Immune Netw.
2011 Feb;11(1):79-94. 10.4110/in.2011.11.1.79.
Deoxypodophyllotoxin Induces a Th1 Response and Enhances the Antitumor Efficacy of a Dendritic Cell-based Vaccine
- Affiliations
-
- 1Department of Biology, College of Natural Sciences, Chosun University, Gwangju 501-759, Korea.
- 2Department of Microbiology and Immunology, School of Medicine, Pusan National University, Yangsan 626-770, Korea. immunpym@pusan.ac.kr
- 3Rangos Research Center, Children's Hospital of Pittsburgh, Department of Pediatrics, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania 15213, USA.
- 4Department of Surgery, Busan Paik Hospital, Inje University, College of Medicine, Busan 614-735, Korea.
- 5Yongsan Hospital College of Medicine Chung-Ang University, Seoul 140-757, Korea.
- 6Department of Pharmacology, College of Medicine, Chung-Ang University, Seoul 156-756, Korea.
- KMID: 2150695
- DOI: http://doi.org/10.4110/in.2011.11.1.79
Abstract
- BACKGROUND
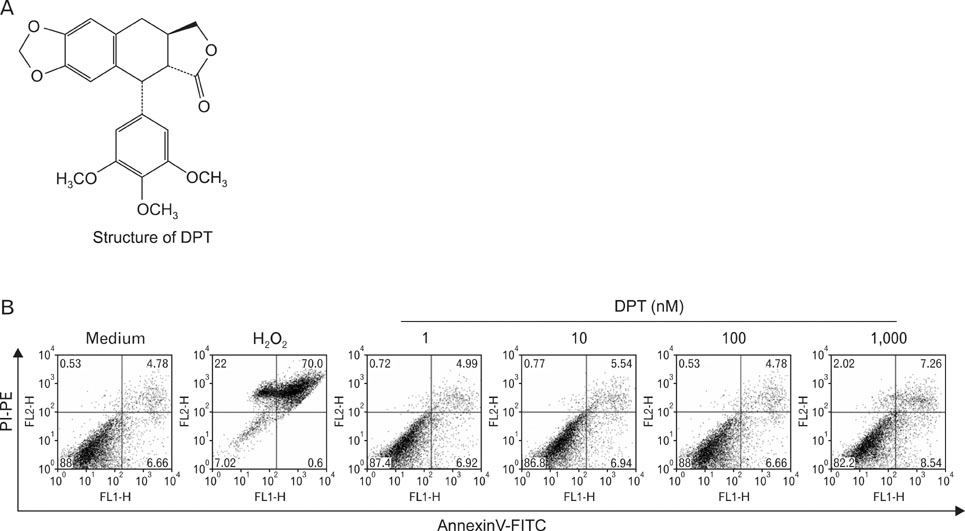
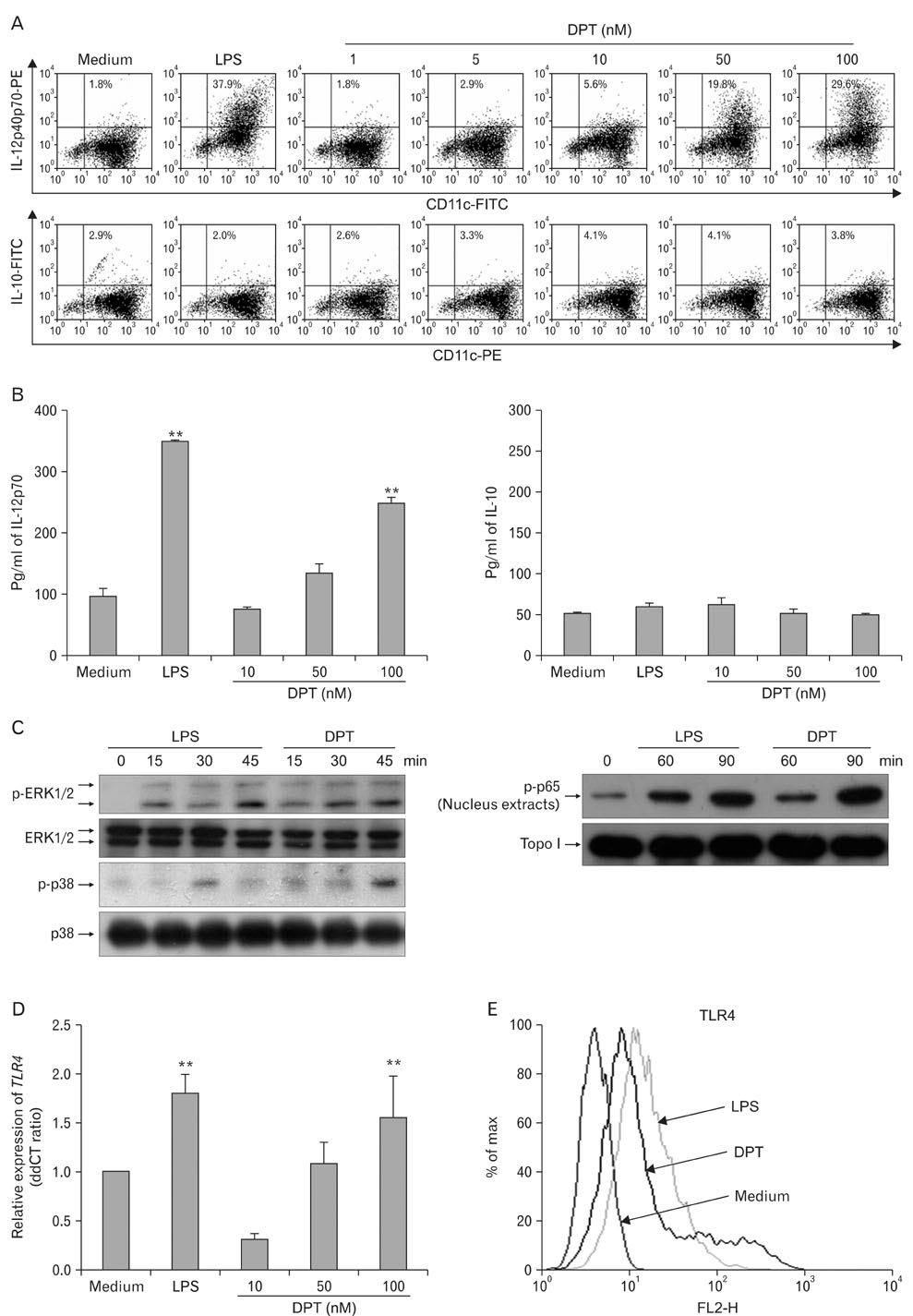
Dendritic cell (DC)-based vaccines are currently being evaluated as a novel strategy for tumor vaccination and immunotherapy. However, inducing long-term regression in established tumor-implanted mice is difficult. Here, we show that deoxypohophyllotoxin (DPT) induces maturation and activation of bone marrow-derived DCs via Toll-like receptor (TLR) 4 activation of MAPK and NF-kappaB.
METHODS
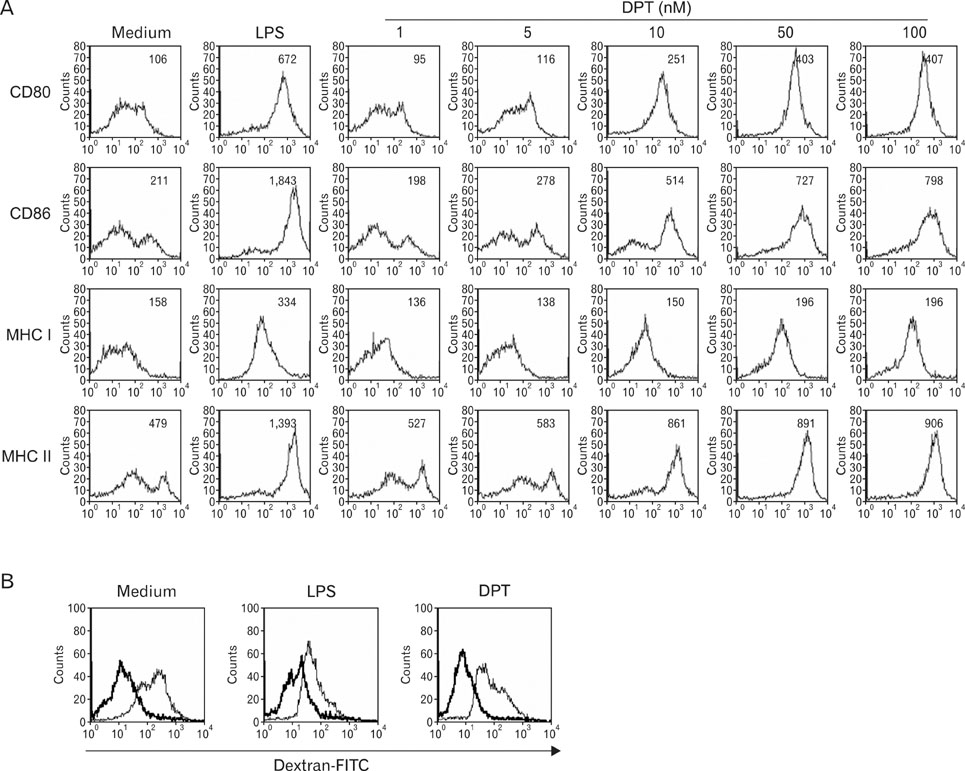
The phenotypic and functional maturation of DPT-treated DCs was assessed by flow cytometric analysis and cytokine production, respectively. DPT-treated DCs was also used for mixed leukocyte reaction to evaluate T cell-priming capacity and for tumor regression against melanoma.
RESULTS
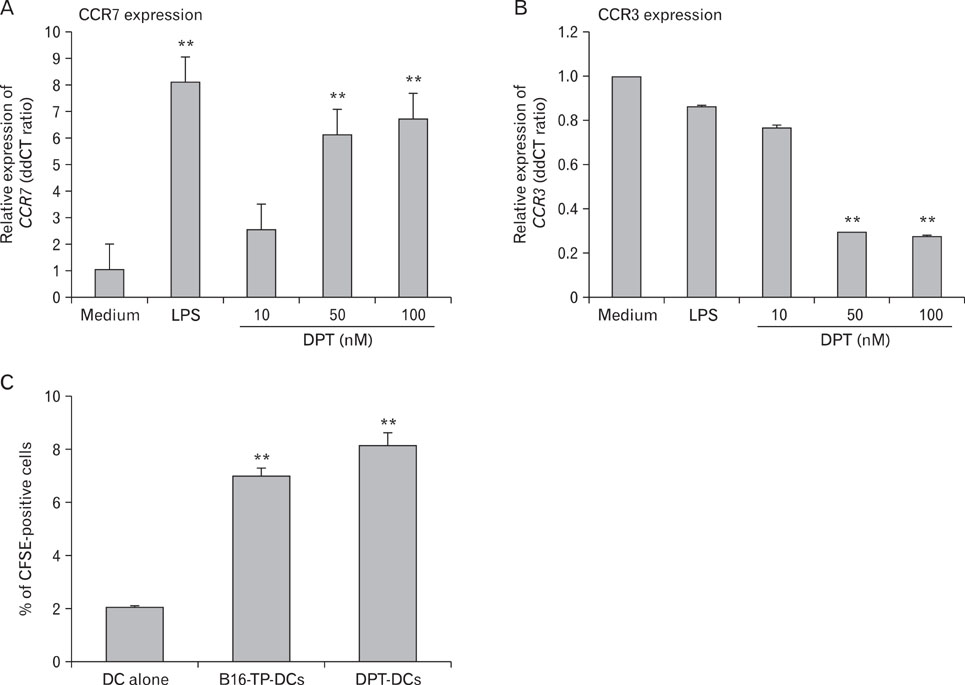
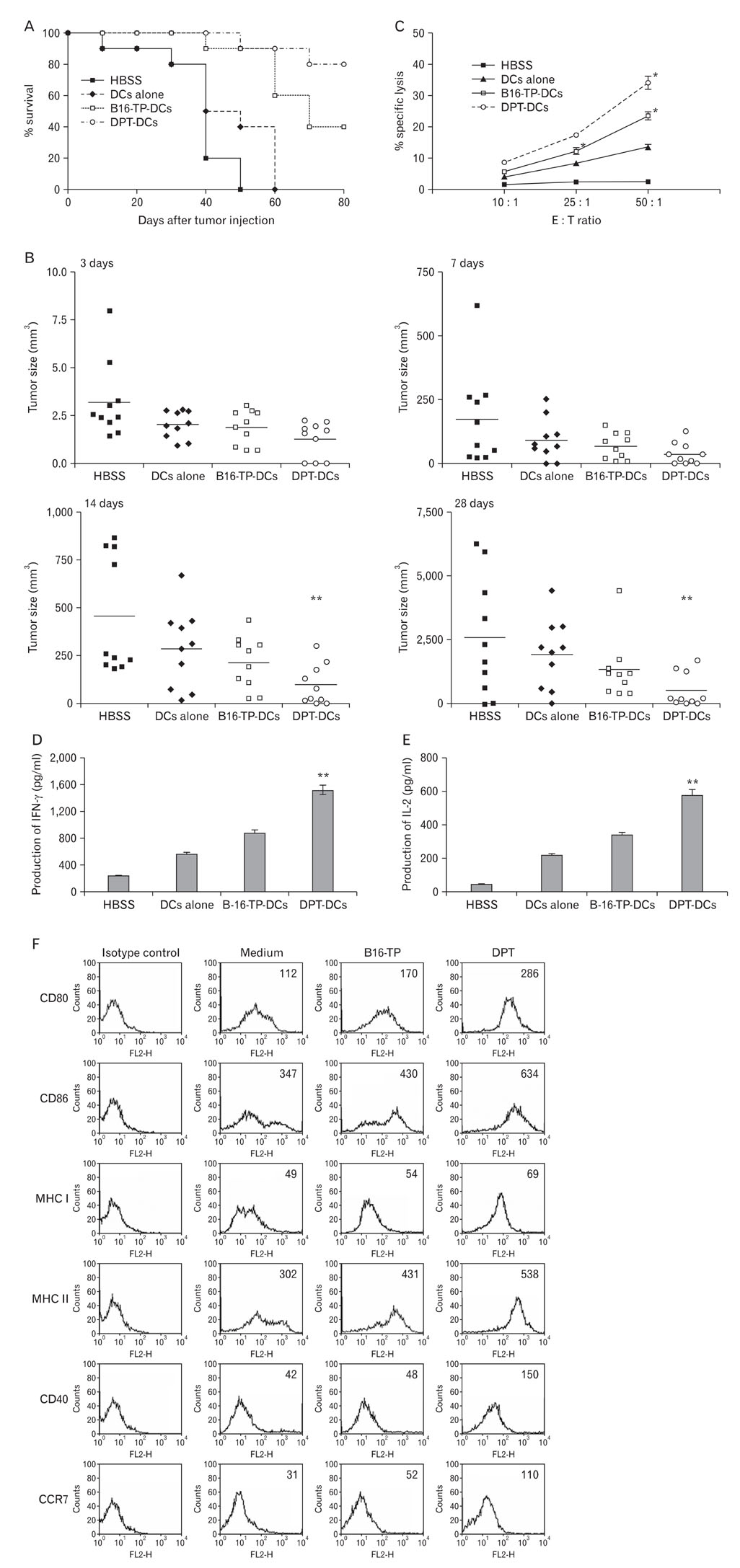
DPT promoted the activation of CD8+ T cells and the Th1 immune response by inducing IL-12 production in DCs. In a B16F10 melanoma-implanted mouse model, we demonstrated that DPT-treated DCs (DPT-DCs) enhance immune priming and regression of an established tumor in vivo. Furthermore, migration of DPT-DCs to the draining lymph nodes was induced via CCR7 upregulation. Mice that received DPT-DCs displayed enhanced antitumor therapeutic efficacy, which was associated with increased IFN-gamma production and induction of cytotoxic T lymphocyte activity.
CONCLUSION
These findings strongly suggest that the adjuvant effect of DPT in DC vaccination is associated with the polarization of T effector cells toward a Th1 phenotype and provides a potential therapeutic antitumor immunity.
MeSH Terms
Figure
Cited by 1 articles
-
Nanoparticle-Based Vaccine Delivery for Cancer Immunotherapy
Yeong-Min Park, Seung Jun Lee, Young Seob Kim, Moon Hee Lee, Gil Sun Cha, In Duk Jung, Tae Heung Kang, Hee Dong Han
Immune Netw. 2013;13(5):177-183. doi: 10.4110/in.2013.13.5.177.
Reference
-
1. Tobiásová Z, Pospísilová D, Miller AM, Minárik I, Sochorová K, Spísek R, Rob L, Bartůnková J. In vitro assessment of dendritic cells pulsed with apoptotic tumor cells as a vaccine for ovarian cancer patients. Clin Immunol. 2007. 122:18–27.
Article2. Bennett SR, Carbone FR, Karamalis F, Miller JF, Heath WR. Induction of a CD8+ cytotoxic T lymphocyte response by cross-priming requires cognate CD4+ T cell help. J Exp Med. 1997. 186:65–70.
Article3. Steinman RM. The dendritic cell system and its role in immunogenicity. Annu Rev Immunol. 1991. 9:271–296.
Article4. Morisaki T, Matsumoto K, Onishi H, Kuroki H, Baba E, Tasaki A, Kubo M, Nakamura M, Inaba S, Yamaguchi K, Tanaka M, Katano M. Dendritic cell-based combined immunotherapy with autologous tumor-pulsed dendritic cell vaccine and activated T cells for cancer patients: rationale, current progress, and perspectives. Hum Cell. 2003. 16:175–182.
Article5. Rock KL, Clark K. Analysis of the role of MHC class II presentation in the stimulation of cytotoxic T lymphocytes by antigens targeted into the exogenous antigen-MHC class I presentation pathway. J Immunol. 1996. 156:3721–3726.6. Salio M, Cerundolo V, Lanzavecchia A. Dendritic cell maturation is induced by mycoplasma infection but not by necrotic cells. Eur J Immunol. 2000. 30:705–708.
Article7. Shimizu K, Thomas EK, Giedlin M, Mulé JJ. Enhancement of tumor lysate- and peptide-pulsed dendritic cell-based vaccines by the addition of foreign helper protein. Cancer Res. 2001. 61:2618–2624.8. Jeannin P, Renno T, Goetsch L, Miconnet I, Aubry JP, Delneste Y, Herbault N, Baussant T, Magistrelli G, Soulas C, Romero P, Cerottini JC, Bonnefoy JY. OmpA targets dendritic cells, induces their maturation and delivers antigen into the MHC class I presentation pathway. Nat Immunol. 2000. 1:502–509.
Article9. Sloan JM, Kershaw MH, Touloukian CE, Lapointe R, Robbins PF, Restifo NP, Hwu P. MHC class I and class II presentation of tumor antigen in retrovirally and adenovirally transduced dendritic cells. Cancer Gene Ther. 2002. 9:946–950.
Article10. Albert ML, Sauter B, Bhardwaj N. Dendritic cells acquire antigen from apoptotic cells and induce class I-restricted CTLs. Nature. 1998. 392:86–89.
Article11. You Z, Hester J, Rollins L, Spagnoli GC, van der Bruggen P, Chen SY. A retrogen strategy for presentation of an intracellular tumor antigen as an exogenous antigen by dendritic cells induces potent antitumor T helper and CTL responses. Cancer Res. 2001. 61:197–205.12. Celluzzi CM, Mayordomo JI, Storkus WJ, Lotze MT, Falo LD Jr. Peptide-pulsed dendritic cells induce antigen-specific CTL-mediated protective tumor immunity. J Exp Med. 1996. 183:283–287.
Article13. Huang J, Tatsumi T, Pizzoferrato E, Vujanovic N, Storkus WJ. Nitric oxide sensitizes tumor cells to dendritic cell-mediated apoptosis, uptake, and cross-presentation. Cancer Res. 2005. 65:8461–8470.
Article14. Liu Y, Bi X, Xu S, Xiang J. Tumor-infiltrating dendritic cell subsets of progressive or regressive tumors induce suppressive or protective immune responses. Cancer Res. 2005. 65:4955–4962.
Article15. Jarnicki AG, Conroy H, Brereton C, Donnelly G, Toomey D, Walsh K, Sweeney C, Leavy O, Fletcher J, Lavelle EC, Dunne P, Mills KH. Attenuating regulatory T cell induction by TLR agonists through inhibition of p38 MAPK signaling in dendritic cells enhances their efficacy as vaccine adjuvants and cancer immunotherapeutics. J Immunol. 2008. 180:3797–3806.
Article16. Zanoni I, Foti M, Ricciardi-Castagnoli P, Granucci F. TLR-dependent activation stimuli associated with Th1 responses confer NK cell stimulatory capacity to mouse dendritic cells. J Immunol. 2005. 175:286–292.
Article17. Kane CM, Cervi L, Sun J, McKee AS, Masek KS, Shapira S, Hunter CA, Pearce EJ. Helminth antigens modulate TLR-initiated dendritic cell activation. J Immunol. 2004. 173:7454–7461.
Article18. Krieg AM. Therapeutic potential of Toll-like receptor 9 activation. Nat Rev Drug Discov. 2006. 5:471–484.
Article19. Chan CW, Crafton E, Fan HN, Flook J, Yoshimura K, Skarica M, Brockstedt D, Dubensky TW, Stins MF, Lanier LL, Pardoll DM, Housseau F. Interferon-producing killer dendritic cells provide a link between innate and adaptive immunity. Nat Med. 2006. 12:207–213.
Article20. Kapsenberg ML. Dendritic-cell control of pathogen-driven T-cell polarization. Nat Rev Immunol. 2003. 3:984–993.
Article21. Lee JS, Lee JC, Lee CM, Jung ID, Jeong YI, Seong EY, Chung HY, Park YM. Outer membrane protein A of Acinetobacter baumannii induces differentiation of CD4+ T cells toward a Th1 polarizing phenotype through the activation of dendritic cells. Biochem Pharmacol. 2007. 74:86–97.
Article22. Sivori S, Falco M, Della Chiesa M, Carlomagno S, Vitale M, Moretta L, Moretta A. CpG and double-stranded RNA trigger human NK cells by Toll-like receptors: induction of cytokine release and cytotoxicity against tumors and dendritic cells. Proc Natl Acad Sci U S A. 2004. 101:10116–10121.
Article23. Kim Y, Kim SB, You YJ, Ahn BZ. Deoxypodophyllotoxin: the cytotoxic and antiangiogenic component from Pulsatilla koreana. Planta Med. 2002. 68:271–274.
Article24. Sudo K, Konno K, Shigeta S, Yokota T. Inhibitory effects of podophyllotoxin derivatives on herpes simplex virus replication. Antivir Chem Chemother. 1998. 9:263–267.
Article25. Jin M, Moon TC, Quan Z, Lee E, Kim YK, Yang JH, Suh SJ, Jeong TC, Lee SH, Kim CH, Chang HW. The naturally occurring flavolignan, deoxypodophyllotoxin, inhibits lipopolysaccharide-induced iNOS expression through the NF-kappaB activation in RAW264.7 macrophage cells. Biol Pharm Bull. 2008. 31:1312–1315.
Article26. Lyons AB. Analysing cell division in vivo and in vitro using flow cytometric measurement of CFSE dye dilution. J Immunol Methods. 2000. 243:147–154.
Article27. Préville X, Ladant D, Timmerman B, Leclerc C. Eradication of established tumors by vaccination with recombinant Bordetella pertussis adenylate cyclase carrying the human papillomavirus 16 E7 oncoprotein. Cancer Res. 2005. 65:641–649.28. Abe M, Zahorchak AF, Colvin BL, Thomson AW. Migratory responses of murine hepatic myeloid, lymphoid-related, and plasmacytoid dendritic cells to CC chemokines. Transplantation. 2004. 78:762–765.
Article29. Mosca PJ, Hobeika AC, Clay TM, Nair SK, Thomas EK, Morse MA, Lyerly HK. A subset of human monocyte-derived dendritic cells expresses high levels of interleukin-12 in response to combined CD40 ligand and interferon-gamma treatment. Blood. 2000. 96:3499–3504.
Article30. Langenkamp A, Messi M, Lanzavecchia A, Sallusto F. Kinetics of dendritic cell activation: impact on priming of TH1, TH2 and nonpolarized T cells. Nat Immunol. 2000. 1:311–316.
Article31. Trinchieri G. Interleukin-12: a proinflammatory cytokine with immunoregulatory functions that bridge innate resistance and antigen-specific adaptive immunity. Annu Rev Immunol. 1995. 13:251–276.
Article32. Lutz MB, Assmann CU, Girolomoni G, Ricciardi-Castagnoli P. Different cytokines regulate antigen uptake and presentation of a precursor dendritic cell line. Eur J Immunol. 1996. 26:586–594.
Article33. An H, Yu Y, Zhang M, Xu H, Qi R, Yan X, Liu S, Wang W, Guo Z, Guo J, Qin Z, Cao X. Involvement of ERK, p38 and NF-kappaB signal transduction in regulation of TLR2, TLR4 and TLR9 gene expression induced by lipopolysaccharide in mouse dendritic cells. Immunology. 2002. 106:38–45.
Article34. MacDonald KP, Kuns RD, Rowe V, Morris ES, Banovic T, Bofinger H, O'Sullivan B, Markey KA, Don AL, Thomas R, Hill GR. Effector and regulatory T-cell function is differentially regulated by RelB within antigen-presenting cells during GVHD. Blood. 2007. 109:5049–5057.
Article35. Sioud M, Fløisand Y. TLR agonists induce the differentiation of human bone marrow CD34+ progenitors into CD11c+ CD80/86+ DC capable of inducing a Th1-type response. Eur J Immunol. 2007. 37:2834–2846.
Article36. Sweet CR, Conlon J, Golenbock DT, Goguen J, Silverman N. YopJ targets TRAF proteins to inhibit TLR-mediated NF-kappaB, MAPK and IRF3 signal transduction. Cell Microbiol. 2007. 9:2700–2715.
Article37. Guiducci C, Ghirelli C, Marloie-Provost MA, Matray T, Coffman RL, Liu YJ, Barrat FJ, Soumelis V. PI3K is critical for the nuclear translocation of IRF-7 and type I IFN production by human plasmacytoid predendritic cells in response to TLR activation. J Exp Med. 2008. 205:315–322.
Article38. Ato M, Stäger S, Engwerda CR, Kaye PM. Defective CCR7 expression on dendritic cells contributes to the development of visceral leishmaniasis. Nat Immunol. 2002. 3:1185–1191.
Article39. Catusse J, Spinks J, Mattick C, Dyer A, Laing K, Fitzsimons C, Smit MJ, Gompels UA. Immunomodulation by herpesvirus U51A chemokine receptor via CCL5 and FOG-2 down-regulation plus XCR1 and CCR7 mimicry in human leukocytes. Eur J Immunol. 2008. 38:763–777.
Article40. Movassagh M, Spatz A, Davoust J, Lebecque S, Romero P, Pittet M, Rimoldi D, Liénard D, Gugerli O, Ferradini L, Robert C, Avril MF, Zitvogel L, Angevin E. Selective accumulation of mature DC-Lamp+ dendritic cells in tumor sites is associated with efficient T-cell-mediated antitumor response and control of metastatic dissemination in melanoma. Cancer Res. 2004. 64:2192–2198.
Article41. Sancho D, Mourão-Sá D, Joffre OP, Schulz O, Rogers NC, Pennington DJ, Carlyle JR, Reis e Sousa C. Tumor therapy in mice via antigen targeting to a novel, DC-restricted C-type lectin. J Clin Invest. 2008. 118:2098–2110.
Article42. Melief CJ. Cancer immunotherapy by dendritic cells. Immunity. 2008. 29:372–383.
Article43. Hatfield P, Merrick AE, West E, O'Donnell D, Selby P, Vile R, Melcher AA. Optimization of dendritic cell loading with tumor cell lysates for cancer immunotherapy. J Immunother. 2008. 31:620–632.
Article44. Okada N, Saito T, Masunaga Y, Tsukada Y, Nakagawa S, Mizuguchi H, Mori K, Okada Y, Fujita T, Hayakawa T, Mayumi T, Yamamoto A. Efficient antigen gene transduction using Arg-Gly-Asp fiber-mutant adenovirus vectors can potentiate antitumor vaccine efficacy and maturation of murine dendritic cells. Cancer Res. 2001. 61:7913–7919.45. Lee JS, Kim JW, Choi CH, Lee WK, Chung HY, Lee JC. Anti-tumor activity of Acinetobacter baumannii outer membrane protein A on dendritic cell-based immunotherapy against murine melanoma. J Microbiol. 2008. 46:221–227.
Article46. Usluoglu N, Pavlovic J, Moelling K, Radziwill G. RIP2 mediates LPS-induced p38 and IkappaBalpha signaling including IL-12 p40 expression in human monocyte-derived dendritic cells. Eur J Immunol. 2007. 37:2317–2325.
Article47. Kelsall BL, Stüber E, Neurath M, Strober W. Interleukin-12 production by dendritic cells. The role of CD40-CD40L interactions in Th1 T-cell responses. Ann N Y Acad Sci. 1996. 795:116–126.48. Iwashita Y, Goto S, Tominaga M, Sasaki A, Ohmori N, Goto T, Sato S, Ohta M, Kitano S. Dendritic cell immunotherapy with poly (D,L-2,4-diaminobutyric acid)-mediated intratumoral delivery of the interleukin-12 gene suppresses tumor growth significantly. Cancer Sci. 2005. 96:303–307.
Article49. Takaoka A, Yanai H, Kondo S, Duncan G, Negishi H, Mizutani T, Kano S, Honda K, Ohba Y, Mak TW, Taniguchi T. Integral role of IRF-5 in the gene induction programme activated by Toll-like receptors. Nature. 2005. 434:243–249.
Article50. Ridge JP, Di Rosa F, Matzinger P. A conditioned dendritic cell can be a temporal bridge between a CD4+ T-helper and a T-killer cell. Nature. 1998. 393:474–478.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- B Cells Promote Th1- Skewed NKT Cell Response by CD1d-TCR Interaction
- The Effectiveness of IL-12 Administration and Fusion on Tumor Antigen Sensitization Methods for Dendritic Cells Derived from Patients with Myelogenous Leukemia
- Regulation of Th2 Cell Immunity by Dendritic Cells
- BCG Immunomodulation Therapy in Asthma
- Strategies to Improve Dendritic Cell-based Immunotherapy against Cancer