cells
- Immunomodulatory Effects of Hypocrellin A on MHC-restricted Antigen Processing
- A Study on the Number of Circulating CD4+CD25+Foxp3+ Regulatory T Cells and CD4+CD25-Foxp3+ T Cells in Psoriasis
- Regulation of Innate Immunity via MHC Class II-mediated Signaling; Non-classical Role of MHC Class II in Innate Immunity